Last updated: April 23, 2026
Risperdal (risperidone) is a long-tenured, off-patent branded antipsychotic that has transitioned into a largely generic, volume-driven market. Across major geographies, financial trajectory is dominated by (1) patent and exclusivity expiry, (2) step-down in brand pricing and pull-through, and (3) durability of demand for the class, partially offset by originator-agnostic switching dynamics and payer controls. The branded business has progressively ceded share to low-cost generics, while the broader risperidone franchise has sustained volume through multiple branded and generic formulations over time.
How did Risperdal’s market shift from branded exclusivity to generic-led demand?
Timeline of key exclusivity and patent effects (high level)
Risperdal’s branded market position eroded as global exclusivity ended and generic competition consolidated. The U.S. and EU were the decisive arenas for early brand-to-generic transition, with downstream impacts in other OECD markets.
Generic competitive mechanics that shaped price and sales
- Entry of multi-source generics pushed wholesale price erosion and retailer/payer contracting on lowest acquisition cost.
- Switching incentives accelerated once payers implemented formulary preferences for AB-rated generic products (same active ingredient, typically same dosage form).
- Patient-level tolerability and prescriber inertia provided only partial brand insulation; the active ingredient name (risperidone) became the primary decision variable.
Persisting demand drivers despite commoditization
- Stable indication base: schizophrenia, bipolar disorder, and irritability associated with autistic disorder (indication-specific availability and labeling varies by jurisdiction).
- Dose flexibility: multiple strengths and (depending on market) long-acting injectable (LAI) offerings improved continuity of treatment for certain patient segments.
- Class durability: second-generation antipsychotics retained guideline relevance in multiple treatment pathways even as brand economics weakened.
What do financial statements and market disclosures show about the brand’s trajectory?
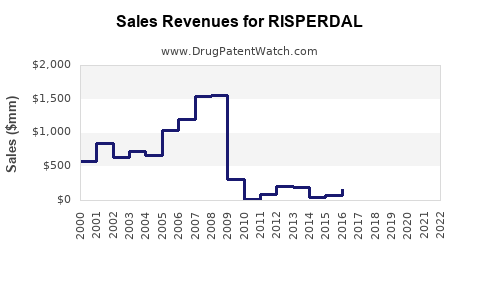
Brand sales are structurally exposed to generic substitution
Because risperidone’s active ingredient is off-patent, the financial pattern for the branded product typically follows:
- Peak during exclusivity (or near-exclusivity windows)
- Step-down during initial generic entries and tender-driven price reductions
- Continued compression as the market becomes low-cost and highly competitive
Company-level implication: brand P&L migration to residual premium plus mix
For originators, residual branded performance usually comes from:
- Remaining brand preference in select channels (where contracting is not yet fully generic-driven)
- Higher ASP in non-transparent channels or lower penetration of automatic substitution
- Ongoing contribution from differentiated presentations (where they exist and where generics are less directly substitutable)
Long-acting and formulation considerations
Risperdal contains oral risperidone and has had long-acting injectable variants historically marketed under the franchise. In many systems, LAIs can maintain brand leverage longer than oral solid doses because of administration infrastructure, adherence, and injection-specific switching barriers. That said, the core market financial trajectory still trends generic-led when the LAI is also exposed to competition.
How does pricing and reimbursement typically evolve for an off-patent antipsychotic like Risperdal?
U.S. reimbursement dynamics (payer control and reimbursement pressure)
In the U.S., the off-patent branded antipsychotic market commonly sees:
- Formulary placement shift: branded versions move from preferred to non-preferred tiers once generic equivalents are established.
- Patient cost-share increases for brand: commercial plans often push higher co-pays for branded drugs on generic availability.
- PBM contracting: lower acquisition costs of generics drive negotiated discounts and preferred status.
EU and other OECD dynamics (tendering and substitution)
In markets with strong generic substitution frameworks:
- Retail substitution reduces brand capture.
- Price regulation and reference pricing compress brand pricing more quickly.
- Hospital formularies and procurement tenders reduce branded usage where generics meet budget thresholds.
What is the financial trajectory expected to look like over the next cycle?
A mature antipsychotic franchise market typically demonstrates:
- Ongoing erosion of branded ASP toward generic parity.
- Share stabilization near low-single-digit branded remnants that persist only where prescribers or payers maintain preference (often tied to non-medical factors like dispensing patterns, channel relationships, or clinical continuity with a specific presentation).
- Volume survivability depends less on brand and more on class treatment prevalence.
Which market segments are most likely to keep volume for the risperidone franchise?
Clinical and channel segments with relative durability
- Chronic schizophrenia management
- Bipolar disorder maintenance
- LAI continuity cohorts (where injection-specific switching barriers apply)
Channel segments
- Institutional/hospital-administered settings can show slower substitution for certain branded presentations.
- Retail experiences faster generic share gains once substituted products become entrenched.
What patent and regulatory reality most affects future brand profitability?
Risperdal’s brand profitability is primarily affected by whether any remaining exclusivity protects specific presentations (formulations, manufacturing processes, or pediatric/conditional exclusivity equivalents depending on jurisdiction). Absent meaningful exclusivity renewal, the economics follow generic substitution.
Investment and R&D implications: what matters for next-stage strategy?
For originators holding residual branded rights
- Cost-to-serve pressure rises as share declines and marketing ROI deteriorates.
- Lifecycle management shifts to differentiated presentations or to defending specific patient segments via access programs rather than pricing power.
- Portfolio rebalancing becomes the norm: brand cash flow transitions into funding other assets.
For generic entrants and investors
- Volume is the business: pricing is the outcome, not the driver.
- Margin depends on manufacturing efficiency and tender access in key channels.
- LAI or specialized dosing forms can preserve differentiation longer than plain oral presentations if competition is slower.
Key Takeaways
- Risperdal’s market dynamics follow a classic post-exclusivity pattern: branded sales compress as generic substitution expands and payer formularies tighten.
- Financial trajectory is dominated by price erosion, AB-rated substitution behavior, and contracting power rather than clinical novelty.
- Remaining branded performance tends to persist only where channel access, formulation specificity, or LAI continuity slows substitution.
- Future profitability is mainly determined by whether any residual exclusivity protects specific presentations; otherwise the franchise stays generic-led and volume-driven.
FAQs
1) Is Risperdal still protected by patent exclusivity in major markets?
Risperdal is widely off-patent in major jurisdictions, and the branded product is exposed to generic substitution dynamics that typically follow exclusivity expiry.
2) What most drives branded sales decline for an antipsychotic like Risperdal?
Branded sales decline is driven by generic market entry, rapid payer formulary shifts, and price erosion tied to procurement and contracting.
3) Which formulation typically preserves brand use longer: oral or long-acting injectables?
Long-acting injectables often experience slower switching than oral tablets due to administration workflow and continuity effects, though this depends on the competitive maturity of each presentation.
4) Does the risperidone class still grow even when the brand loses share?
The class can retain or grow demand with population and disease prevalence, but branded share usually declines as generic penetration increases.
5) What is the most important metric for tracking Risperdal’s financial trajectory?
Generic-led market share and branded net price (after rebates/discounts) are the primary indicators; volume matters, but pricing compression typically dominates branded profitability.
References
[1] U.S. Food and Drug Administration. Risperdal (risperidone) label and prescribing information. FDA. https://www.accessdata.fda.gov/
[2] European Medicines Agency. Risperdal EPAR (product information and EPAR documents). EMA. https://www.ema.europa.eu/
[3] U.S. Patent and Trademark Office (USPTO). Patent records for risperidone and Risperdal-related inventions (bibliographic and legal status records). https://patentscope.wipo.int/ and https://www.uspto.gov/
[4] IQVIA Institute for Human Data Science. Medicines use and spending reports (generic penetration and market trends). IQVIA. https://www.iqvia.com/insights/the-iqvia-institute/reports