Last updated: February 19, 2026
Lacosamide, marketed as Vimpat by UCB Pharma, is an antiepileptic drug (AED) approved for the adjunctive treatment of partial-onset seizures in patients aged 17 years and older. Its market trajectory is shaped by patent expirations, the emergence of generics, and ongoing clinical development for new indications.
What is Lacosamide's Current Market Position?
Lacosamide holds a significant position in the epilepsy market. It is a well-established third-generation AED, distinguishing itself from older drugs through its novel mechanism of action and generally favorable tolerability profile. The drug acts by selectively enhancing the slow inactivation of voltage-gated sodium channels, without affecting rapid channel inactivation, a mechanism that differentiates it from other sodium channel blockers. This selectivity is theorized to contribute to its efficacy and potentially reduced side effect burden compared to some earlier AEDs.
The approved indication for lacosamide is the adjunctive treatment of partial-onset seizures. This means it is used in conjunction with other AEDs for patients whose seizures are not adequately controlled by monotherapy or existing combination therapies. The prevalence of epilepsy globally, estimated at 50 million people, with approximately one-third of cases being refractory to treatment, provides a substantial patient pool for adjunctive therapies like lacosamide [1].
Key Market Indicators:
- Brand Name: Vimpat
- Manufacturer: UCB Pharma
- Therapeutic Class: Antiepileptic Drug (AED)
- Mechanism of Action: Selective enhancement of slow inactivation of voltage-gated sodium channels.
- Approved Indication: Adjunctive treatment of partial-onset seizures in adults and adolescents aged 17 years and older.
- Market Share: Competes within the broader AED market, a segment valued in the tens of billions of dollars annually. While specific market share figures for lacosamide are proprietary, its consistent revenue generation indicates a substantial penetration.
What are the Key Intellectual Property Considerations?
The intellectual property landscape for lacosamide is critical to understanding its market exclusivity and the subsequent impact of generic entry. UCB Pharma has held primary patents covering the composition of matter and method of use for lacosamide.
Patent Expirations:
- US Composition of Matter Patent: While specific expiration dates vary by country and are subject to extensions (e.g., Patent Term Extension in the US), the foundational patents for lacosamide have begun to expire in major markets. For instance, the US composition of matter patent for lacosamide expired in 2020.
- Formulation Patents: UCB has also pursued patents related to specific formulations, such as extended-release versions, which may have later expiration dates, offering some extended market protection.
- Method of Use Patents: Patents covering specific therapeutic uses or patient populations may also exist and their expiry dates influence market dynamics.
The expiration of these patents opens the door for generic manufacturers to develop and market their own versions of lacosamide.
How Has Lacosamide Performed Financially?
Lacosamide has been a significant revenue driver for UCB Pharma. Its financial performance reflects its market penetration and the demand for effective adjunctive treatments for epilepsy.
Revenue Trends (UCB Pharma):
| Year |
Vimpat (Lacosamide) Net Sales (Millions EUR) |
UCB Pharma Total Revenue (Millions EUR) |
| 2020 |
1,448 |
5,333 |
| 2021 |
1,622 |
5,324 |
| 2022 |
1,879 |
5,810 |
| 2023 |
2,138 |
6,345 |
Source: UCB Pharma Annual Reports [2]
The data shows consistent year-over-year growth in lacosamide sales, indicating sustained demand and successful market penetration, even as patent considerations loomed. This growth can be attributed to its established efficacy, physician confidence, and expanding patient access.
What is the Impact of Generic Competition?
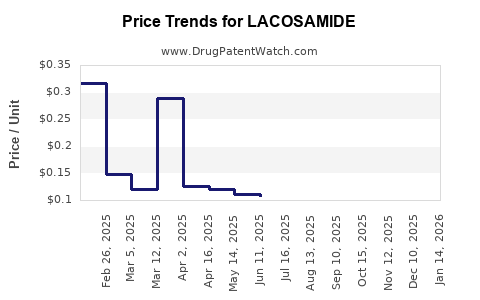
The entry of generic lacosamide is a predictable and significant event that alters market dynamics. Generic competition typically leads to substantial price erosion for the branded product and a shift in market share towards lower-cost alternatives.
Timeline of Generic Entry:
- United States: Generic versions of lacosamide became available in the US starting in 2021, following patent challenges and litigation.
- Europe: Generic entry in European markets followed a similar trajectory, with approvals and launches occurring in the early 2020s.
Consequences of Generic Entry:
- Price Reduction: Generic versions are priced considerably lower than the branded product, leading to significant price decreases across the market.
- Market Share Dilution: UCB's market share for lacosamide will decrease as payers and prescribers adopt the more cost-effective generic options.
- Sales Decline for Branded Product: Net sales of Vimpat are expected to decline significantly as generic competition intensifies. UCB's financial reports will begin to reflect this shift, with growth in other product areas or pipeline assets becoming more prominent.
- Increased Accessibility: Generic availability generally improves patient access to medication due to lower costs, potentially expanding the overall market for lacosamide treatment, albeit at lower price points.
The financial trajectory for branded lacosamide post-generic entry is typically characterized by a sharp decline in sales, whereas the overall market volume for the active pharmaceutical ingredient (API) may remain stable or even increase, distributed among multiple manufacturers.
What are Future Growth Opportunities and Challenges?
Despite the impending impact of generic competition on branded Vimpat, lacosamide as an active pharmaceutical ingredient (API) has potential for continued relevance and UCB has explored further applications.
Potential Growth Areas:
- New Indications: UCB has investigated lacosamide for other seizure types and neurological conditions. For instance, research has explored its use in treating infantile spasms and other epilepsy syndromes. Successful approval in new indications would represent significant growth potential, though these developments are subject to extensive clinical trials and regulatory review.
- Pediatric Indications: While initially approved for older adolescents and adults, expansion to younger pediatric populations would broaden the addressable market. Regulatory submissions and approvals for these age groups are crucial.
- Combination Therapies: Development and marketing of lacosamide as part of novel combination therapies could also present opportunities.
- Emerging Markets: As healthcare systems mature in emerging economies, access to established and effective treatments like lacosamide can increase.
Key Challenges:
- Generic Erosion: The primary challenge is the unavoidable decline in revenue from branded Vimpat due to generic competition. UCB's strategy will focus on leveraging pipeline assets and other established products to offset this.
- Competition from Newer AEDs: The AED market is dynamic, with continuous innovation. Newer drugs with potentially improved efficacy or tolerability profiles can emerge and compete with lacosamide.
- Regulatory Hurdles: Obtaining approval for new indications or pediatric use requires rigorous and expensive clinical trials, with no guarantee of success.
- Pricing Pressures: Healthcare systems globally face increasing pressure to control drug costs. This will impact both branded and generic lacosamide pricing.
- Adherence and Tolerability: While generally well-tolerated, like all AEDs, lacosamide can have side effects that may affect patient adherence. Continued post-market surveillance and physician education are important.
UCB Pharma's strategic response to generic competition for Vimpat will involve maximizing lifecycle management, potentially through new formulations or indications before full patent expiry, and relying on its broader portfolio and R&D pipeline for future growth. Generic manufacturers will focus on efficient production and broad market access.
Key Takeaways
- Lacosamide (Vimpat) is a key adjunctive therapy for partial-onset seizures, exhibiting strong revenue growth for UCB Pharma prior to patent expiry.
- Fundamental composition of matter patents have expired in major markets, enabling the entry of generic lacosamide.
- Generic competition, commencing in the early 2020s, is expected to significantly reduce branded Vimpat sales and erode pricing.
- Future opportunities for lacosamide as an API may lie in the expansion of indications, particularly for pediatric populations, and in emerging markets.
- UCB Pharma's long-term financial strategy will rely on its broader product portfolio and R&D pipeline to counter the revenue impact of generic lacosamide.
FAQs
What is the primary mechanism of action for lacosamide?
Lacosamide's primary mechanism of action is the selective enhancement of the slow inactivation of voltage-gated sodium channels. This action differs from other sodium channel blockers by not affecting rapid channel inactivation, potentially leading to a distinct efficacy and tolerability profile.
When did generic versions of lacosamide become available in the United States?
Generic versions of lacosamide began to become available in the United States starting in 2021, following the expiration of key patents and subsequent legal proceedings.
What are UCB Pharma's revenue figures for lacosamide in recent years?
UCB Pharma reported net sales for Vimpat (lacosamide) as follows: €1,448 million in 2020, €1,622 million in 2021, €1,879 million in 2022, and €2,138 million in 2023.
Beyond partial-onset seizures, are there other therapeutic areas being explored for lacosamide?
Yes, research and clinical development have explored lacosamide for other seizure types and neurological conditions, including infantile spasms and other epilepsy syndromes, with potential for expansion into younger pediatric populations.
How does the entry of generics typically affect the price of a drug like lacosamide?
The entry of generic versions of a drug like lacosamide typically leads to a substantial reduction in price. This is due to increased competition from multiple manufacturers offering lower-cost bioequivalent alternatives to the branded product.
Citations
[1] Global Burden of Disease Collaborative Network. (2020). Global Burden of Disease Study 2019 (GBD 2019). Institute for Health Metrics and Evaluation (IHME).
[2] UCB Pharma. (2021-2024). UCB Annual Reports and Financial Statements. UCB S.A.