TRELEGY ELLIPTA Drug Patent Profile
✉ Email this page to a colleague
When do Trelegy Ellipta patents expire, and what generic alternatives are available?
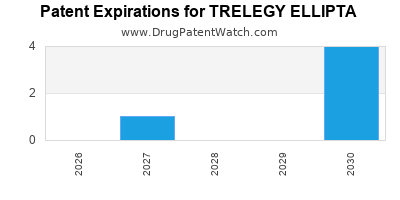
Trelegy Ellipta is a drug marketed by Glaxosmithkline and is included in one NDA. There are six patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and ninety-nine patent family members in forty-nine countries.
The generic ingredient in TRELEGY ELLIPTA is fluticasone furoate; umeclidinium bromide; vilanterol trifenatate. There are twenty-nine drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the fluticasone furoate; umeclidinium bromide; vilanterol trifenatate profile page.
DrugPatentWatch® Generic Entry Outlook for Trelegy Ellipta
Trelegy Ellipta was eligible for patent challenges on May 10, 2017.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be November 29, 2030. This may change due to patent challenges or generic licensing.
There have been six patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for TRELEGY ELLIPTA?
- What are the global sales for TRELEGY ELLIPTA?
- What is Average Wholesale Price for TRELEGY ELLIPTA?
Summary for TRELEGY ELLIPTA
| International Patents: | 199 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 5 |
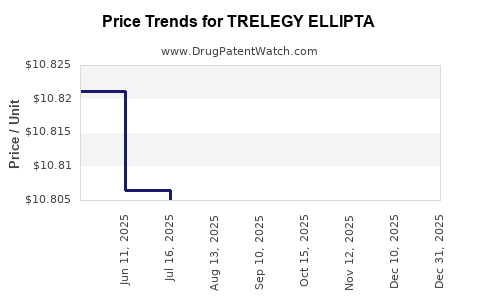
| Drug Prices: | Drug price information for TRELEGY ELLIPTA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TRELEGY ELLIPTA |
| What excipients (inactive ingredients) are in TRELEGY ELLIPTA? | TRELEGY ELLIPTA excipients list |
| DailyMed Link: | TRELEGY ELLIPTA at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TRELEGY ELLIPTA
Generic Entry Date for TRELEGY ELLIPTA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
POWDER;INHALATION |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for TRELEGY ELLIPTA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Alabama at Birmingham | PHASE2 |
| Baylor Research Institute | Phase 4 |
| GlaxoSmithKline | Phase 4 |
Pharmacology for TRELEGY ELLIPTA
Paragraph IV (Patent) Challenges for TRELEGY ELLIPTA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TRELEGY ELLIPTA | Powder for Inhalation | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | 100 mcg/62.5 mcg/ 25 mcg | 209482 | 1 | 2025-11-26 |
US Patents and Regulatory Information for TRELEGY ELLIPTA
TRELEGY ELLIPTA is protected by six US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TRELEGY ELLIPTA is ⤷ Start Trial.
This potential generic entry date is based on patent 12,396,986.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | RX | Yes | Yes | 11,090,294 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-002 | Sep 9, 2020 | RX | Yes | Yes | 7,488,827 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | RX | Yes | Yes | 7,488,827 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TRELEGY ELLIPTA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | 8,309,572 | ⤷ Start Trial |
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-002 | Sep 9, 2020 | 6,537,983 | ⤷ Start Trial |
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | 6,878,698 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for TRELEGY ELLIPTA
When does loss-of-exclusivity occur for TRELEGY ELLIPTA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 10326798
Patent: Combinations of a muscarinic receptor antagonist and a beta-2 adrenoreceptor agonist
Estimated Expiration: ⤷ Start Trial
Patent: 14204459
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Patent: 16262698
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Patent: 17238392
Patent: Mannose-derived antagonists of FimH useful for treating disease
Estimated Expiration: ⤷ Start Trial
Patent: 18282427
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Patent: 21204302
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Patent: 23219901
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2012012925
Patent: produto de combinação farmacêutico, inalador de pó seco, e, uso de um produto
Estimated Expiration: ⤷ Start Trial
Patent: 2018069147
Patent: composto da fórmula iii, composição farmacêutica, método de inibição da função de fimh e método de tratamento de uma doença mediada por fimh
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 81487
Patent: COMBINAISONS D'UN ANTAGONISTE DE RECEPTEUR MUSCARINIQUE ET D'UN AGONISTE DU RECEPTEUR BETA-2 ADRENERGIQUE (COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST)
Estimated Expiration: ⤷ Start Trial
Patent: 18345
Patent: ANTAGONISTES DERIVES DU MANNOSE DE FIMH UTILES POUR LE TRAITEMENT D'UNE MALADIE (MANNOSE-DERIVED ANTAGONISTS OF FIMH USEFUL FOR TREATING DISEASE)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 12001432
Patent: Producto de combinacion farmacéutica que comprende bromuro de 4-[hidroxi-(difenil)-metil]-1-{2-[(fenil-metil)-oxi]-etil}-1-azonia-biciclo-[2.2.2]-octano y trifenil-acetato de 4-{(1r)-2-[(6-{2-[(2,6-dicloro-bencil)-oxi]-etoxi}-hexil)-amino]-1-hidroxi-etil}-2-(hidroxi-metil)-fenol; y su uso para tratar enfermedades respiratorias.
Estimated Expiration: ⤷ Start Trial
China
Patent: 2724974
Patent: Combinations of a muscarinic receptor antagonist and a beta-2 adrenoreceptor agonist
Estimated Expiration: ⤷ Start Trial
Patent: 7412229
Patent: 毒蕈碱受体拮抗剂和β‑2肾上腺素受体激动剂的组合 (COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST)
Estimated Expiration: ⤷ Start Trial
Patent: 8778288
Patent: 可用于治疗疾病的FimH的甘露糖衍生的拮抗剂 (Mannose-derived antagonists of FimH useful for treating disease)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 41613
Patent: COMBINACIONES DE UN ANTAGONISTA DE LOS RECEPTORES MUSCARÍNICOS Y UN AGONISTA DEL ADRENO-RECEPTOR BETA 2
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 120265
Patent: COMBINACIONES DE UN ANTAGONISTA DE LOS RECEPTORES MUSCARÍNICOS Y UN AGONISTA DEL ADRENO-RECEPTOR BETA-2
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0180312
Estimated Expiration: ⤷ Start Trial
Patent: 0240688
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 20058
Estimated Expiration: ⤷ Start Trial
Patent: 18017
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 06844
Estimated Expiration: ⤷ Start Trial
Patent: 35707
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 012000148
Patent: COMBINACIONES DE UN ANTAGONISTA DE LOS RECEPTORES MUSCARINICOS Y UN AGONISTA DEL ADRENO-RECEPTOR BETA-2
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 3839
Patent: КОМБИНАЦИИ АНТАГОНИСТА МУСКАРИНОВЫХ РЕЦЕПТОРОВ И АГОНИСТА БЕТА-2-АДРЕНОРЕЦЕПТОРОВ (COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST)
Estimated Expiration: ⤷ Start Trial
Patent: 1290266
Patent: КОМБИНАЦИИ АНТАГОНИСТА МУСКАРИНОВЫХ РЕЦЕПТОРОВ И АГОНИСТА БЕТА-2-АДРЕНОРЕЦЕПТОРОВ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 06844
Patent: Combinaisons d'un antagoniste de récepteur muscarinique et d'un agoniste du récepteur bêta-2 adrénergique (Combinations of a muscarinic receptor antagonist and a beta-2 adrenoreceptor agonist)
Estimated Expiration: ⤷ Start Trial
Patent: 35707
Patent: COMBINAISONS D'UN ANTAGONISTE DE RÉCEPTEUR MUSCARINIQUE ET D'UN AGONISTE DU RÉCEPTEUR BÊTA-2 ADRÉNERGIQUE (COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST)
Estimated Expiration: ⤷ Start Trial
Patent: 32892
Patent: ANTAGONISTES DÉRIVÉS DU MANNOSE DE FIMH UTILES POUR LE TRAITEMENT D'UNE MALADIE (MANNOSE-DERIVED ANTAGONISTS OF FIMH USEFUL FOR TREATING DISEASE)
Estimated Expiration: ⤷ Start Trial
Patent: 54645
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 35707
Estimated Expiration: ⤷ Start Trial
Patent: 0180019
Estimated Expiration: ⤷ Start Trial
France
Patent: C1022
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 49407
Patent: 毒蕈碱受體拮抗劑和β-2腎上腺素受體激動劑的組合 (COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 36216
Estimated Expiration: ⤷ Start Trial
Patent: 66359
Estimated Expiration: ⤷ Start Trial
Patent: 800027
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 9893
Patent: שילובים של אנטגוניסט לקולטן מוסקריני ואגוניסט לקולטן בתא-2-אדרנו (Combinations of a muscarinic receptor antagonist and a beta-2 adrenorecptor agonist)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 16631
Estimated Expiration: ⤷ Start Trial
Patent: 13512270
Estimated Expiration: ⤷ Start Trial
Patent: 19509315
Patent: 疾患の処置に有用なFimHのマンノース由来アンタゴニスト
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 06844
Estimated Expiration: ⤷ Start Trial
Patent: 35707
Estimated Expiration: ⤷ Start Trial
Patent: 506844
Estimated Expiration: ⤷ Start Trial
Patent: 2018011
Estimated Expiration: ⤷ Start Trial
Luxembourg
Patent: 0077
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 4864
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Patent: 7466
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 1290
Patent: COMBINACIONES DE UN ANTAGONISTA DE LOS RECEPTORES MUSCARÍNICOS Y UN AGONISTA DEL ADRENO-RECEPTOR BETA-2. (COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST.)
Estimated Expiration: ⤷ Start Trial
Patent: 12006310
Patent: COMBINACIONES DE UN ANTAGONISTA DE LOS RECEPTORES MUSCARINICOS Y UN AGONISTA DEL ADRENO-RECEPTOR BETA-2. (COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST.)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 965
Patent: KOMBINACIJE ANTAGONISTA MUSKARINSKOG RECEPTORA I AGONISTA ВЕТА - 2 ADRENORECEPTORA (Combinations of a muscarinic receptor antagonist and a beta-2 adrenoreceptor agonist)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 853
Patent: تركيبة مضاد استقبال المسكارينية ومحضر استقبال بتي ـ 2 الأدرينالية
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 0026
Patent: Combinations of a muscarinic receptor antagonist and a beta-2 adrenoreceptor agonist
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 18018
Estimated Expiration: ⤷ Start Trial
Patent: 06844
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 130042
Patent: COMBINACIONES DE UN ANTAGONISTA DE LOS RECEPTORES MUSCARINICOS Y UN AGONISTA DEL ADRENO-RECEPTOR BETA-2
Estimated Expiration: ⤷ Start Trial
Patent: 170915
Patent: COMBINACIONES DE UN ANTAGONISTA DE LOS RECEPTORES MUSCARINICOS Y UN AGONISTA DEL ADRENO-RECEPTOR BETA-2
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 012501083
Patent: COMBINATIONS OF MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 06844
Estimated Expiration: ⤷ Start Trial
Patent: 35707
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 06844
Estimated Expiration: ⤷ Start Trial
Patent: 35707
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 18131440
Patent: Антагонисты FimH, являющиеся производными маннозы, пригодные для лечения заболевания
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01800129
Estimated Expiration: ⤷ Start Trial
Patent: 02400176
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 848
Patent: KOMBINACIJE ANTAGONISTA MUSKARINSKOG RECEPTORA I AGONISTA BETA-2 ADRENORECEPTORA (COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST)
Estimated Expiration: ⤷ Start Trial
Patent: 494
Patent: KOMBINACIJE ANTAGONISTA MUSKARINSKOG RECEPTORA I AGONISTA BETA-2-ADRENORECEPTORA (COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201407864U
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Patent: 1087
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 06844
Estimated Expiration: ⤷ Start Trial
Patent: 35707
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1203890
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1742140
Estimated Expiration: ⤷ Start Trial
Patent: 1830728
Estimated Expiration: ⤷ Start Trial
Patent: 120092163
Patent: COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST
Estimated Expiration: ⤷ Start Trial
Patent: 170061719
Patent: 무스카린성 수용체 길항제 및 베타-2 아드레날린 수용체 효능제의 조합물 (- COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST)
Estimated Expiration: ⤷ Start Trial
Patent: 180128937
Patent: 질환을 치료하는데 유용한 FIMH의 만노스-유래 길항제
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 59330
Estimated Expiration: ⤷ Start Trial
Patent: 79060
Estimated Expiration: ⤷ Start Trial
Turkey
Patent: 1802921
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 6775
Patent: КОМБІНАЦІЯ АНТАГОНІСТА МУСКАРИНОВОГО РЕЦЕПТОРА ТА АГОНІСТА БЕТА-2 АДРЕНОРЕЦЕПТОРА (COMBINATIONS OF MUSCARINIC ACETYLCHOLINE RECEPTOR ANTAGONIST AND BETA 2 AGONIST)
Estimated Expiration: ⤷ Start Trial
United Kingdom
Patent: 21075
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TRELEGY ELLIPTA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Malaysia | 129796 | NOVEL ANTI-INFLAMMATORY ANDROSTANE DERIVATIVES | ⤷ Start Trial |
| Hong Kong | 1249407 | ⤷ Start Trial | |
| Peru | 20130042 | COMBINACIONES DE UN ANTAGONISTA DE LOS RECEPTORES MUSCARINICOS Y UN AGONISTA DEL ADRENO-RECEPTOR BETA-2 | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TRELEGY ELLIPTA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2506844 | 300942 | Netherlands | ⤷ Start Trial | PRODUCT NAME: EEN FARMACEUTISCH COMBINATIEPRODUCT OMVATTENDE EEN FARMACEUTISCH AANVAARDBAAR ZOUT VAN UMECLIDINIUM (BIJVOORBEELD UMECLIDINIUMBROMIDE), VILANTEROL OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN (BIJVOORBEELD VILANTEROLTRIFENATAAT) EN FLUTICASONFUROAAT; REGISTRATION NO/DATE: EU/1/17/1236 20171117 |
| 1740177 | C01740177/02 | Switzerland | ⤷ Start Trial | PRODUCT NAME: UMECLIDINIUM BROMID + VILANTEROL; REGISTRATION NO/DATE: SWISSMEDIC 63152 14.07.2014 |
| 1425001 | 2014C/026 | Belgium | ⤷ Start Trial | PRODUCT NAME: VILANTEROL TRIFENATATE; AUTHORISATION NUMBER AND DATE: EU/1/13/886/001 20131114 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for TRELEGY ELLIPTA
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.