Last updated: April 22, 2026
How has risperidone’s market position evolved?
Risperidone is a long-standing, high-volume antipsychotic that transitioned into a mature-and-generic market. The commercial center of gravity has shifted from branded penetration to broad generic availability, driving unit-price compression and steady volume, with earnings increasingly tied to manufacturing scale, gross margin discipline, and regional mix.
Market structure (high level)
- Originator-led phase: branded risperidone with patent-protected market exclusivity in early years.
- Generic-led phase: after expiries, multiple entrants drove price competition.
- Therapeutic durability: ongoing demand from schizophrenia and bipolar disorder indications supported baseline volume even as pricing fell.
What are the key demand drivers for risperidone?
Risperidone demand tracks to psychiatric disease prevalence and regimen persistence, with specific drivers that typically stabilize volume in mature categories:
-
Stable addressable patient base
- Schizophrenia remains a long-duration treatment setting.
- Bipolar disorder maintenance increases the likelihood of continued prescriptions in chronic patients.
-
Formulation breadth supporting adherence
- Oral tablets cover acute and maintenance use.
- Long-acting injectable (LAI) variants support persistence by reducing missed doses.
-
Clinical practice patterns
- Clinicians often use established antipsychotics with known tolerability profiles, supporting ongoing prescribing in cost-sensitive markets after generic entry.
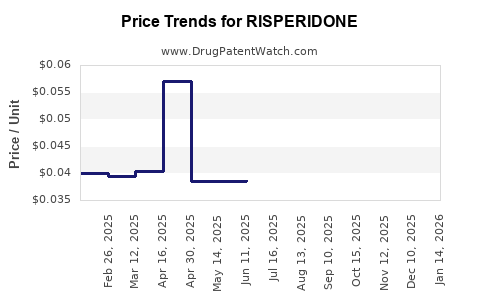
What market dynamics pressured prices and margins?
Risperidone pricing reflects generic competition intensity and the breadth of supplier networks.
Primary margin pressures
- Generic substitution: pharmacy-level switching to lower-cost equivalents.
- Regional tendering and reimbursement: payers increasingly select lowest-cost options.
- Supply normalization post-launch of multiple generics: competitive intensity rises after the first wave of entrants.
Common commercial outcomes for mature generics
- Revenue often becomes less sensitive to new patient starts and more sensitive to:
- persistence,
- share of formulary placements,
- inventory cycles and wholesaler purchasing behavior,
- contract pricing.
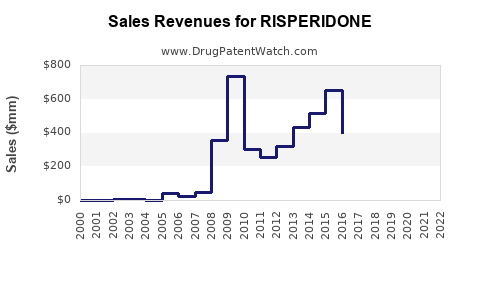
How should the financial trajectory be read for risperidone?
Risperidone’s financial trajectory in most markets follows a recognizable mature-drug pattern:
Trajectory shape
- Revenue: peaks during brand exclusivity; then declines as generics capture share.
- Gross margin: compresses after generic proliferation; recovery only occurs when a firm holds cost leadership or specialized segments (for example, LAI platforms, specific dosage strengths, or geography-specific contracting advantages).
- Volume: typically remains resilient relative to revenue because demand persists.
Investor lens
- Upside after generic entry is usually operational, not commercial:
- scale and manufacturing yield,
- supply reliability and allocation management,
- formula/dose breadth,
- payer contracting and reimbursement fit.
Where does risperidone still generate premium economics?
Even in a generic-dominated market, premium economics can remain where the product mix includes protected or harder-to-substitute segments, and where formulation or delivery reduces adherence failures.
Residual value pools
- LAI formulations: typically have more complex procurement and can sustain steadier demand if administered through established channels.
- Segmented dosage strengths: some strengths or pack configurations may face slower substitution due to pharmacy stocking patterns and contracting.
How do major regulatory timelines affect market access economics?
Risperidone’s market access has been shaped by a timeline of patent expiry and marketing authorization across jurisdictions. The resulting generic market depth tends to increase over successive years as additional entrants clear regulatory requirements.
Commercial impact of these timelines
- Patent expiry triggers:
- accelerated generic entries,
- payer formulary updates,
- wholesale price normalization.
- Subsequent years tend to show:
- additional bidders expanding supply,
- further price pressure and margin erosion,
- consolidation around cost leaders.
What is the typical financial pattern after patent expiry?
A mature generic pattern for a product like risperidone usually looks like this:
- Year 0 to Year 1 post-expiry
- Revenue drop as substitution accelerates.
- Gross margin pressure from competitive pricing.
- Year 1 to Year 3
- Volume stabilizes but at lower net revenue per unit.
- Margin improves only for suppliers with the lowest landed costs and strongest contract coverage.
- Year 3 onward
- Market becomes a procurement-and-contracting game.
- Differentiation concentrates in:
- supply reliability,
- service levels,
- LAI availability where applicable.
How do financial results typically map to risperidone’s competitive set?
Risperidone is widely manufactured by multiple generic firms, which turns pricing into a function of capacity and cost structure. For branded holders, financial trajectory generally declines after exclusivity, and for generic manufacturers, profitability depends on:
- manufacturing cost per unit,
- ability to secure and retain payer and pharmacy contracts,
- distribution execution,
- inventory and allocation strategies.
Benchmarking approach
Use two metrics to interpret financial trajectory:
- Net sales vs. unit shipments: divergence indicates pricing stress.
- Gross margin vs. production utilization: divergence indicates capacity-driven cost efficiency and pricing power (often limited in mature segments).
What market segments matter most for revenue continuity?
For risperidone, revenue continuity usually depends on maintaining share in:
- long-term treatment populations,
- institutional channels (where LAI administration occurs),
- regions with slower generic price erosion due to contract design or distribution structure.
Key segment levers
- formulary preference,
- tender participation,
- pack-size and strength availability,
- LAI channel relationships.
What are the main risks to financial trajectory?
Even with persistent demand, the financial trajectory can deviate due to category dynamics.
Material risk categories
- Pricing resets: aggressive tender cycles can compress margins further.
- Competitive entry cadence: incremental generic launches deepen price pressure.
- Formulation or delivery disruptions: supply interruptions can cost share, especially where clinicians rely on continuity.
- Competitive substitution by other antipsychotics: payer preferencing and guideline shifts can reduce share even if total schizophrenia and bipolar treatment volumes remain stable.
What would an actionable financial readout look like for a generic entrant?
For firms considering investment or expansion in risperidone supply, the financial trajectory should be evaluated through operating economics rather than growth assumptions typical of branded drugs.
Actionable commercial metrics
- Contract coverage breadth (share of revenue tied to framework agreements)
- Unit-cost position (manufacturing plus freight plus chargebacks)
- Penetration by dosage strength and pack configuration
- Supply allocation and fill-rate reliability
Key Takeaways
- Risperidone’s commercial arc is mature and generic-led: revenue and margins have been structurally pressured by substitution, while volume demand persists due to chronic psychiatric indications.
- Financial trajectory is best interpreted as a pricing-and-contracting story post-patent expiry rather than growth in patient numbers.
- Residual value pools can persist in LAI-related channels and where procurement structures reduce rapid substitution.
- For investors and R&D planners, value creation concentrates in cost leadership, supply reliability, and payer contracting, not in new patient growth.
FAQs
-
Is risperidone revenue still supported after generic entry?
Yes, typically via persistent demand in chronic indications, but at lower net revenue per unit due to competitive pricing.
-
What drives profitability for generic risperidone makers?
Cost per unit, contract pricing, supply reliability, and dosage-strength mix that influences net realized pricing.
-
Does long-acting injectable risperidone change the economics?
It can, because LAI channels and procurement patterns can sustain steadier demand and can be harder to substitute than oral products.
-
What market event most affects risperidone’s financial trajectory?
Patent expiry and the resulting wave of generic launches, followed by payer contract resets.
-
What is the main threat to a mature risperidone business model?
Additional generic entrants and aggressive tender cycles that reset pricing and compress gross margins.
References
[1] FDA. Risperdal (risperidone) prescribing information. U.S. Food and Drug Administration. https://www.accessdata.fda.gov/
[2] EMA. Risperdal EPAR and product information (European Public Assessment Report). European Medicines Agency. https://www.ema.europa.eu/
[3] IQVIA Institute. Medicines use and spending / global pharmaceutical trends reports. IQVIA. https://www.iqvia.com/insights/the-iqvia-institute/reports