DICLOFENAC Drug Patent Profile
✉ Email this page to a colleague
When do Diclofenac patents expire, and when can generic versions of Diclofenac launch?
Diclofenac is a drug marketed by Aurobindo Pharma Ltd, Bionpharma, Strides Pharma, Alkem Labs Ltd, Annora Pharma, Par Form, Chartwell Rx, Novast Labs, Rk Pharma, Rubicon, Senores Pharms, Sun Pharm Industries, Teva, Umedica, Watson Labs Teva, Actavis Mid Atlantic, Alembic, Amneal, Amneal Pharms, Aurolife Pharma Llc, Cipla, Encube, Glenmark Pharms Ltd, Hikma, Padagis Israel, Perrigo Pharma Intl, Taro, Akorn, Altaire Pharms Inc, Bausch And Lomb, Falcon Pharms, Rising, Sandoz, Apotex, Epic Pharma Llc, Lupin Ltd, Lupin Pharms, Novel Labs Inc, Pai Holdings Pharm, Twi Pharms, Watson Labs Inc, Zydus Lifesciences, Actavis Elizabeth, Aurobindo Pharma Usa, Carlsbad, Micro Labs, Pliva, Roxane, Teva Pharms, Unique, Dexcel Ltd, Riconpharma Llc, Vpna, Actavis Labs Fl Inc, Exela Holdings, Yung Shin Pharm, and Zydus Pharms. and is included in seventy-eight NDAs.
The generic ingredient in DICLOFENAC is diclofenac sodium; misoprostol. There are forty-seven drug master file entries for this compound. Eleven suppliers are listed for this compound. Additional details are available on the diclofenac sodium; misoprostol profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Diclofenac
A generic version of DICLOFENAC was approved as diclofenac sodium; misoprostol by ACTAVIS LABS FL INC on July 9th, 2012.
Summary for DICLOFENAC
| US Patents: | 0 |
| Applicants: | 57 |
| NDAs: | 78 |
| Formulation / Manufacturing: | see details |
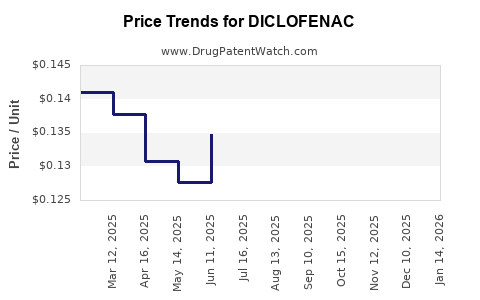
| Drug Prices: | Drug price information for DICLOFENAC |
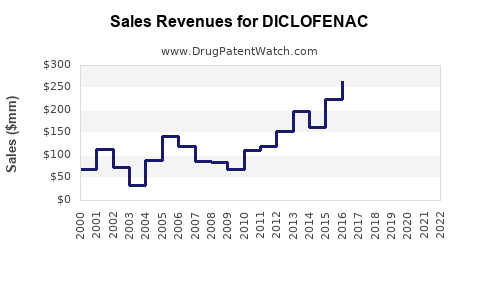
| Drug Sales Revenues: | Drug sales revenues for DICLOFENAC |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for DICLOFENAC |
| What excipients (inactive ingredients) are in DICLOFENAC? | DICLOFENAC excipients list |
| DailyMed Link: | DICLOFENAC at DailyMed |
See drug prices for DICLOFENAC
Recent Clinical Trials for DICLOFENAC
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Trauma Center | N/A |
| ZetrOZ, Inc. | Phase 1 |
| Daré Bioscience, Inc. | Phase 1 |
Medical Subject Heading (MeSH) Categories for DICLOFENAC
Paragraph IV (Patent) Challenges for DICLOFENAC
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ZORVOLEX | Capsules | diclofenac | 18 mg and 35 mg | 204592 | 1 | 2014-06-06 |
US Patents and Regulatory Information for DICLOFENAC
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rubicon | DICLOFENAC SODIUM | diclofenac sodium | TABLET, DELAYED RELEASE;ORAL | 216548-001 | May 11, 2023 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Zydus Pharms | DICLOFENAC SODIUM AND MISOPROSTOL | diclofenac sodium; misoprostol | TABLET, DELAYED RELEASE;ORAL | 206771-002 | Jun 12, 2023 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Apotex | DICLOFENAC SODIUM | diclofenac sodium | SOLUTION;TOPICAL | 202027-001 | May 27, 2014 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bausch And Lomb | DICLOFENAC SODIUM | diclofenac sodium | SOLUTION/DROPS;OPHTHALMIC | 078792-001 | Dec 28, 2007 | AT | RX | No | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


