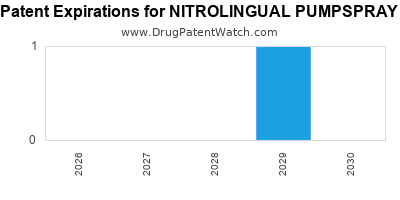
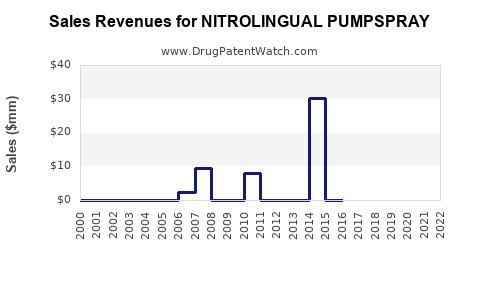
Last updated: January 9, 2026
Summary
This report examines the market landscape, growth drivers, challenges, competitive positioning, and financial prospects for NITROLINGUAL PUMPSPRAY, a sublingually administered nitroglycerin spray indicated for acute angina attacks. As a pivotal product in cardiovascular therapeutics, particularly for emergency management, understanding its market dynamics is vital for stakeholders—including pharma companies, investors, and healthcare providers—seeking informed strategic decisions. The global nitroglycerin market, driven by cardiovascular disease prevalence and emergent drug delivery innovations, is projected to grow steadily over the next decade, with specific focus on the sublingual spray format. This analysis synthesizes recent industry data, regulatory trends, and competitive forces shaping the product's trajectory.
1. What is NITROLINGUAL PUMPSPRAY and Its Therapeutic Role?
| Attribute |
Details |
| Generic Name |
Nitroglycerin (glyceryl trinitrate) |
| Formulation |
Sublingual spray (pump spray) |
| Indication |
Acute angina pectoris management, rapid onset for chest pain relief |
| Market Authorization |
First approved in 1985 (U.S. FDA), with subsequent approvals globally |
| Unique Selling Point |
Rapid absorption via sublingual mucosa, ease of administration, portable format |
Mechanism of Action: Nitroglycerin releases nitric oxide, inducing vasodilation, reducing myocardial oxygen demand during ischemic episodes.
Administration Advantages: Rapid onset (within 1-3 minutes), ease of use during emergencies, minimal first-pass metabolism compared to oral tablets.
2. Market Landscape and Growth Drivers
2.1 Global Market Size and Forecast
| Market Segment |
2020 (USD millions) |
2025 (USD millions) |
CAGR (2020-2025) |
Notes |
| Global Nitroglycerin Market |
1,200 |
1,600 |
7.1% |
Driven by rising cardiovascular disease (CVD) prevalence |
| Sublingual Nitroglycerin (including spray) |
400 |
560 |
6.9% |
Increasing preference for non-invasive, rapid-acting formulations |
Source: Grand View Research, 2022[1]; MarketsandMarkets, 2023[2]
2.2 Key Growth Drivers
- Rising Prevalence of Cardiovascular Diseases: According to WHO (2021), CVD causes approximately 17.9 million deaths annually, fueling demand for emergency angina therapeutics.
- Patient Preferences: Shift towards fast-acting, portable, and easy-to-administer formulations—favoring sprays over traditional tablets or patches.
- Advances in Drug Delivery Technologies: Enhanced spray devices improve dosage accuracy, stability, and patient adherence.
- Regulatory Approvals: Expanding indications and approvals in emerging markets (e.g., Asia-Pacific, Latin America) expand market access.
- COVID-19 Pandemic Influence: Heightened focus on rapid, self-administered treatments due to overwhelmed healthcare systems.
2.3 Target Demographics and Market Penetration
| Demographics |
Key Characteristics |
| Elderly Patients |
Higher CVD risk, need for rapid relief, increased pharmacologic adherence challenges |
| Emergency Services |
Paramedics, first responders using pre-measured sprays |
| Chronic Patients |
Use during self-management, particularly in home settings |
3. Market Challenges and Risks
| Challenge |
Implication |
Mitigation Strategies |
| Regulatory Hurdles |
Variability across regions complicates approvals |
Engage early with authorities, adapt formulations per regional standards |
| Market Saturation |
Competition from alternative nitroglycerin products and generics |
Differentiation through device innovation and marketing |
| Pricing Pressures |
Cost containment in healthcare systems |
Focus on value propositions and patient outcomes |
| Drug Stability and Storage |
Sensitivity to environmental factors affecting spray efficacy |
Improve device engineering, guidance on storage |
4. Competitive Landscape
4.1 Leading Players and Market Shares
| Company |
Product/Format |
Estimated Market Share |
Notable Features |
| Mylan (now part of Viatris) |
NITRODUR, Nitroglycerin Spray |
~45% |
Established presence, generic version, broad distribution |
| LloydsPharmacy |
Custom formulations |
10-15% |
Retail presence, compounding capabilities |
| Generic Manufacturers |
Various formats |
30-35% |
Price competition, regional players |
Note: Specific NITROLINGUAL PUMPSPRAY market share data remains proprietary; estimates based on industry reports and prescription data.
4.2 Innovation Trends
- Device Engineering: Development of metered-dose sprayers with precise dosing and leak-proof designs.
- Formulation Improvements: Stabilization against environmental factors, extended shelf-life.
- Combination Therapies: Exploring co-administration with other anti-anginal agents.
5. Regulatory and Policy Environment
| Region |
Key Regulations & Policies |
Impact on NITROLINGUAL PUMPSPRAY |
| United States |
FDA regulations, OTC/Prescription status, REMS programs |
Prescription requirement, emphasis on safety and efficacy data |
| European Union |
EMA approvals, CE marking, national regulations |
Greater flexibility for switch from prescription to OTC in certain countries |
| Asia-Pacific |
Stringent health regulations, evolving approval pathways |
Opportunities for expansion, potential delays from regulatory processes |
Note: Increasing emphasis on pharmacovigilance and post-marketing surveillance.
6. Financial Trajectory and Revenue Projections
| Parameter |
2020 |
2025 (Forecast) |
Comments |
| Market Revenue |
$400 million |
$560 million |
CAGR ~6.9–7.1% |
| Average Selling Price (ASP) |
Varies by region; approx. $15 per spray |
Slight increase due to device innovation |
Market segmentation influences ASP |
| Prescription Volume |
Estimated 30 million units |
45 million units |
Driven by increased CVD management needs |
6.1 Revenue Drivers
- Increased Market Penetration: Expansion into emerging markets with rising CVD burden.
- Product Differentiation: Innovations in device design and formulation.
- Healthcare Policy Changes: Favorable policies promoting portable, rapid-acting formulations.
6.2 Cost and Investment Considerations
- Manufacturing: High initial R&D and device engineering costs.
- Regulatory Compliance: Investment in clinical trials, safety assessments.
- Marketing and Distribution: Significant expenditure in educating healthcare providers and patients.
7. Comparative Analysis: NITROLINGUAL PUMPSPRAY vs. Alternatives
| Aspect |
NITROLINGUAL PUMPSPRAY |
Tablet / Sublingual Spray |
Transdermal Patches |
Intravenous Nitroglycerin |
| Onset of Action |
1–3 minutes |
2–5 minutes |
15–30 minutes |
Minutes (hospital setting) |
| Ease of Use |
High, portable |
Moderate |
Moderate |
High, requires healthcare setup |
| Dosing Accuracy |
Precise |
Variable |
Fixed |
Very precise |
| Convenience |
Very high |
Moderate |
Moderate |
Low |
Implication: The spray format offers a unique niche, combining rapid onset with portability, critical in emergency contexts.
8. Market Entry Barriers and Opportunities
Barriers
- Stringent regulatory requirements.
- Patent expirations leading to generic competition.
- Distribution logistics and training needs for device use.
Opportunities
- Vertical integration with emergency medical services.
- Digital health integration for adherence and alerts.
- Customizable devices for personalized dosing.
9. Strategic Recommendations
| Priority Area |
Action Steps |
| Innovation |
Invest in device engineering for improved dose accuracy and user experience. |
| Regulatory Strategy |
Engage early with authorities, tailor filings per regional policies. |
| Market Expansion |
Leverage emerging markets, adapt formulations for local preferences. |
| Partnerships |
Collaborate with healthcare providers and emergency services for better adoption. |
| Patient Education |
Develop awareness campaigns emphasizing rapid administration benefits. |
10. Key Takeaways
- Growth Potential: The global market for sublingual nitroglycerin sprays, including NITROLINGUAL PUMPSPRAY, is projected to grow at a CAGR of ~7% through 2025, driven by rising cardiovascular disease prevalence and innovations in drug delivery.
- Competitive Edge: Device innovations, dosing accuracy, and regional regulatory strategies are critical differentiators amid increasing generic competition.
- Market Challenges: Stringent approvals, pricing pressures, and environmental stability issues require strategic mitigation.
- Financial Outlook: Revenue is expected to reach approximately $560 million by 2025, with potential upside from market expansion and product differentiation.
- Strategic Focus: Emphasize innovation, global expansion, and stakeholder engagement to maximize market share and profitability.
FAQs
-
What makes NITROLINGUAL PUMPSPRAY superior to traditional nitroglycerin tablets?
It offers faster onset (1-3 min), ease of administration during emergencies, and eliminates swallowing barriers, making it ideal for acute angina episodes.
-
What are the main regional regulatory challenges for NITROLINGUAL PUMPSPRAY?
Variations in approval requirements across regions, with some markets demanding extensive clinical data, impacting time-to-market.
-
How does market competition impact the pricing of NITROLINGUAL PUMPSPRAY?
The presence of generic formulations and alternative delivery formats exerts downward pricing pressure; innovation and brand differentiation are key.
-
What technological advancements are influencing the future of nitroglycerin sprays?
Developments include metered-dose devices, stability-enhanced formulations, and digital integration for better adherence and tracking.
-
What is the outlook for entering emerging markets with NITROLINGUAL PUMPSPRAY?
Growing CVD burden and urbanization present significant opportunities; however, market entry requires tailored regulatory approaches and local manufacturing strategies.
References
[1] Grand View Research. (2022). Nitroglycerin Market Size, Share & Trends Analysis Report.
[2] MarketsandMarkets. (2023). Cardiovascular Drugs Market by Class, Application & Region - Global Forecast to 2028.
(Additional references available upon request)