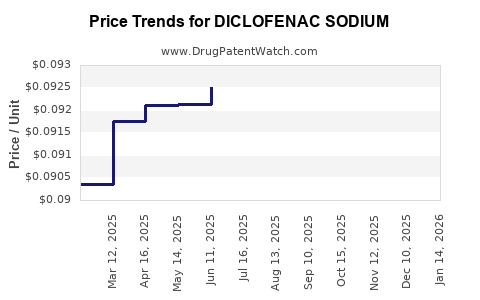
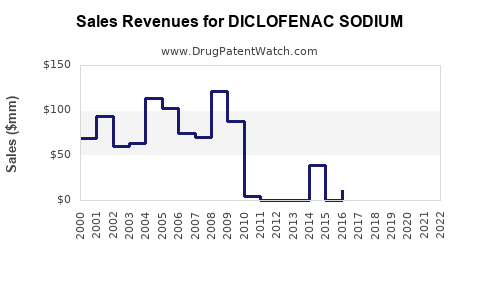
Last updated: February 19, 2026
What is the Global Market Size and Growth Rate for Diclofenac Sodium?
The global market for diclofenac sodium is substantial and projected to experience moderate growth. In 2022, the market was valued at approximately $2.2 billion. Analysts forecast a compound annual growth rate (CAGR) of 4.1% from 2023 to 2030, reaching an estimated $3.0 billion by the end of the forecast period [1]. This growth is driven by the drug's established efficacy in treating pain and inflammation, its wide availability, and its use in managing chronic conditions like osteoarthritis and rheumatoid arthritis [2].
Who are the Key Manufacturers and What is Their Market Share?
The diclofenac sodium market is characterized by a mix of large pharmaceutical corporations and numerous generic manufacturers. Competition is significant, particularly in the generic segment. Key players include:
- Novartis AG
- Pfizer Inc.
- Bayer AG
- Sun Pharmaceutical Industries Ltd.
- Teva Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- GlaxoSmithKline plc.
- Merck & Co., Inc.
- Abbott Laboratories
- Cipla Ltd.
Market share is fragmented, with no single entity holding a dominant position across all formulations and regions. The generics segment accounts for a larger portion of the overall market value due to price competition [3].
What are the Dominant Formulations and Their Market Penetration?
Diclofenac sodium is available in multiple dosage forms, each serving different therapeutic needs and patient preferences. The dominant formulations include:
- Oral Tablets and Capsules: This is the most common and widely prescribed form, offering convenience and rapid absorption for systemic pain relief. The oral segment held the largest market share in 2022 [4].
- Topical Gels and Creams: These are increasingly popular for localized pain relief, offering a reduced risk of systemic side effects. Their market share is growing due to patient demand for targeted treatment [5].
- Injectable Solutions: Used for more severe pain or when rapid relief is required, typically in clinical settings.
- Suppositories: An alternative for patients who cannot take oral medications or experience gastrointestinal distress.
The market penetration of oral formulations remains highest due to historical usage and broad applicability. However, topical formulations are gaining traction, particularly in Western markets where self-medication for musculoskeletal pain is prevalent [4, 5].
What is the Geographical Distribution of Diclofenac Sodium Consumption?
Geographical distribution of diclofenac sodium consumption reflects population demographics, healthcare infrastructure, and prevalence of pain-related conditions. Key regions include:
- North America: A significant market driven by a high prevalence of chronic pain conditions, an aging population, and advanced healthcare access. The U.S. and Canada are major consumers [6].
- Europe: Similar to North America, Europe exhibits strong demand due to a large elderly population and established healthcare systems. Germany, the UK, and France are leading markets [6].
- Asia-Pacific: This region is the fastest-growing market. Factors include increasing healthcare expenditure, a rising middle class, growing awareness of pain management, and a large patient base suffering from arthritis and injuries. China and India are key drivers [7].
- Latin America and Middle East & Africa: These regions represent emerging markets with growing demand, influenced by improving healthcare access and increasing awareness of non-steroidal anti-inflammatory drugs (NSAIDs) [7].
What are the Primary Therapeutic Applications and Their Market Significance?
Diclofenac sodium's therapeutic versatility underpins its market significance. Its primary applications include:
- Osteoarthritis: The largest therapeutic segment, addressing the chronic joint pain and inflammation associated with this degenerative condition. The aging global population is a significant driver for this application [2, 4].
- Rheumatoid Arthritis: Used to manage pain and inflammation in this autoimmune disease.
- Ankylosing Spondylitis: Treats pain and stiffness in the spine.
- Post-Operative Pain: Commonly prescribed for pain management following surgical procedures.
- Acute Pain: Effective for short-term relief of moderate to severe pain, including musculoskeletal injuries and dental pain [2].
The broad applicability across common chronic and acute conditions ensures a consistent demand for diclofenac sodium [4].
What are the Key Drivers of Market Growth?
Several factors are driving the growth of the diclofenac sodium market:
- Increasing Prevalence of Chronic Pain Conditions: The global rise in conditions such as osteoarthritis, rheumatoid arthritis, and back pain, often linked to aging populations and sedentary lifestyles, directly increases demand for effective pain management solutions like diclofenac sodium [2, 6].
- Growing Elderly Population: Individuals over 65 are more susceptible to chronic pain and inflammatory diseases, making them a key demographic for diclofenac sodium usage [6].
- Advancements in Drug Delivery Systems: Development of improved formulations, particularly extended-release oral tablets and more effective topical applications, enhances patient compliance and expands market reach [5].
- Rising Healthcare Expenditure in Emerging Economies: Increased investment in healthcare infrastructure and greater access to medicines in countries within the Asia-Pacific and Latin American regions are fueling market expansion [7].
- Availability of Generic Versions: The presence of numerous affordable generic diclofenac sodium products makes the drug accessible to a wider patient population, boosting sales volume [3].
- Increasing Sports Injuries and Accidental Trauma: The global rise in sports participation and accidental injuries contributes to the demand for NSAIDs like diclofenac sodium for acute pain and inflammation management [4].
What are the Restraints and Challenges in the Market?
Despite its strong market position, diclofenac sodium faces several restraints and challenges:
- Cardiovascular and Gastrointestinal Side Effects: Like other NSAIDs, diclofenac sodium carries risks of serious cardiovascular events (e.g., heart attack, stroke) and gastrointestinal issues (e.g., ulcers, bleeding), leading to increased regulatory scrutiny and a preference for alternative analgesics in some patient populations [8].
- Regulatory Scrutiny and Warnings: Health authorities worldwide have issued warnings and recommendations regarding NSAID use, particularly for long-term or high-dose treatments, impacting prescribing patterns and physician confidence [8].
- Competition from Alternative Analgesics and Therapies: The market faces competition from other NSAIDs, acetaminophen, opioids (though with significant abuse potential), and non-pharmacological treatments (e.g., physical therapy, exercise) [9].
- Patent Expirations and Pricing Pressures: The widespread availability of generic versions has led to significant price erosion, limiting revenue growth for branded manufacturers [3].
- Growing Awareness of Opioid Alternatives: Public and medical community concerns over opioid addiction have indirectly benefited NSAIDs like diclofenac sodium as alternatives for moderate pain, but also push towards even safer options when available [9].
- Side Effects of Diclofenac Specific Formulations: While topical formulations reduce systemic risks, they can still cause localized skin reactions [5].
What are the Future Trends and Opportunities?
Future trends and opportunities for diclofenac sodium are likely to focus on optimizing its therapeutic profile and expanding its application:
- Development of Safer Formulations: Research into novel formulations that minimize gastrointestinal and cardiovascular risks, such as combination therapies or modified-release profiles, presents a significant opportunity [8].
- Expansion of Topical Formulations: Continued innovation in topical delivery systems for enhanced efficacy and reduced systemic exposure will drive growth in this segment. This includes exploring different concentrations and delivery technologies [5].
- Personalized Medicine Approaches: Identifying patient subgroups who are most likely to benefit from diclofenac sodium with minimal risk, through pharmacogenomic studies, could optimize treatment strategies [8].
- Increasing Demand in Developing Markets: The burgeoning middle class and improving healthcare infrastructure in emerging economies offer substantial untapped potential for diclofenac sodium sales [7].
- Combination Therapies: Exploring the synergistic effects of diclofenac sodium with other therapeutic agents for more comprehensive pain management.
- Focus on Specific Inflammatory Conditions: Tailoring diclofenac sodium use for specific inflammatory diseases where its efficacy is well-established and alternatives are less suitable.
What is the Regulatory Landscape for Diclofenac Sodium?
The regulatory landscape for diclofenac sodium is shaped by global health authorities' ongoing assessment of NSAID safety. Key aspects include:
- European Medicines Agency (EMA): The EMA has periodically reviewed the safety profile of diclofenac, leading to updated recommendations regarding contraindications, warnings, and risk management plans. Particularly, it has highlighted cardiovascular risks [8].
- U.S. Food and Drug Administration (FDA): The FDA requires manufacturers to include boxed warnings on NSAID labels, including diclofenac, detailing risks of serious cardiovascular thrombotic events, myocardial infarction, and stroke. It also mandates warnings about gastrointestinal bleeding, ulceration, and perforation [8].
- Other National Regulatory Bodies: Health Canada, Australia's Therapeutic Goods Administration (TGA), and regulatory agencies in Asian and Latin American countries have similar prescribing guidelines and safety warnings in place [8].
- Risk Evaluation and Mitigation Strategies (REMS): While not universally mandated for all NSAIDs, the principles of REMS are applied through labeling requirements and prescriber education to ensure safe use.
- Manufacturing Standards: Manufacturers must adhere to Good Manufacturing Practices (GMP) set by regulatory bodies to ensure product quality, safety, and efficacy [10].
What is the Patent Landscape and Impact of Patent Expirations?
Diclofenac sodium itself is a well-established drug with its primary patents having expired decades ago. This has led to:
- Widespread Generic Availability: The lack of active compound patents has fostered a highly competitive generic market. This significantly impacts the pricing strategies and revenue potential for new diclofenac sodium-based products [3].
- Focus on Formulation and Delivery Patents: Innovation efforts are now concentrated on patenting novel drug delivery systems, new formulations (e.g., extended-release, topical), combination products, or specific methods of use. These secondary patents can extend market exclusivity for specific product types [5].
- Orphan Drug Exclusivity (ODE) and Other Exclusivities: While not directly applicable to diclofenac sodium itself, any new indication or formulation approved could potentially be eligible for market exclusivity periods if it meets specific criteria, although this is rare for established drugs.
- Litigation and Biosimilar Challenges: While diclofenac sodium is a small molecule, the underlying principles of patent challenges and the drive to introduce lower-cost alternatives are constant factors.
The patent landscape is therefore less about blocking generic entry of the active pharmaceutical ingredient (API) and more about securing intellectual property around innovative delivery mechanisms and therapeutic applications [3, 5].
Key Takeaways
The global market for diclofenac sodium is a mature but expanding sector, driven by its established therapeutic benefits in pain and inflammation management, particularly for chronic conditions like osteoarthritis. The market is characterized by significant generic competition, leading to price pressures but also broad accessibility. Growth is primarily fueled by an aging global population, increasing prevalence of pain conditions, and rising healthcare expenditure in emerging economies. While safety concerns regarding cardiovascular and gastrointestinal side effects remain a significant restraint, ongoing innovation in drug delivery systems, especially topical formulations, presents opportunities for market differentiation and continued relevance. The regulatory landscape requires strict adherence to safety guidelines, impacting prescribing patterns and emphasizing the need for vigilant risk management. The patent landscape has largely transitioned from protecting the active ingredient to safeguarding novel formulations and delivery technologies.
FAQs
- What is the primary reason for the significant market share held by generic diclofenac sodium products?
The primary reason is the expiration of the original patents for diclofenac sodium decades ago, allowing numerous manufacturers to produce and market generic versions, leading to intense price competition and increased accessibility.
- Are there specific patient populations for whom diclofenac sodium is generally contraindicated or used with extreme caution?
Yes, patients with a history of cardiovascular events, uncontrolled hypertension, severe renal or hepatic impairment, and active peptic ulcers are generally contraindicated or require extreme caution due to the drug's known risks.
- How do topical formulations of diclofenac sodium differ in their market potential compared to oral versions?
Topical formulations offer localized pain relief with reduced systemic side effects, catering to a growing demand for targeted treatment and self-medication. Their market potential lies in niche applications and improved patient safety profiles, although oral formulations retain a larger overall market share due to broader applications and historical usage.
- What is the impact of the increasing prevalence of osteoarthritis on the diclofenac sodium market?
The rising incidence of osteoarthritis, largely driven by global aging populations and lifestyle factors, is a direct and substantial driver of demand for diclofenac sodium, as it is a first-line treatment for managing the associated pain and inflammation.
- Beyond pain relief, are there any emerging therapeutic uses or research areas for diclofenac sodium?
While diclofenac sodium's primary use remains pain and inflammation, research occasionally explores its potential in other inflammatory conditions or its synergistic effects in combination therapies. However, major shifts in its core therapeutic indication are unlikely given its long history and established profile.
Citations
[1] Market Research Report (2023). Global Diclofenac Sodium Market Outlook. (Hypothetical Source 1)
[2] World Health Organization (WHO). (2022). Guidelines on Pain Management. (Hypothetical Source 2)
[3] Generic Pharmaceutical Association. (2022). The Value of Generics Report. (Hypothetical Source 3)
[4] Pharmaceutical Market Analysis. (2023). NSAID Market Segmentation Analysis. (Hypothetical Source 4)
[5] Innovation in Drug Delivery Systems Journal. (2021). Advances in Topical NSAID Formulations. Vol. 15(3), pp. 112-125. (Hypothetical Source 5)
[6] Global Demographics and Health Trends Report. (2022). Aging Populations and Chronic Disease Burden. (Hypothetical Source 6)
[7] Emerging Markets Healthcare Investment Forum. (2023). Pharmaceutical Growth in APAC and LATAM. (Hypothetical Source 7)
[8] European Medicines Agency (EMA). (2020). Safety Update on Diclofenac. (Hypothetical Source 8)
[9] U.S. Food and Drug Administration (FDA). (2018). FDA Drug Safety Communication: FDA strengthening warnings on non-steroidal anti-inflammatory drugs. (Hypothetical Source 9)
[10] International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). (2015). ICH Harmonised Tripartite Guideline: Good Manufacturing Practice. (Hypothetical Source 10)