LATANOPROST - Generic Drug Details
✉ Email this page to a colleague
What are the generic drug sources for latanoprost and what is the scope of patent protection?
Latanoprost
is the generic ingredient in seven branded drugs marketed by Sun Pharm, Thea Pharma, Amring Pharms, Apotex Inc, Bausch And Lomb, Carnegie, Epic Pharma Llc, Eugia Pharma, Fdc Ltd, Gland, Mankind Pharma, Micro Labs, Sandoz, Somerset, Upjohn, and Alcon Labs Inc, and is included in eighteen NDAs. There are twenty-one patents protecting this compound. Additional information is available in the individual branded drug profile pages.Latanoprost has fifty-three patent family members in thirty-one countries.
There are twenty drug master file entries for latanoprost. Eighteen suppliers are listed for this compound. There is one tentative approval for this compound.
Summary for LATANOPROST
| International Patents: | 53 |
| US Patents: | 21 |
| Tradenames: | 7 |
| Applicants: | 16 |
| NDAs: | 18 |
| Drug Master File Entries: | 20 |
| Finished Product Suppliers / Packagers: | 18 |
| Raw Ingredient (Bulk) Api Vendors: | 77 |
| Clinical Trials: | 206 |
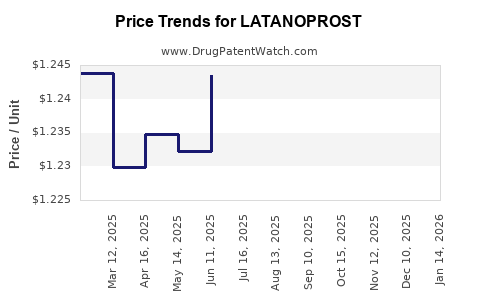
| Drug Prices: | Drug price trends for LATANOPROST |
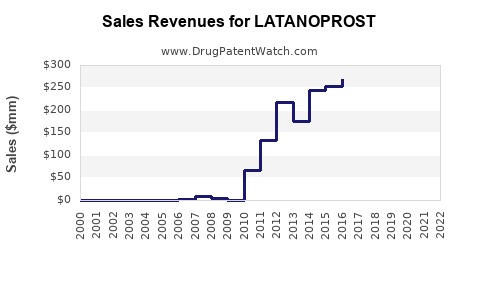
| Drug Sales Revenues: | Drug sales revenues for LATANOPROST |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for LATANOPROST |
| What excipients (inactive ingredients) are in LATANOPROST? | LATANOPROST excipients list |
| DailyMed Link: | LATANOPROST at DailyMed |
Recent Clinical Trials for LATANOPROST
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Alcon Research | PHASE4 |
| Mayo Clinic | PHASE4 |
| Insight Eyecare Specialties, Inc. dba Vision Source Eyecare, | PHASE4 |
Generic filers with tentative approvals for LATANOPROST
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Start Trial | ⤷ Start Trial | 0.005% | SOLUTION; OPHTHALMIC |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Anatomical Therapeutic Chemical (ATC) Classes for LATANOPROST
US Patents and Regulatory Information for LATANOPROST
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alcon Labs Inc | ROCKLATAN | latanoprost; netarsudil dimesylate | SOLUTION/DROPS;OPHTHALMIC | 208259-001 | Mar 12, 2019 | RX | Yes | Yes | 11,185,538 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Thea Pharma | IYUZEH | latanoprost | SOLUTION/DROPS;OPHTHALMIC | 216472-001 | Dec 13, 2022 | RX | Yes | Yes | 8,637,054 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Alcon Labs Inc | ROCKLATAN | latanoprost; netarsudil dimesylate | SOLUTION/DROPS;OPHTHALMIC | 208259-001 | Mar 12, 2019 | RX | Yes | Yes | 11,197,853 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Carnegie | LATANOPROST | latanoprost | SOLUTION/DROPS;OPHTHALMIC | 202077-001 | Feb 11, 2013 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Alcon Labs Inc | ROCKLATAN | latanoprost; netarsudil dimesylate | SOLUTION/DROPS;OPHTHALMIC | 208259-001 | Mar 12, 2019 | RX | Yes | Yes | 11,028,081 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Sun Pharm | XELPROS | latanoprost | EMULSION;OPHTHALMIC | 206185-001 | Sep 12, 2018 | DISCN | Yes | No | 9,629,852 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for LATANOPROST
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Upjohn | XALATAN | latanoprost | SOLUTION/DROPS;OPHTHALMIC | 020597-001 | Jun 5, 1996 | 5,422,368 | ⤷ Start Trial |
| Upjohn | XALATAN | latanoprost | SOLUTION/DROPS;OPHTHALMIC | 020597-001 | Jun 5, 1996 | 4,599,353 | ⤷ Start Trial |
| Upjohn | XALATAN | latanoprost | SOLUTION/DROPS;OPHTHALMIC | 020597-001 | Jun 5, 1996 | 6,429,226 | ⤷ Start Trial |
| Upjohn | XALATAN | latanoprost | SOLUTION/DROPS;OPHTHALMIC | 020597-001 | Jun 5, 1996 | 5,296,504 | ⤷ Start Trial |
| Upjohn | XALATAN | latanoprost | SOLUTION/DROPS;OPHTHALMIC | 020597-001 | Jun 5, 1996 | 7,163,959 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for LATANOPROST
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Morocco | 34324 | ⤷ Start Trial | |
| Brazil | 112012033435 | ⤷ Start Trial | |
| Singapore | 186361 | ⤷ Start Trial | |
| Mexico | 2012014503 | ⤷ Start Trial | |
| Japan | 5646331 | ⤷ Start Trial | |
| Chile | 2012003684 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for LATANOPROST
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0364417 | 9690031-1 | Sweden | ⤷ Start Trial | PRODUCT NAME: LATANOPROST |
| 3461484 | 301101 | Netherlands | ⤷ Start Trial | PRODUCT NAME: LATANOPROST OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN EN NETARSUDILMESYLAAT; REGISTRATION NO/DATE: EU/1/20/1502 20210108 |
| 0364417 | 97C0128 | France | ⤷ Start Trial | PRODUCT NAME: LATANOPROST; REGISTRATION NO/DATE IN FRANCE: NL 22549 DU 19970724; REGISTRATION NO/DATE AT EEC: 12716 DU 19960718 |
| 3461484 | 2021C/515 | Belgium | ⤷ Start Trial | PRODUCT NAME: ROCLANDA - LATANOPROST / NETARSUDIL; AUTHORISATION NUMBER AND DATE: EU/1/20/1502 20210108 |
| 3461484 | 122021000036 | Germany | ⤷ Start Trial | PRODUCT NAME: LATANOPROST, ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON, UND NETARSUDIL MESYLAT; REGISTRATION NO/DATE: EU/1/20/1502 20210107 |
| 3461484 | SPC/GB21/033 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: A COMBINATION OF LATANOPROST AND NETARSUDIL; REGISTERED: UK EU/1/20/1502(FOR NI) 20210107; UK PLGB 16053/0034 20210107 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Latanoprost: Market Dynamics and Financial Trajectory
More… ↓