RYALTRIS Drug Patent Profile
✉ Email this page to a colleague
When do Ryaltris patents expire, and when can generic versions of Ryaltris launch?
Ryaltris is a drug marketed by Glenmark Speclt and is included in one NDA. There are sixteen patents protecting this drug.
This drug has one hundred patent family members in thirty-four countries.
The generic ingredient in RYALTRIS is mometasone furoate; olopatadine hydrochloride. There are thirty drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the mometasone furoate; olopatadine hydrochloride profile page.
DrugPatentWatch® Generic Entry Outlook for Ryaltris
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 8, 2039. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for RYALTRIS?
- What are the global sales for RYALTRIS?
- What is Average Wholesale Price for RYALTRIS?
Summary for RYALTRIS
| International Patents: | 100 |
| US Patents: | 16 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
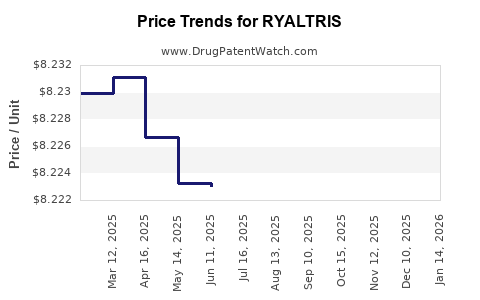
| Drug Prices: | Drug price information for RYALTRIS |
| What excipients (inactive ingredients) are in RYALTRIS? | RYALTRIS excipients list |
| DailyMed Link: | RYALTRIS at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for RYALTRIS
Generic Entry Date for RYALTRIS*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
SPRAY, METERED;NASAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for RYALTRIS
| Drug Class | Corticosteroid Histamine-1 Receptor Antagonist Histamine-1 Receptor Inhibitor Mast Cell Stabilizer |
| Mechanism of Action | Corticosteroid Hormone Receptor Agonists Histamine H1 Receptor Antagonists |
| Physiological Effect | Decreased Histamine Release |
US Patents and Regulatory Information for RYALTRIS
RYALTRIS is protected by sixteen US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of RYALTRIS is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
International Patents for RYALTRIS
When does loss-of-exclusivity occur for RYALTRIS?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 18260934
Estimated Expiration: ⤷ Start Trial
Patent: 19224850
Estimated Expiration: ⤷ Start Trial
Patent: 23270308
Patent: Dispensing device and pharmaceutical composition for the treatment of rhinitis
Estimated Expiration: ⤷ Start Trial
Patent: 25203489
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering RYALTRIS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| South Korea | 20200125625 | 모메타손 및 올로파타딘의 조합을 사용하는 소아 대상체의 알레르기성 비염의 치료 | ⤷ Start Trial |
| South Korea | 102709429 | ⤷ Start Trial | |
| Russian Federation | 2015128497 | СТАБИЛЬНАЯ ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ С ФИКСИРОВАННОЙ ДОЗОЙ, СОДЕРЖАЩАЯ МОМЕТАЗОН И ОЛОПАТАДИН | ⤷ Start Trial |
| European Patent Office | 3468532 | ⤷ Start Trial | |
| China | 112870203 | 用于治疗鼻炎的分配装置和药物组合物 (Dispensing device and pharmaceutical composition for treating rhinitis) | ⤷ Start Trial |
| South Korea | 102562797 | ⤷ Start Trial | |
| Japan | 2016534142 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for RYALTRIS
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3043773 | 2021045 | Norway | ⤷ Start Trial | PRODUCT NAME: MOMETASON ELLER ET SALT DERAV OG OLOPATADIN ELLER ET SALT DERAV; NAT. REG. NO/DATE: 20-13278 20210709; FIRST REG. NO/DATE: 140638 20210426 |
| 3043773 | 122021000085 | Germany | ⤷ Start Trial | PRODUCT NAME: MOMETASON ODER EIN SALZ DAVON UND OLOPATADIN ODER EIN SALZ DAVON; NAT. REGISTRATION NO/DATE: 2205824.00.00 20211117; FIRST REGISTRATION: OESTERREICH 140638 20210426 |
| 0112669 | 96C0002 | Belgium | ⤷ Start Trial | PRODUCT NAME: MOMETASONE FUROATE; NAT. REGISTRATION NO/DATE: NL 19601 19950919; FIRST REGISTRATION: GR - 10681/89 19900622 |
| 3043773 | 2022C/520 | Belgium | ⤷ Start Trial | PRODUCT NAME: MOMETASONE OF EEN ZOUT HIERVAN EN OLOPATADINE OF EEN ZOUT HIERVAN; AUTHORISATION NUMBER AND DATE: BE595626 20220203 |
| 3043773 | CA 2021 00050 | Denmark | ⤷ Start Trial | PRODUCT NAME: MOMETASON ELLER ET SALT DERAF OG OLOPATADIN ELLER ET SALT DERAF; NAT. REG. NO/DATE: 63820 20210706; FIRST REG. NO/DATE: AT 140638 20210426 |
| 3043773 | SPC/GB21/077 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: MOMETASONE OR A SALT THEREOF AND OLOPATADINE OR A SALT THEREOF; REGISTERED: AT 140638 20210426; UK PL 25258/0331 - 0001 20210511 |
| 3043773 | C202130060 | Spain | ⤷ Start Trial | PRODUCT NAME: MOMETASONA O UNA SAL DE LA MISMA Y OLOPATADINA O UNA SAL DE LA MISMA; NATIONAL AUTHORISATION NUMBER: 86059-SE/H/2040/001/DC; DATE OF AUTHORISATION: 20210701; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): 140638; DATE OF FIRST AUTHORISATION IN EEA: 20210419 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
RYALTRIS Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.