Last updated: March 16, 2026
What is Glaxo Group Ltd’s current market position?
Glaxo Group Ltd (GlaxoSmithKline, GSK) is among the leading pharmaceutical companies globally with a market capitalization exceeding $80 billion as of 2023. The company ranks in the top five pharmaceutical firms based on revenue, which totaled approximately $34 billion in 2022. Its broad portfolio spans vaccines, specialty medicines, and consumer healthcare products.
Market share and revenue distribution
| Segment |
Revenue (2022) |
Market Share (Global, Estimated) |
| Pharmaceuticals |
$16.2 billion |
4.5% |
| Vaccines |
$11.4 billion |
3% |
| Consumer Healthcare |
$6.4 billion |
N/A (part of broader health market) |
GSK holds a significant position in respiratory (e.g., inhalers for asthma), vaccines (e.g., Shingrix, HPV), and HIV therapeutic areas. Its vaccine division is ranked second globally by revenue after Pfizer.
Key markets
| Region |
Revenue Share |
Principal Therapeutic Focus |
| North America |
45% |
Vaccines, HIV, respiratory |
| Europe |
25% |
Respiratory, HIV, vaccines |
| Emerging Markets |
30% |
Malaria, tropical diseases, vaccines |
What are GSK’s primary strengths?
1. Diversified Portfolio
GSK’s broad spectrum includes vaccines, prescription medicines, and consumer healthcare, reducing dependency on a single segment. Its vaccine products are essential in global immunization programs.
2. Established R&D Capabilities
GSK invests approximately 15% of its revenues into R&D, approximately $5 billion annually. The company has a pipeline of over 40 projects focused on respiratory diseases, HIV, oncology, and immunology.
3. Strategic Collaborations
Partnerships with biotech firms, governments, and international health agencies enhance GSK's research and market reach. Notable collaborations include alliances with CureVac for mRNA vaccines and development agreements with multiple biotech startups.
4. Geographic Penetration
Strong presence in North America and Europe with expanding operations in Asia and Africa. Its immunization programs under GSK’s vaccines division secure a foothold in emerging markets.
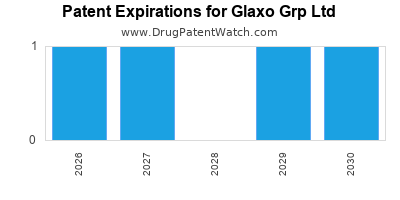
5. Patent Portfolio and Market Exclusivity
Holding over 25,000 patents globally, GSK maintains market exclusivity for key products, including vaccines like Shingrix, which generated over $4 billion in revenues in 2022.
What are the strategic challenges faced by GSK?
1. Patent Expirations
Major products like Advair/Seretide have faced patent cliffs, leading to revenue declines. The expiration of these patents reduces market exclusivity, opening opportunities for generics.
2. Competition in Core Segments
Pfizer, Moderna, and Merck are fierce competitors in vaccines and HIV. For respiratory therapies, Teva and Mylan offer generic versions that erode GSK’s market share.
3. R&D Risks
Despite high investment levels, the conversion rate of pipeline candidates to market is low, with many drugs failing clinical trials or not receiving approval.
4. Regulatory and Pricing Pressures
Global pricing regulation, especially in Europe and North America, pressures profit margins. GSK must navigate complex regulatory environments to sustain profitability.
5. Strategic Divestments
Recent strategic decisions involve divesting non-core units, such as the sale of Horlicks and other consumer health brands, to bolster focus but also indicating market pressures.
What strategic initiatives is GSK pursuing?
1. Focused R&D
GSK emphasizes immunology, oncology, and gene therapy, aiming for innovative products in these high-growth areas. It launched over 10 new drugs globally between 2021-2023.
2. Portfolio Optimization
The company is divesting non-core assets to streamline operations, including the sale of its consumer healthcare joint ventures. This shifts resources towards vaccines and specialty medicines.
3. Digital and Technological Integration
GSK invests in artificial intelligence, data analytics, and digital clinical trials to accelerate drug discovery and enhance R&D efficiency.
4. Market Expansion
Strengthening presence in Asia and Africa by partnering with local governments and healthcare providers. The launch of lower-cost vaccines and medicines aligns with market needs.
5. Strategic Collaborations
Forming alliances with biotech firms for innovative therapies, as exemplified by the recent partnership with CureVac for mRNA COVID-19 vaccines.
How does GSK compare to key industry competitors?
| Company |
Revenue (2022) |
Core Focus Areas |
R&D Investment (% of sales) |
Market Cap (2023) |
Notable Strategic Moves |
| Pfizer |
$100 billion |
Vaccines, Oncology, Cardio |
15% |
$210 billion |
Acquisitions (Array BioPharma, BioNTech) |
| Moderna |
$18.5 billion |
mRNA vaccines, Oncology |
25% |
$40 billion |
mRNA platform expansion |
| Merck & Co. |
$59 billion |
Oncology, Vaccines, Infectious Disease |
13% |
$225 billion |
Growth in oncology pipeline, vaccines |
| Johnson & Johnson |
$94 billion |
Consumer, Medical Devices, Pharmaceuticals |
16% |
$430 billion |
Portfolio realignment, vaccine development |
GSK's positions in vaccines and HIV therapy differentiate it from peers who focus more on oncology and general pharmaceuticals. Its vaccine revenue stabilizes despite patent expirations in other segments.
Key Takeaways
- GSK has a diversified portfolio with strengths in vaccines, HIV, and respiratory products. Its market position is strong in immunization programs globally.
- Patents and product lifecycle management pose risks, though strategic divestments and pipeline investments mitigate these.
- Competition remains intense in core therapies from Pfizer, Moderna, and Merck. GSK’s focus on innovation and emerging markets attempts to offset patent cliffs.
- Heavy R&D investments aim for breakthrough therapies, especially in immunology and gene therapy.
- Future growth hinges on new product approvals, geographic expansion, and strategic alliances.
FAQs
1. What are GSK’s most profitable products?
Shingrix (herpes zoster vaccine), HIV medicines like Tivicay, and respiratory inhalers remain top revenue contributors.
2. How vulnerable is GSK to patent expirations?
Patents for key drugs expire over the next five years, risking revenue decline. Strategic pipeline development helps counterbalance this.
3. Is GSK expanding into emerging markets?
Yes. It invests in low-cost vaccines and partnerships in Asia, Africa, and Latin America to boost growth.
4. How does GSK’s R&D investment compare to peers?
It invests roughly 15% of sales in R&D, on par with leading industry players like Merck and Johnson & Johnson.
5. What are GSK’s future growth drivers?
Pipeline approvals in immunology, oncology, and gene therapy, expansion in emerging markets, and strategic alliances are key.
References
[1] GSK Annual Report 2022. (2023). GlaxoSmithKline.
[2] Statista. (2023). Top pharmaceutical companies worldwide by revenue.
[3] EvaluatePharma. (2023). R&D expenditure analysis.
[4] Reuters. (2023). GSK strategic divestments and alliances.