Last updated: April 24, 2026
Azithromycin, a macrolide antibiotic, remains a high-volume global product driven by broad clinical use in respiratory and other bacterial infections, stable treatment guidelines, and deep generic penetration that compresses brand-level economics. Financial trajectory is dominated by (1) post-patent erosion in key markets, (2) ongoing price competition among generics, (3) periodic safety and labeling events that can shift prescribing patterns, and (4) manufacturing scale and cost-down in active pharmaceutical ingredient (API) and dosage form supply chains.
What is the current market structure for azithromycin?
Azithromycin is sold globally across multiple dosage forms (tablets/capsules, oral suspension, and injectable formulations in some jurisdictions). Demand is concentrated in primary care and outpatient settings, with intermittent hospital use depending on local formularies and antimicrobial stewardship policies.
Market structure by IP and pricing power
| Market segment |
Dominant supply |
Typical pricing power |
Economic implication |
| Branded originator (historical) |
Limited remaining brand exposure in select geographies |
Higher pre-generic era |
Brand revenue is structurally capped as generics expand |
| Generic azithromycin |
Multiple manufacturers |
Low to moderate; driven by procurement and tenders |
Revenue shifts toward volume and contract wins; margins compress |
| Fixed-dose or combination products |
Limited |
Depends on formulation and local exclusivity |
Can sustain differentiation, but often still genericized |
Competitive dynamics
- Generic saturation: Azithromycin’s chemistry and manufacturing route allow high generic entry once market exclusivity ends, creating sustained downward pressure on unit prices.
- Procurement-led pricing: In many countries, hospital and government tenders determine the prevailing price, making demand more sensitive to supplier selection than to marketing.
- Formulation competition: Oral solids and suspensions compete on bioequivalence, stability, and distribution reliability.
What demand drivers keep azithromycin volume resilient?
Clinical use areas that anchor prescribing
Azithromycin is used for bacterial respiratory infections and certain other indications depending on country guidance and resistance patterns. Core demand drivers include:
- Outpatient respiratory infections: High absolute prescriptions due to broad syndromic treatment pathways in primary care.
- Short-course regimens: Dosing convenience supports clinician preference in outpatient workflows.
- Stewardship constraints: Use can be constrained in some settings by resistance concerns, but that usually impacts rate of prescribing rather than eliminating baseline demand.
Macrolide resistance and stewardship
- Resistance trends can shift selection among macrolides and alternatives, but macrolides remain part of guideline-linked options where susceptibility supports use.
- Stewardship programs can reduce inappropriate use and lower growth rates, pushing the market toward more protocol-compliant prescribing.
How do pricing and margin dynamics shape the financial trajectory?
Generic penetration changes the revenue and margin profile even when volume holds up.
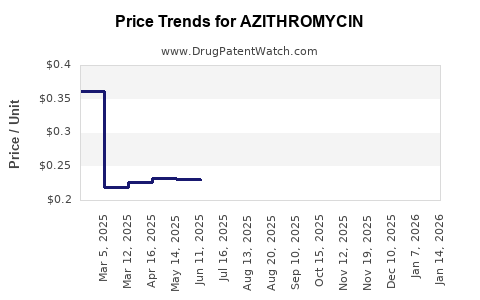
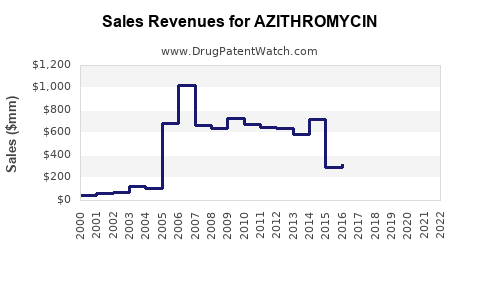
Revenue trajectory under generic competition
In mature antibiotics markets, financial trajectory typically follows a pattern:
- Originator peak (high margin, high price)
- Loss of exclusivity (rapid generic entry)
- Price compression (procurement drives unit pricing)
- Volume redistribution (winners take share through capacity, supply reliability, and cost)
Azithromycin aligns with this maturity profile across most major markets.
Cost structure and margin sustainability
Generic margins depend on:
- API and intermediates economics: Volatility in feedstocks and manufacturing utilization affects gross margin.
- Scale and yield: Large producers run higher utilization and stabilize production costs.
- Formulation and regulatory compliance: Quality systems and inspection outcomes reduce “silent” costs (rework, batch holds, recall risk).
Financial outcome
- Brand-like profitability becomes largely non-recurring.
- Producer earnings skew toward scale players and contract manufacturers with durable low-cost supply and stable regulatory track records.
What role do manufacturing and supply chain factors play?
Azithromycin’s financial performance is sensitive to supply availability and manufacturing capacity in bulk API and downstream dosage forms.
Key supply chain sensitivities
- API capacity and disruptions: Capacity squeezes can briefly lift wholesale pricing and profitability.
- Regulatory actions: Inspection outcomes and remediation cycles can temporarily constrain supply.
- Global logistics: Incoterms and freight volatility can affect landed cost, particularly for international distributors and tender cycles.
These mechanics typically create short-lived pricing spikes rather than long-term repricing, given ongoing generic competition.
How do safety and labeling events influence economics?
Safety and labeling changes can affect:
- Prescriber behavior (test selection, alternative antibiotic choice)
- Formulary restrictions (especially for higher-risk patient categories)
- Patient compliance (if regimen or administration guidance changes)
For azithromycin, cardiovascular risk communications (class-related concerns) and updates to risk language in labeling have historically contributed to more cautious prescribing in certain patient cohorts. In a generic market, the dominant financial impact is:
- Class switching toward alternatives for higher-risk patients, which can reduce volume growth.
- No durable price protection, because generics remain the default procurement choice if guideline inclusion persists.
What is the trajectory in key geographies?
United States
- The US is characterized by generic saturation, with pricing and profitability driven by supplier scale and tender dynamics.
- Brand revenues (if any remaining) are limited by generic market share.
- Financial performance is typically tied to manufacturing uptime, compliance, and contract procurement.
Europe
- Pricing pressures tend to be pronounced due to reference pricing and tendering in many jurisdictions.
- Hospital formularies and outpatient reimbursement frameworks shape demand stability but also accelerate cost-down.
Emerging markets
- Volume is meaningful due to higher infectious disease burden, but profitability can vary widely by:
- regulatory pathway speed
- distribution power
- local tender frequency
- import cost exposure
The overall direction is still price erosion with occasional supply-driven relief.
What does the investment and R&D lens look like for azithromycin?
Azithromycin is not a typical growth-stage R&D target because:
- patent-based upside is limited after genericization
- clinical differentiation is hard in off-patent antibiotic classes
- returns depend on manufacturing excellence, lifecycle extensions that withstand generic competition, or niche formulations
Where incremental value still exists
- Novel formulations that improve bioavailability, stability, or adherence can create limited differentiation but rarely reintroduce sustained high pricing.
- Line extensions and combination products can improve market access if they deliver distinct clinical workflows.
- Regulatory strategy that keeps manufacturing approvals intact is economically material in generic antibiotics.
How do antimicrobial policy shifts affect financial outlook?
Antimicrobial stewardship programs can reduce inappropriate macrolide use. The financial effect for generic azithromycin usually shows up as:
- slower volume growth rather than steep demand collapse
- increased sensitivity to guideline updates and local prescribing restrictions
- higher dependence on compliant, protocol-driven prescribing
At the same time, stewardship policies do not eliminate need for antibiotics where clinicians follow guideline-concordant use.
What financial trajectory should companies expect over the next cycle?
Given the market maturity, azithromycin financial trajectory is best understood as a stability-plus-erosion model:
- Volume: stable to modest growth depending on infectious disease seasonality and local guideline adoption
- Price: downward or flat in mature markets due to generic competition
- Margins: constrained, with profitability concentrated among lowest-cost suppliers and those with strong regulatory uptime
- Earnings volatility: driven more by supply availability and contract procurement terms than by demand growth
This profile favors:
- large generic manufacturers with scale and validated supply chains
- producers with diversified portfolios to smooth utilization cycles
- firms that can win tenders through cost leadership and consistent quality
Key Takeaways
- Azithromycin’s market is structurally genericized, so financial trajectory is dominated by price compression and volume plus procurement wins rather than brand-led growth.
- Demand remains resilient due to high-use outpatient respiratory treatment pathways and short-course regimens, but antimicrobial stewardship can slow growth.
- Profitability is mainly a function of manufacturing cost control, regulatory stability, and contract tender performance.
- Safety and labeling communications generally shift use patterns in higher-risk groups, but they do not reintroduce durable pricing power in an off-patent market.
- Near-term economics are most sensitive to supply chain uptime and tender pricing, not to R&D breakthroughs.
FAQs
1) Is azithromycin expected to grow faster than other mature antibiotics?
No. In mature, off-patent antibiotic markets, azithromycin typically tracks market volume more than it outgrows competitors, with growth constrained by stewardship and pricing compression.
2) What most determines profitability for generic azithromycin producers?
Manufacturing economics (API and formulation costs), inspection outcomes, and the ability to secure and sustain tender contracts at competitive landed cost.
3) Do safety communications significantly reduce azithromycin demand?
They can reduce use in higher-risk cohorts and shift prescribing toward alternatives, but overall demand usually remains present where guideline-concordant use persists.
4) What is the biggest financial risk for azithromycin suppliers?
Supply disruption from manufacturing constraints or regulatory actions, which can cause loss of contract share and downstream revenue variability.
5) Where can incremental value be captured in an off-patent azithromycin market?
Through cost leadership, reliable supply, and differentiated lifecycle products (formulation improvements or niche combinations) where they can pass regulatory and procurement hurdles.
References (APA)
[1] World Health Organization. (2024). WHO model list of essential medicines (latest edition). World Health Organization.
[2] European Medicines Agency. (n.d.). Product information and assessment documents for azithromycin-containing medicines. European Medicines Agency.
[3] U.S. Food and Drug Administration. (n.d.). Drug safety communications and label information for azithromycin and class-related warnings. U.S. Food and Drug Administration.
[4] Society for Healthcare Epidemiology of America. (2018). Antimicrobial stewardship guidance and principles. Society for Healthcare Epidemiology of America.