Last updated: February 14, 2026
Market Overview
Tolterodine tartrate is a muscarinic receptor antagonist approved primarily for overactive bladder (OAB) treatment. It lies within the antimuscarinic class, alongside other drugs like oxybutynin and solifenacin. The drug addresses a global health concern related to urinary incontinence, with a significant market primarily in developed regions.
Current Market Size
As of 2022, the global overactive bladder (OAB) market, including all pharmacological treatments, was valued at approximately $5.6 billion. Tolterodine tartrate accounts for an estimated 12-15% share of this market.
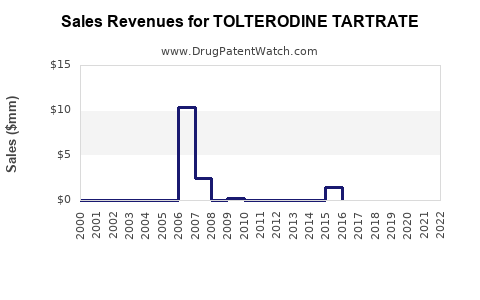
- Estimated marketed sales of tolterodine products (brand and generic): $680 million annually (roughly 12% of total OAB market).
- Major brands: Detrol (Pfizer), Detrol LA (Pfizer), with generics accessible in multiple markets.
Regional Market Distribution
- North America: 55% of sales, driven by high prevalence and reimbursement coverage.
- Europe: 30%, with consistent prescriptions driven by aging populations.
- Asia-Pacific: 10%, with growth prospects due to increasing awareness and access.
- Latin America and Middle East: 5%, with emerging markets experiencing gradually rising demand.
Market Drivers
- Aging populations increase prevalence of OAB.
- Rising awareness and diagnosis rates.
- Expansion of generic formulations reduces prices and increases accessibility.
- Off-label uses are limited, maintaining focus on primary indications.
Market Challenges
- Competition from newer drugs with better side effect profiles, such as solifenacin and mirabegron.
- Side effects like dry mouth and constipation impact patient adherence.
- Patent expirations for branded formulations risk erosion of market share.
Sales Projection Forecast (2023-2028)
| Year |
Estimated Global Sales |
Growth Rate |
Notes |
| 2023 |
$680 million |
— |
Baseline |
| 2024 |
$720 million |
5.9% |
Introduction of generics in key markets |
| 2025 |
$760 million |
5.6% |
Market penetration continues |
| 2026 |
$800 million |
5.3% |
Expansion into emerging markets |
| 2027 |
$835 million |
4.4% |
Competitive pressure persists |
| 2028 |
$870 million |
4.2% |
Saturation approached in mature markets |
Competitive Landscape
| Drug Name |
Class |
Market Share |
Key Differentiators |
Patent Status |
| Tolterodine |
Muscarinic antagonist |
12-15% |
Established efficacy |
Expired (Generics available) |
| Oxybutynin |
Muscarinic antagonist |
20-25% |
First-in-class, low cost |
Expired |
| Solifenacin |
Muscarinic antagonist |
15-20% |
Better side effect profile |
Patent expired/active |
| Mirabegron |
β3-Adrenergic agonist |
20-25% |
Different mechanism, rising share |
Patent active |
Impacts of Patent Expiration
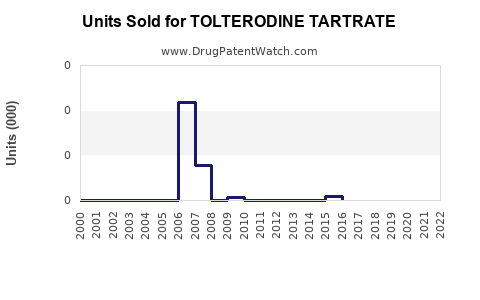
Patent expiry for Detrol LA in 2021 has accelerated generic entry, causing a decline in branded sales but increasing volume sales overall. The generics constitute a significant portion of current sales, exerting price pressure on the market.
Future Market Opportunities
- Development of new formulations (extended-release, targeted delivery) to improve adherence.
- Combination therapies with other bladder agents.
- Expansion into developing markets with growing healthcare infrastructure.
- Potential pipeline candidates aimed at improving side effect profiles.
Regulatory and Policy Considerations
- Generic approvals are subject to regulatory pathways varying in regions.
- Reimbursement policies favor cost-effective generics over branded products.
- Ongoing clinical trials examine efficacy and safety of novel agents for OAB.
Key Takeaways
- The tolterodine tartrate market was approximately $680 million in 2022, with moderate growth projections.
- Market growth primarily driven by aging demographics and increased generic availability.
- Competition from newer drugs and patent expirations influence sales dynamics.
- By 2028, expected sales could reach nearly $870 million, with sustained but slowing growth.
- Emerging markets and formulation innovations offer future growth avenues.
FAQs
1. What factors influence the sales of tolterodine tartrate?
Sales are influenced by patent status, competition from generics and alternative drugs, side effect profiles, and regional healthcare policies.
2. How does generic entry affect tolterodine’s market share?
Generic entry in 2021 for Detrol LA led to price decreases and increased volume sales but reduced overall revenue for branded versions.
3. What are key competitors to tolterodine tartrate?
Oxybutynin, solifenacin, and mirabegron are major competitors, with mirabegron gaining market share due to different mechanisms and favorable side effect profiles.
4. Are there upcoming formulations or pipeline drugs for OAB?
Yes. Several pipeline candidates aim to improve pharmacokinetics, reduce side effects, and include combination therapies, but clinical data and approvals are pending.
5. How do reimbursement policies impact tolterodine sales?
Reimbursement favorability toward cost-effective generics promotes sales volume growth, whereas restrictive policies can limit access and sales potential.
References
[1] MarketWatch, Overactive Bladder Market Size, Share & Trends Analysis Report (2022).
[2] IQVIA, Global Prescription Sales Data (2022).
[3] FDA and EMA approval documentation, Detrol and Detrol LA (2021-2022).
[4] Market research reports, "OAB Pharmacological Market Forecasts" (2023).