GLAXOSMITHKLINE Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GLAXOSMITHKLINE
GLAXOSMITHKLINE has one hundred and fifty-two approved drugs.
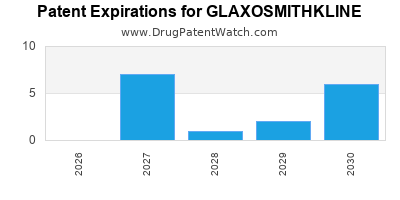
There are thirty-seven US patents protecting GLAXOSMITHKLINE drugs.
There are eight hundred and five patent family members on GLAXOSMITHKLINE drugs in sixty-two countries and one hundred and eighty-four supplementary protection certificates in twenty-one countries.
Summary for GLAXOSMITHKLINE
| International Patents: | 805 |
| US Patents: | 37 |
| Tradenames: | 107 |
| Ingredients: | 93 |
| NDAs: | 152 |
| Drug Master File Entries: | 8 |
| Patent Litigation for GLAXOSMITHKLINE: | See patent lawsuits for GLAXOSMITHKLINE |
| PTAB Cases with GLAXOSMITHKLINE as petitioner: | See PTAB cases with GLAXOSMITHKLINE as petitioner |
Drugs and US Patents for GLAXOSMITHKLINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glaxosmithkline | ARNUITY ELLIPTA | fluticasone furoate | POWDER;INHALATION | 205625-002 | Aug 20, 2014 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | |||||
| Glaxosmithkline | CEPTAZ | ceftazidime | INJECTABLE;INJECTION | 050646-003 | Sep 27, 1990 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Glaxosmithkline | WELLBUTRIN SR | bupropion hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 020358-004 | Jun 14, 2002 | AB1 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ||||
| Glaxosmithkline | VENTOLIN | albuterol sulfate | SOLUTION;INHALATION | 019269-002 | Jan 16, 1987 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Glaxosmithkline | JESDUVROQ | daprodustat | TABLET;ORAL | 216951-002 | Feb 1, 2023 | DISCN | Yes | No | 8,557,834 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Glaxosmithkline | MAREZINE | cyclizine lactate | INJECTABLE;INJECTION | 009495-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Glaxosmithkline | JESDUVROQ | daprodustat | TABLET;ORAL | 216951-004 | Feb 1, 2023 | DISCN | Yes | No | 8,324,208 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for GLAXOSMITHKLINE
Paragraph IV (Patent) Challenges for GLAXOSMITHKLINE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Tablets | 6 mg | ➤ Subscribe | 2009-07-22 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2007-10-30 |
| ➤ Subscribe | Orally Disintegrating Tablets | 25 mg, 50 mg, 100 mg, and 200 mg | ➤ Subscribe | 2009-12-21 |
| ➤ Subscribe | Injection | 6 mg/0.5 mL, 0.5 mL (prefilled syringes) | ➤ Subscribe | 2006-05-09 |
| ➤ Subscribe | Tablets | 3 mg, 4 mg and 5 mg | ➤ Subscribe | 2005-02-04 |
| ➤ Subscribe | Tablets | 250 mg/100 mg | ➤ Subscribe | 2009-04-03 |
| ➤ Subscribe | Extended-release Capsules | 325 mg | ➤ Subscribe | 2006-11-07 |
| ➤ Subscribe | Oral Suspension | 750 mg/5 mL | ➤ Subscribe | 2009-10-20 |
| ➤ Subscribe | Extended-release Tablets | 4 mg | ➤ Subscribe | 2008-10-31 |
| ➤ Subscribe | Extended-release Tablets | 3 mg | ➤ Subscribe | 2009-01-08 |
| ➤ Subscribe | Extended-release Tablets | 2 mg | ➤ Subscribe | 2008-10-14 |
| ➤ Subscribe | Extended-release Tablets | 3 mg | ➤ Subscribe | 2009-01-08 |
| ➤ Subscribe | Injection | 6 mg/0.5 mL, 0.5 mL vials | ➤ Subscribe | 2004-10-25 |
| ➤ Subscribe | Tablets | 0.25 mg, 0.5 mg, 1 mg and 2 mg | ➤ Subscribe | 2004-12-22 |
| ➤ Subscribe | Tablets | 100 mg | ➤ Subscribe | 2007-10-31 |
| ➤ Subscribe | Tablets | 62.5 mg/25 mg | ➤ Subscribe | 2010-09-14 |
| ➤ Subscribe | Extended-release Capsules | 225 mg and 425 mg | ➤ Subscribe | 2006-10-11 |
| ➤ Subscribe | Extended-release Tablets | 25 mg, 50 mg, 100 mg, 200 mg, 250 mg, and 300 mg | ➤ Subscribe | 2014-02-12 |
| ➤ Subscribe | Extended-release Tablets | 8 mg | ➤ Subscribe | 2008-11-03 |
| ➤ Subscribe | Extended-release Tablets | 12 mg | ➤ Subscribe | 2009-02-05 |
International Patents for GLAXOSMITHKLINE Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| South Africa | 200600679 | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2011067212 | ⤷ Start Trial |
| Colombia | 6541613 | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2018183354 | ⤷ Start Trial |
| Brazil | PI0412909 | ⤷ Start Trial |
| Taiwan | 201919476 | ⤷ Start Trial |
| Japan | 6840909 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for GLAXOSMITHKLINE Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3494972 | 202440019 | Slovenia | ⤷ Start Trial | PRODUCT NAME: DOLUTEGRAVIR OR ITS PHARMACEUTICALLY ACCEPTABLE SALT, INCLUDING SODIUM DOLUTEGRAVIR, AND LAMIVUDINE; NATIONAL AUTHORISATION NUMBER: EU/1/19/1370; DATE OF NATIONAL AUTHORISATION: 20190701; AUTHORITY FOR NATIONAL AUTHORISATION: EU |
| 1480644 | 2016/056 | Ireland | ⤷ Start Trial | PRODUCT NAME: A COMBINATION OF CEFTAZIDIME OR A SALT THEREOF, AND AVIBACTAM OR A SALT THEREOF; REGISTRATION NO/DATE: EU/1/16/1109/001 20160624 |
| 2109608 | 122018000048 | Germany | ⤷ Start Trial | PRODUCT NAME: NIRAPARIB ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ, STEREOISOMER ODER TAUTOMER DAVON, EINSCHLIESSLICH DES TOSYLATS DAVON; REGISTRATION NO/DATE: EU/1/17/1235 20171116 |
| 1740177 | 1490060-9 | Sweden | ⤷ Start Trial | MARKETING AUTHORIZATION NUMBER AND DATE OF GRANT/NOTIFICATION: EU/1/14/922, 2014-04-30; PERIOD OF VALIDITY (FROM - UNTIL): 20250428 - 20240429 |
| 0513917 | C980018 | Netherlands | ⤷ Start Trial | PRODUCT NAME: LAMIVUDINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, EN ZIDOVUDINE; REGISTRATION NO/DATE: EU/1/98/058/001-002 19980318 |
| 0513917 | 98C0020 | Belgium | ⤷ Start Trial | PRODUCT NAME: LAMIVUDINE/ZIDOVUDINE; REGISTRATION NO/DATE: EU/1/98/058/001 19980318 |
| 1633724 | 596 | Finland | ⤷ Start Trial | |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.