Alcon Labs Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ALCON LABS INC, and what generic alternatives to ALCON LABS INC drugs are available?
ALCON LABS INC has seventeen approved drugs.
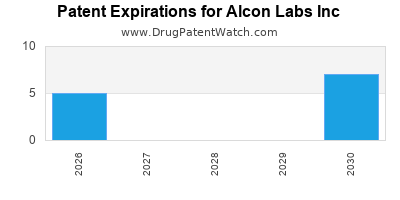
There are thirty-nine US patents protecting ALCON LABS INC drugs.
There are two hundred and seventeen patent family members on ALCON LABS INC drugs in thirty-one countries and twenty-seven supplementary protection certificates in ten countries.
Summary for Alcon Labs Inc
| International Patents: | 217 |
| US Patents: | 39 |
| Tradenames: | 14 |
| Ingredients: | 12 |
| NDAs: | 17 |
Drugs and US Patents for Alcon Labs Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alcon Labs Inc | EYSUVIS | loteprednol etabonate | SUSPENSION/DROPS;OPHTHALMIC | 210933-001 | Oct 26, 2020 | RX | Yes | Yes | 9,056,057 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Alcon Labs Inc | EYSUVIS | loteprednol etabonate | SUSPENSION/DROPS;OPHTHALMIC | 210933-001 | Oct 26, 2020 | RX | Yes | Yes | 10,646,436 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Alcon Labs Inc | INVELTYS | loteprednol etabonate | SUSPENSION/DROPS;OPHTHALMIC | 210565-001 | Aug 22, 2018 | RX | Yes | Yes | 10,688,045 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Alcon Labs Inc | RHOPRESSA | netarsudil mesylate | SOLUTION/DROPS;OPHTHALMIC | 208254-001 | Dec 18, 2017 | RX | Yes | Yes | 10,588,901 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Alcon Labs Inc | RHOPRESSA | netarsudil mesylate | SOLUTION/DROPS;OPHTHALMIC | 208254-001 | Dec 18, 2017 | RX | Yes | Yes | 10,654,844 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Alcon Labs Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Alcon Labs Inc | PATADAY TWICE DAILY RELIEF | olopatadine hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 020688-001 | Dec 18, 1996 | 4,923,892 | ⤷ Try a Trial |
| Alcon Labs Inc | PATADAY ONCE DAILY RELIEF | olopatadine hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 021545-001 | Dec 22, 2004 | 7,402,609*PED | ⤷ Try a Trial |
| Alcon Labs Inc | PATADAY ONCE DAILY RELIEF | olopatadine hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 021545-001 | Dec 22, 2004 | 5,116,863*PED | ⤷ Try a Trial |
| Alcon Labs Inc | PATADAY TWICE DAILY RELIEF | olopatadine hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 020688-001 | Dec 18, 1996 | 4,871,865 | ⤷ Try a Trial |
| Alcon Labs Inc | PATADAY TWICE DAILY RELIEF | olopatadine hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 020688-001 | Dec 18, 1996 | 5,116,863*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Alcon Labs Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Hong Kong | 1225733 | ⤷ Try a Trial |
| Spain | 2584858 | ⤷ Try a Trial |
| Cyprus | 1117835 | ⤷ Try a Trial |
| Denmark | 2442790 | ⤷ Try a Trial |
| Mexico | 2014013315 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Alcon Labs Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3461484 | 21C1024 | France | ⤷ Try a Trial | PRODUCT NAME: ASSOCIATION DE NETARSUDIL OU L'UN DE SES SELS ET DE LATANOPROST; REGISTRATION NO/DATE: EU/1/20/1502 20210108 |
| 3043773 | 2022C/520 | Belgium | ⤷ Try a Trial | PRODUCT NAME: MOMETASONE OF EEN ZOUT HIERVAN EN OLOPATADINE OF EEN ZOUT HIERVAN; AUTHORISATION NUMBER AND DATE: BE595626 20220203 |
| 1631293 | 2014C/042 | Belgium | ⤷ Try a Trial | PRODUCT NAME: MIRVASO (BRIMONIDINE) EN FARMACEUTISCHE ZOUTEN DAARVAN VOOR GEBRUIK ALS MEDICIJN VOOR HET BEHANDELEN VAN ROSACEA GEINDUCEERDE ROODHEID; AUTHORISATION NUMBER AND DATE: EU/1/13/904 20140221 |
| 1631293 | 300683 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDINE EN FARMACEUTISCH AANVAARDBARE ZOUTEN DAARVAN VOOR TOEPASSING ALS GENEESMIDDEL VOOR DE BEHANDELING VAN DOOR ROSACEA GEINDUCEERDE ROODHEID; REGISTRATION NO/DATE: EU/1/13/904 20140225 |
| 1631293 | 92462 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDINE ET SES SELS PHARMACEUTIQUES POUR L UTILISATION COMME MEDICAMENT POUR LE TRAITEMENT DES ROUGEURS INDUITES PAR LA ROSACEA.FIRST REGISTRATION: 20140225 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.