Last updated: February 19, 2026
This analysis examines the market dynamics and financial trajectory of tolterodine tartrate, a muscarinic receptor antagonist used to treat overactive bladder (OAB). The drug faces significant generic competition, impacting its revenue potential. Key factors influencing its market position include patent expiries, generic market penetration, and the evolving OAB treatment landscape.
What is Tolterodine Tartrate and Its Therapeutic Use?
Tolterodine tartrate is a selective muscarinic receptor antagonist primarily prescribed for the treatment of urinary urgency, frequency, and incontinence associated with OAB. It functions by inhibiting the binding of acetylcholine to muscarinic receptors in the bladder detrusor muscle, thereby reducing involuntary bladder contractions.
The drug is available in various formulations, including immediate-release (IR) and extended-release (ER) tablets. The ER formulation, such as Detrol® LA (Pfizer), was developed to provide a more convenient dosing schedule and potentially improve patient adherence.
Key indications for tolterodine tartrate include:
- Symptomatic treatment of overactive bladder.
- Reduction of urinary urgency, frequency, and urge incontinence.
What is the Patent Landscape for Tolterodine Tartrate?
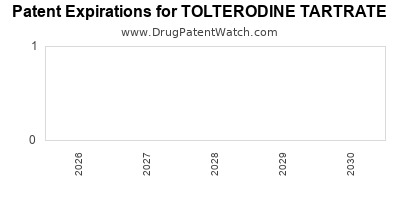
The original patents protecting tolterodine tartrate have expired, paving the way for generic market entry. Pfizer’s Detrol® and Detrol® LA were the originator products.
- Original Composition of Matter Patent: The foundational patent for tolterodine expired in the early to mid-2000s. Specific dates vary by jurisdiction, but U.S. patent protection for the active ingredient generally concluded by 2006.
- Formulation Patents: Patents covering specific extended-release formulations, such as Detrol® LA, also faced expiry. For Detrol® LA, key patent protections concluded around 2010 in the U.S.
- Market Exclusivity: Following patent expiry, the market opened to generic manufacturers. The entry of generics significantly impacted the pricing and revenue of the originator product.
How Has the Generic Market for Tolterodine Tartrate Evolved?
The advent of generic tolterodine tartrate has reshaped the market significantly.
- Entry of Generic Manufacturers: Numerous pharmaceutical companies have introduced generic versions of tolterodine tartrate IR and ER tablets since the expiry of key patents. Major players in the generic space now offer these products.
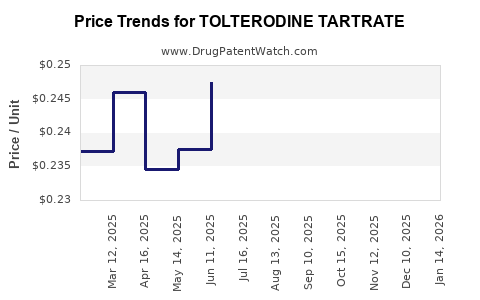
- Price Erosion: The presence of multiple generic competitors leads to substantial price reductions compared to the originator brand. This price erosion is a standard characteristic of markets after patent expiry.
- Market Share Shift: Generic tolterodine tartrate formulations now constitute the vast majority of prescriptions and sales volume for the drug class. Brand loyalty and physician preference for originator products diminish as lower-cost generic alternatives become widely available and accepted.
- Regulatory Approvals: Generic versions require approval from regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Approvals are granted based on demonstrating bioequivalence to the originator product.
What is the Current Market Size and Projected Growth for Tolterodine Tartrate?
The global market for tolterodine tartrate is substantial but characterized by mature growth and significant price competition due to its generic status.
- Market Size: While precise, up-to-the-minute figures for tolterodine tartrate alone are difficult to isolate due to its broad categorization within OAB treatments and generic drug markets, the overall OAB therapeutics market is estimated to be in the billions of dollars globally. Tolterodine tartrate, as a widely prescribed generic, captures a significant portion of this market by volume. Estimates suggest the global market for tolterodine tartrate sales (all generic manufacturers combined) is in the hundreds of millions of dollars annually.
- Growth Projection: The market for tolterodine tartrate is expected to exhibit low single-digit compound annual growth rates (CAGR), if any, in developed markets. Growth will be driven primarily by increasing OAB prevalence due to aging populations and improved diagnosis rates, rather than by new product innovation or market expansion.
- Regional Dynamics: The U.S. and European markets represent the largest shares due to higher healthcare spending and established OAB treatment protocols. Emerging markets may show slightly higher growth potential as access to diagnostics and treatments improves.
What are the Competitive Dynamics and Key Players in the Tolterodine Tartrate Market?
The competitive landscape is dominated by generic manufacturers, with limited innovation occurring post-patent expiry.
- Key Generic Manufacturers: Companies such as Teva Pharmaceutical Industries, Mylan (now Viatris), Sun Pharmaceutical Industries, Aurobindo Pharma, and Accord Healthcare are prominent suppliers of generic tolterodine tartrate.
- Competition Factors: Competition is primarily based on price, supply chain reliability, and distribution networks.
- Lack of Product Differentiation: Beyond IR and ER formulations, there is little to differentiate generic tolterodine tartrate products from one another. The focus is on cost-effectiveness.
- Emerging Therapies: While tolterodine tartrate is established, it competes with other OAB treatments, including other anticholinergics (e.g., oxybutynin, solifenacin), beta-3 adrenergic agonists (e.g., mirabegron), and newer therapeutic modalities like onabotulinumtoxinA injections and neuromodulation. These newer therapies may target specific patient segments or offer different efficacy/side effect profiles, influencing tolterodine tartrate's market share.
What are the Financial Implications and Revenue Trends for Tolterodine Tartrate?
The financial trajectory of tolterodine tartrate is largely dictated by its generic status.
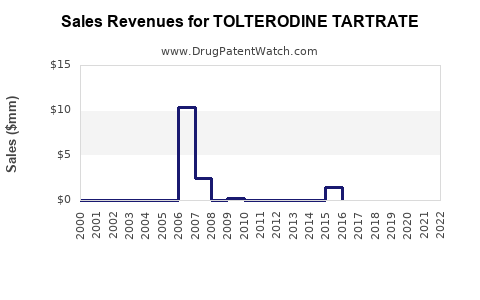
- Originator Product Decline: Pfizer's Detrol® and Detrol® LA experienced significant revenue decline following generic entry. This is typical for branded drugs facing robust generic competition.
- Generic Revenue Contribution: The collective revenue generated by all generic tolterodine tartrate manufacturers is substantial but fragmented. Individual generic manufacturers generate revenue based on their market share, pricing strategies, and manufacturing efficiency.
- Pricing Pressure: Continuous pricing pressure from payers, pharmacy benefit managers (PBMs), and direct competition limits revenue upside for generic manufacturers. Prices for generic tolterodine tartrate can fluctuate based on supply, demand, and competitive bidding.
- Profit Margins: While volumes are high, profit margins for generic tolterodine tartrate are generally thinner compared to branded drugs, particularly for well-established generics with intense competition. Manufacturers rely on economies of scale and efficient production to maintain profitability.
- Cost of Goods Sold (COGS): Active pharmaceutical ingredient (API) sourcing, manufacturing costs, and regulatory compliance are key components of COGS. Efficient supply chain management is crucial for profitability.
What are the Challenges and Opportunities for Tolterodine Tartrate Manufacturers?
Manufacturers face a landscape defined by cost management and market access.
Challenges:
- Intense Price Competition: The primary challenge is the downward pressure on prices from a crowded generic market.
- Regulatory Scrutiny: Ensuring ongoing compliance with evolving manufacturing standards and quality control is critical.
- Supply Chain Vulnerabilities: Reliance on global API suppliers can expose manufacturers to supply disruptions.
- Evolving Treatment Guidelines: Shifts in medical guidelines favoring newer or different OAB therapies could impact prescription volumes.
- Antimicrobial Resistance Concerns (Indirect): While not directly antimicrobial, the broader regulatory and public health focus on drug safety and resistance may indirectly influence scrutiny of all pharmaceutical production and environmental impact.
Opportunities:
- Market Penetration in Emerging Economies: Expanding access and distribution in regions where OAB treatment is less established presents growth opportunities.
- Supply Chain Optimization: Companies that can secure cost-effective API sources and streamline manufacturing processes can gain a competitive edge.
- Portfolio Diversification: Leveraging existing manufacturing capabilities to produce other generics or related therapeutic areas can mitigate reliance on a single product.
- Contract Manufacturing: Providing manufacturing services for other pharmaceutical companies can be a supplementary revenue stream.
- Long-Term Demand: The persistent prevalence of OAB, particularly in aging populations, ensures continued demand for effective and affordable treatments like tolterodine tartrate.
Key Takeaways
Tolterodine tartrate is a mature generic pharmaceutical product facing significant price competition. Its market trajectory is characterized by stable but low single-digit growth, driven by OAB prevalence, and is dominated by numerous generic manufacturers. Profitability relies heavily on efficient manufacturing, cost control, and robust supply chain management. While significant new revenue growth is unlikely, continued demand ensures its place as a staple treatment for overactive bladder.
Frequently Asked Questions
- What is the current average wholesale acquisition cost (WAC) for a 30-day supply of generic tolterodine tartrate?
The WAC for a 30-day supply of generic tolterodine tartrate (e.g., 4mg extended-release capsules) typically ranges from $10 to $30, depending on the manufacturer, pharmacy, and contracted pricing. This figure is subject to change based on market dynamics and payer agreements.
- Are there any ongoing clinical trials for novel formulations or delivery methods of tolterodine tartrate?
As of late 2023, there are no significant, publicly disclosed clinical trials for novel formulations or new delivery methods of tolterodine tartrate. The focus in OAB therapeutics has shifted towards newer drug classes and combination therapies.
- What are the primary side effects that limit tolterodine tartrate's use in certain patient populations?
Common side effects of tolterodine tartrate include dry mouth, constipation, blurred vision, and cognitive impairment (especially in the elderly). These side effects can limit its use in patients with pre-existing conditions such as glaucoma, severe constipation, or cognitive dysfunction.
- How does tolterodine tartrate compare in efficacy and safety to other first-line OAB medications like oxybutynin?
Tolterodine tartrate, particularly the extended-release formulation, is generally considered to have a more favorable side effect profile, specifically reduced dry mouth and constipation, compared to immediate-release oxybutynin. Efficacy is comparable for many patients, though individual responses vary.
- What is the projected impact of biosimil competition on the market for tolterodine tartrate?
Biosimil competition is not applicable to small molecule drugs like tolterodine tartrate. The relevant competitive dynamic is generic drug entry, which has already occurred and significantly impacted the market.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/drugs-information- Mugs-consumers/search-drug-database (Note: Specific patent expiry dates are not publicly listed here but are derived from patent filings and legal databases).
[2] Pfizer Inc. (2004). Detrol® LA (tolterodine tartrate) extended release capsules prescribing information.
[3] Various generic pharmaceutical manufacturers' product information and market reports. (Access dates vary).
[4] Global market research reports on the Overactive Bladder therapeutics market. (Reports from firms like Grand View Research, MarketsandMarkets, Allied Market Research; specific report dates vary).