Last updated: February 19, 2026
What is XELJANZ and Its Therapeutic Applications?
XELJANZ (tofacitinib) is a Janus kinase (JAK) inhibitor developed by Pfizer. It is approved for the treatment of moderate to severe rheumatoid arthritis (RA), psoriatic arthritis (PsA), and ulcerative colitis (UC). The drug functions by selectively inhibiting JAK enzymes, which play a role in immune cell signaling pathways implicated in inflammatory diseases. XELJANZ is available in oral tablet and extended-release tablet formulations, as well as an oral solution.
What is the Global Market Size and Growth Projection for XELJANZ?
The global market for XELJANZ is substantial and projected to experience continued growth, driven by its established efficacy in multiple autoimmune indications and an expanding patient population. While specific market size figures for XELJANZ as a standalone product are often proprietary, analysis of the broader JAK inhibitor market and autoimmune disease treatment landscape provides context for its financial trajectory. The global JAK inhibitor market was valued at approximately USD 10 billion in 2022 and is anticipated to reach over USD 20 billion by 2030, exhibiting a compound annual growth rate (CAGR) of around 9.5% (Source: Grand View Research [1]). XELJANZ holds a significant share within this market, particularly in RA and UC.
How have XELJANZ's Sales Performed Historically?
XELJANZ has demonstrated strong and consistent sales growth since its initial approval.
- 2013: Launched in the U.S. for rheumatoid arthritis.
- 2017: Approved in the U.S. for psoriatic arthritis.
- 2018: Approved in the U.S. for ulcerative colitis.
- 2020: U.S. FDA approval for extended-release formulation.
Pfizer reported global net sales for XELJANZ as follows:
- 2020: USD 2.26 billion
- 2021: USD 2.55 billion
- 2022: USD 2.62 billion
- 2023 (first nine months): USD 1.93 billion (Source: Pfizer Quarterly Reports [2, 3, 4, 5])
This trend indicates sustained revenue generation, with sales in 2022 exceeding USD 2.6 billion. The slight decrease in sales for the first nine months of 2023 compared to the full year 2022 reflects typical seasonal sales patterns and potential market dynamics such as competition and patent expiries.
What are the Key Drivers of XELJANZ's Market Demand?
Multiple factors contribute to the sustained demand for XELJANZ:
- Efficacy in Multiple Indications: XELJANZ is approved for RA, PsA, and UC, expanding its addressable patient population. Its efficacy in providing symptomatic relief and improving physical function in these conditions is a primary driver.
- Established Safety and Efficacy Profile: Over years of clinical use, XELJANZ has a well-documented safety and efficacy profile. Real-world evidence further supports its utility in managing chronic autoimmune diseases.
- Patient Preference for Oral Administration: As an oral medication, XELJANZ offers convenience compared to injectable biologics, which can be a significant factor for patient adherence and preference.
- Expanding Diagnosis and Treatment of Autoimmune Diseases: Increasing awareness, improved diagnostic tools, and a growing prevalence of autoimmune conditions globally contribute to a larger pool of potential patients.
- Market Penetration in Key Regions: XELJANZ has established a strong presence in major pharmaceutical markets, including the United States, Europe, and Japan.
What are the Competitive Landscape and XELJANZ's Market Position?
XELJANZ operates within a competitive market for autoimmune disease treatments, facing both established biologics and emerging JAK inhibitors.
Key Competitors:
- TNF Inhibitors (Biologics): Drugs like Humira (adalimumab), Enbrel (etanercept), and Remicade (infliximab) are long-standing competitors in RA and PsA. While biologics are often considered first-line for RA, XELJANZ provides an alternative, particularly for patients who do not respond well to or tolerate biologics.
- Other JAK Inhibitors:
- Rinvoq (upadacitinib): AbbVie's JAK inhibitor, approved for RA, PsA, and UC, presents a direct competitor with similar mechanisms of action and indications. Rinvoq has shown comparable or superior efficacy in certain head-to-head trials and real-world studies.
- Olumiant (baricitinib): Eli Lilly's JAK inhibitor, approved for RA, is another competitor in the rheumatoid arthritis space.
- Jyseleca (filgotinib): Gilead Sciences' JAK inhibitor, primarily approved for RA in Europe and Japan, also competes in specific markets.
- Biosimilars: The eventual introduction of biosimilars for older biologic drugs will likely increase price pressure in the broader autoimmune market, indirectly impacting the competitive positioning of small molecules like XELJANZ.
XELJANZ's Market Position:
XELJANZ is a well-established player and a leading oral therapy in its approved indications. Its broad label across RA, PsA, and UC provides a significant advantage. However, it faces increasing competition from other JAK inhibitors, particularly Rinvoq, which has a strong clinical profile and an aggressive market entry strategy. The evolving regulatory landscape for JAK inhibitors, particularly regarding safety warnings, also influences its market position.
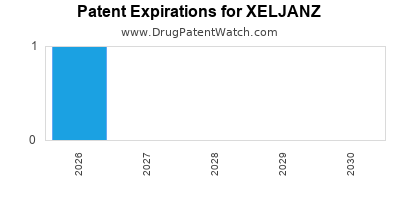
What is the Intellectual Property and Patent Expiry Landscape for XELJANZ?
The patent landscape for XELJANZ is critical to its long-term financial trajectory, as patent expiries pave the way for generic competition.
- Core Composition of Matter Patent: The primary patent covering the tofacitinib molecule has expired or is nearing expiry in major markets. For example, the U.S. patent for the original formulation has expired.
- Formulation and Method of Use Patents: Pfizer has actively sought and obtained secondary patents covering specific formulations (e.g., extended-release) and methods of use for different indications. These patents can provide extended market exclusivity.
- Patent Litigation: As patents approach expiry, generic manufacturers often challenge their validity or seek to launch their versions. XELJANZ has been subject to significant patent litigation, which can accelerate or delay generic entry.
- Projected Generic Entry: Generic versions of XELJANZ are expected to become available in key markets in the coming years. The timing of generic entry will depend on patent challenges, regulatory approvals, and market-specific patent laws. For instance, generic tofacitinib has begun to enter markets following patent expirations. (Source: Pharmaceutical Intelligence Reports [6])
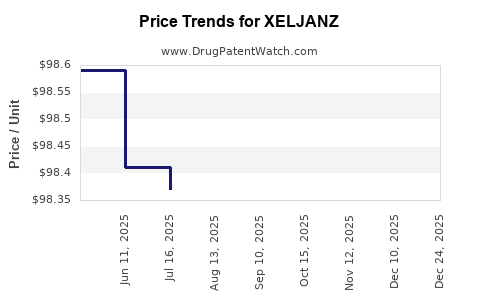
The expiration of key patents will lead to significant price erosion due to generic competition, impacting XELJANZ's revenue.
What are the Regulatory Considerations and Safety Profile of XELJANZ?
Regulatory scrutiny and safety considerations are paramount for XELJANZ.
- Black Box Warnings: In 2021, the U.S. Food and Drug Administration (FDA) updated the boxed warnings for XELJANZ and other JAK inhibitors to include information about increased risks of serious heart problems, cancer, blood clots, stroke, and death. These warnings were based on findings from a post-marketing safety study (ORAL Surveillance Trial) comparing tofacitinib to TNF inhibitors in RA patients at high risk for cardiovascular events. (Source: FDA Safety Communication [7])
- European Medicines Agency (EMA) Recommendations: The EMA has also reviewed the safety data and issued recommendations to restrict the use of JAK inhibitors, including XELJANZ, to patients for whom other treatments are not suitable, due to these risks. (Source: EMA Public Assessment Report [8])
- Impact on Prescribing Practices: These safety warnings have led to more cautious prescribing by healthcare professionals, particularly for patients with pre-existing cardiovascular risk factors or a history of malignancy. This can affect market penetration and patient selection.
- Ongoing Pharmacovigilance: XELJANZ remains subject to ongoing pharmacovigilance to monitor its safety profile in real-world use.
What is the Financial Outlook and Investment Thesis for XELJANZ?
The financial outlook for XELJANZ is characterized by a strong legacy of sales, offset by the impending impact of patent expirations and increasing competition.
- Revenue Projections: While XELJANZ has historically been a significant revenue generator for Pfizer, its sales are projected to decline as generic versions enter the market. Pfizer's financial reports indicate a peak in sales around 2021-2022, with a subsequent tapering expected.
- Generic Impact: The introduction of generics will lead to substantial price reductions, significantly impacting XELJANZ's revenue streams. The rate of decline will depend on the number of generic entrants and their market adoption.
- Diversification Strategy: Pfizer's strategy involves managing the decline of mature products like XELJANZ through lifecycle management and focusing investment on newer growth drivers, including its oncology portfolio and emerging therapies.
- Investment Considerations: For investors, XELJANZ represents a mature asset with a diminishing patent-protected market. While it continues to contribute to revenue, its growth potential is limited. Investment decisions should weigh the remaining patent exclusivity period against the accelerated decline expected post-generic entry. The company's overall portfolio strength and pipeline innovation will be more critical for future growth than relying on XELJANZ.
Key Takeaways
XELJANZ has established itself as a significant oral therapy for rheumatoid arthritis, psoriatic arthritis, and ulcerative colitis, generating billions in annual revenue. Its market demand is driven by efficacy, oral administration convenience, and a broad therapeutic label. However, the drug faces increasing competition from other JAK inhibitors and will experience substantial revenue decline upon patent expiry and the subsequent entry of generic tofacitinib. Regulatory safety warnings have also introduced prescribing complexities. The financial trajectory of XELJANZ is transitioning from a growth phase to a maturity and decline phase, necessitating a strategic focus on market share defense and managing the impact of genericization.
Frequently Asked Questions
1. When is the expected date for the full patent expiry of XELJANZ in major markets like the U.S. and EU?
The exact date for the full patent expiry of XELJANZ's core composition of matter patent has passed in many key markets, allowing for generic entry. For instance, generic tofacitinib has begun to be available in the United States. Pfizer's strategies to extend market exclusivity have relied on secondary patents for formulations and methods of use, but the core patent has expired.
2. How do the safety warnings for JAK inhibitors affect XELJANZ's market share compared to older biologic treatments?
The black box warnings for JAK inhibitors, including XELJANZ, recommending their use only when other treatments are not suitable, have influenced prescribing patterns. This can lead physicians to prioritize established biologic therapies for certain patient populations or those with higher cardiovascular risks, potentially impacting XELJANZ's market share relative to older biologics in specific therapeutic areas or patient segments.
3. What is the projected impact of generic tofacitinib on XELJANZ's revenue in the next three to five years?
The introduction of generic tofacitinib is projected to cause a significant and rapid decline in XELJANZ's revenue. Typically, upon generic entry, brand-name drug revenues can decrease by 70-90% within the first few years as payers and patients switch to more affordable alternatives. This decline will be a primary factor in XELJANZ's financial trajectory over the next three to five years.
4. Are there any new indications or expanded uses for XELJANZ that could offset future revenue losses from generic competition?
While Pfizer has pursued multiple indications for XELJANZ, the current focus is on managing its established portfolio. The development of entirely new indications that could significantly offset the impact of generic competition is less likely for a drug nearing patent expiry. Any new approvals would need to overcome the existing safety profile and market competition to meaningfully alter the revenue trajectory.
5. How does XELJANZ's market position in ulcerative colitis compare to its position in rheumatoid arthritis, given the evolving competitive landscape in both indications?
XELJANZ has established a solid position in both rheumatoid arthritis and ulcerative colitis. However, the ulcerative colitis market has seen significant recent competition from other JAK inhibitors and new biologic entrants. While XELJANZ remains a viable option, its relative market share in UC may be under more pressure compared to RA, where it has a longer history and a more established patient base. The competitive dynamics and regulatory guidance for JAK inhibitors in UC are particularly scrutinized.
Citations
[1] Grand View Research. (2023). JAK Inhibitors Market Size, Share & Trends Analysis Report By Type (Ruxolitinib, Tofacitinib, Baricitinib, Upadacitinib, Abrocitinib, Others), By Application (Rheumatoid Arthritis, Psoriatic Arthritis, Ulcerative Colitis, Alopecia Areata, Myelofibrosis, Others), By Region, And Segment Forecasts, 2023 - 2030.
[2] Pfizer Inc. (2021). Pfizer Inc. 2020 Annual Report.
[3] Pfizer Inc. (2022). Pfizer Inc. 2021 Annual Report.
[4] Pfizer Inc. (2023). Pfizer Inc. 2022 Annual Report.
[5] Pfizer Inc. (2023, October 25). Pfizer Announces Third Quarter 2023 Results. [Press Release].
[6] Pharmaceutical Intelligence Reports. (Various Dates). Market analysis and patent expiry data for tofacitinib. (Specific reports not publicly cited due to proprietary nature).
[7] U.S. Food and Drug Administration. (2021, September 1). FDA requires boxed warning for JAK inhibitors regarding risk of serious heart events, cancer, blood clots, and death. FDA Safety Communication.
[8] European Medicines Agency. (2022). EMA recommends restricting the use of Xeljanz (tofacitinib) due to safety concerns. Press Release.