Last updated: February 19, 2026
This analysis examines the market dynamics and financial trajectory of RENVELA (sevelamer carbonate), a phosphate binder used in the management of hyperphosphatemia in adult patients with chronic kidney disease (CKD) on hemodialysis or on peritoneal dialysis. Key patent expiries, market competition, and revenue trends are assessed to inform R&D and investment decisions.
What is RENVELA's Market Position?
RENVELA is a branded pharmaceutical product approved by the U.S. Food and Drug Administration (FDA). It is manufactured by Sanofi S.A. (formerly Genzyme). The drug's primary indication is for the control of serum phosphorus in adult patients with CKD on dialysis.
The market for phosphate binders is characterized by patient adherence, cost-effectiveness, and the availability of generic alternatives. RENVELA, as a branded drug, competes with other phosphate binders, including both non-calcium-based binders and calcium-based binders.
Competitive Landscape
The competitive landscape for RENVELA includes:
- Other Non-Calcium Phosphate Binders:
- Fosrenol (lanthanum carbonate): Another non-calcium binder with a similar mechanism of action.
- Velphoro (sucroferric oxyhydroxide): A ferric iron-based phosphate binder.
- Calcium-Based Phosphate Binders:
- Calcium acetate (various brands and generics): A widely used, cost-effective option.
- Calcium carbonate (various brands and generics): Another common calcium-based binder.
- Newer Agents: While less direct competitors for RENVELA's established market, newer oral and intravenous phosphate binders are in development and have entered the market, potentially impacting future market share.
The introduction of generic versions of sevelamer carbonate has significantly altered RENVELA's market dynamics. Generic competition typically leads to price erosion and a shift in market share towards lower-cost alternatives.
U.S. FDA Approval and Exclusivity
RENVELA (sevelamer carbonate) received its initial U.S. FDA approval on May 12, 2009, for the control of serum phosphorus in adult patients with CKD on dialysis [1]. Later approvals extended its use to patients on peritoneal dialysis.
- Original NDA Number: 021776
- Dosage Forms: Tablets and powder for oral suspension.
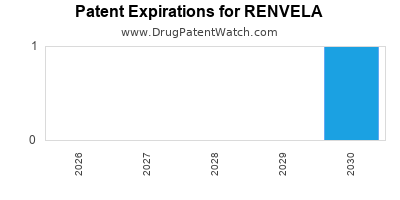
The patent landscape for RENVELA is crucial for understanding its market exclusivity. Key patents that protected the original composition of matter and methods of use for sevelamer hydrochloride (the precursor to sevelamer carbonate) have expired. Sevelamer carbonate, a distinct salt form, also had its own patent protection.
- U.S. Patent 5,958,907: This patent, covering sevelamer hydrochloride, expired in 2018 [2].
- U.S. Patent 7,781,516: This patent, related to sevelamer carbonate and its use, was a significant protection for the branded product. It expired in 2021 [3].
The expiry of these primary patents opened the door for generic manufacturers to enter the U.S. market.
Generic Entry and Market Impact
Generic versions of sevelamer carbonate have become available in the U.S. following the expiry of key patents. This has led to a decrease in the market share held by branded RENVELA and a significant reduction in its average selling price (ASP).
- First Generic Approvals: Generic sevelamer carbonate approvals began to appear around the time of patent expiry. For example, Teva Pharmaceuticals received tentative approval for its generic sevelamer carbonate tablets in 2019, with full approval following patent challenges [4]. Other generic manufacturers, including Apotex and Mylan, also have generic versions on the market.
- Price Erosion: Generic competition typically results in price reductions of 50% to 90% compared to the branded product's peak pricing. This directly impacts the revenue generated by RENVELA.
The availability of multiple generic sevelamer carbonate products intensifies competition, further driving down prices and influencing prescribing patterns. Healthcare providers and payers often favor generic options due to their lower cost.
What is RENVELA's Financial Trajectory?
RENVELA's financial trajectory has been significantly impacted by the expiry of its patent protection and the subsequent entry of generic competition. Prior to patent expiry, the drug generated substantial revenue as a leading branded phosphate binder. Post-expiry, revenue has declined due to price erosion and market share loss.
Revenue Trends
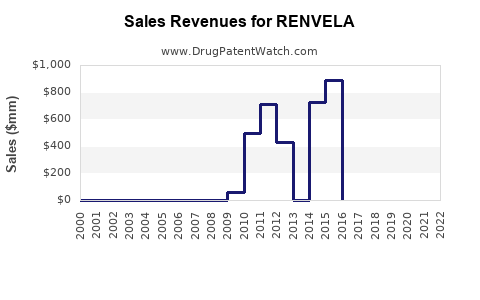
- Pre-Patent Expiry: During its period of market exclusivity, RENVELA achieved significant sales. Specific historical sales figures for RENVELA as a standalone product are often consolidated within Sanofi's broader renal or specialty care portfolios. However, industry reports indicated strong performance. For instance, in 2014, Genzyme (then part of Sanofi) reported that sevelamer products (including RENVELA and the older product RENAGEL) had combined net sales of approximately $1.3 billion globally [5]. RENVELA contributed a significant portion of this.
- Post-Patent Expiry (U.S. Market):
- 2019: Sales of branded RENVELA began to feel the initial pressure of generic anticipation.
- 2021-2022: Following the expiry of the U.S. Patent 7,781,516 in 2021, a marked decline in RENVELA's revenue is expected and has been observed in market data. While exact figures for branded RENVELA post-generic entry are proprietary, typical patterns show a steep decline.
- Current Market: The U.S. market for sevelamer carbonate is now heavily dominated by generics. Branded RENVELA's market share is minimal, primarily serving niche markets or specific formularies where brand loyalty or contractual agreements may persist.
Impact of Generic Competition on Pricing
The introduction of generics directly affects RENVELA's pricing power.
- Average Selling Price (ASP): The ASP for branded RENVELA has decreased significantly from its peak. The ASP is the average price at which a drug is sold, net of rebates and discounts. Generic entry typically drives down the ASP for both branded and generic versions of the same drug.
- Reimbursement Landscape: Insurers and pharmacy benefit managers (PBMs) favor generic drugs due to their lower cost. This leads to higher co-pays for branded drugs or preferred placement of generics on formularies. This pricing pressure directly translates to lower net revenue for the branded product.
Global Market Considerations
While the U.S. market is a primary focus, RENVELA's financial trajectory is also influenced by its performance in other key markets. Patent expiries and generic introductions vary by country.
- Europe: Patent expiries and generic availability in European countries also contributed to global revenue declines for branded RENVELA.
- Other Regions: Sanofi's global strategy for RENVELA involved managing its lifecycle across different regulatory and market environments.
Manufacturing and Supply Chain
Sanofi maintains manufacturing capabilities for RENVELA. The transition to a genericized market requires efficient supply chain management to compete on volume and cost.
- Cost of Goods Sold (COGS): As a branded drug faces generic competition, maintaining profitability requires optimizing COGS. Sanofi's manufacturing costs for RENVELA would be compared against the lower price points set by generic competitors.
- Supply Chain Efficiency: A robust and cost-effective supply chain is essential for branded products to remain competitive in a post-expiry environment, even with reduced market share.
What are the Key Patent Expiries and Their Implications?
The patent landscape for RENVELA is fundamental to understanding its market exclusivity and subsequent genericization. The expiry of these patents has directly led to the loss of market exclusivity and the erosion of its financial performance as a branded product.
Primary Patent Expiries
The core patents protecting sevelamer, the active pharmaceutical ingredient (API) in RENVELA, have expired.
- U.S. Patent 5,958,907 (Sevelamer Hydrochloride): This foundational patent expired in 2018. While it covered the hydrochloride salt, it represented an early stage of patent protection for the sevelamer molecule.
- U.S. Patent 7,781,516 (Sevelamer Carbonate): This patent was critical for RENVELA, protecting the carbonate salt form and its specific uses. It expired in 2021. This expiration marked the definitive end of primary patent protection for RENVELA in the U.S. [3].
Secondary Patents and Litigation
Pharmaceutical companies often pursue secondary patents (e.g., for manufacturing processes, formulations, or new uses) to extend market exclusivity. Sanofi would have likely explored such avenues for RENVELA.
- Patent Litigation: The period leading up to and following patent expiry is often characterized by patent litigation. Generic companies challenge the validity or infringement of remaining patents, while the brand manufacturer defends its intellectual property. These legal battles can influence the timing of generic entry. For sevelamer carbonate, litigation focused on challenging the validity and enforceability of key patents, such as the '516 patent, to secure an earlier generic launch.
- Orphan Drug Exclusivity: RENVELA is not designated as an orphan drug, so it did not benefit from the 7-year exclusivity period granted to drugs treating rare diseases.
Implications of Patent Expiry for Market Entry
The expiry of key patents triggers a cascade of market events:
- Generic Launch Readiness: Generic manufacturers actively prepare for patent expiry, conducting bioequivalence studies and filing Abbreviated New Drug Applications (ANDAs) with the FDA.
- Market Entry: Upon patent expiry (or earlier if a patent challenge is successful), generic versions of sevelamer carbonate become available.
- Price Competition: Multiple generic manufacturers entering the market leads to intense price competition.
- Market Share Shift: Prescribers and payers switch to lower-cost generic alternatives, leading to a rapid decline in the branded product's market share.
- Revenue Decline: The combined effect of price reduction and market share loss results in a significant decrease in revenue for the branded product.
The timeline of patent expiry directly correlates with RENVELA's shift from a high-revenue branded product to a highly commoditized generic market.
What are the Future Market Projections and Opportunities?
The future market for RENVELA, as a branded product, is characterized by limited growth potential and a continued dominance of generic alternatives. However, opportunities may exist in specific market segments or through lifecycle management strategies, albeit with reduced impact.
Market Outlook for Branded RENVELA
- Continued Generic Dominance: The U.S. market for sevelamer carbonate is expected to remain dominated by generic products. Branded RENVELA's market share will likely be marginal, serving only a fraction of the patient population.
- Price Pressure: Ongoing price pressure from generic competition will continue to suppress any potential for significant revenue growth for the branded product.
- Limited Brand Loyalty: In the phosphate binder market, efficacy, safety, and cost are primary drivers. Brand loyalty for RENVELA is unlikely to counteract the economic advantages of generics.
Opportunities in the Generic Sevelamer Market
While the focus is on branded RENVELA, the broader market for sevelamer carbonate (including generics) presents ongoing activity.
- Generic Manufacturing: Opportunities exist for generic pharmaceutical companies to manufacture and market sevelamer carbonate. Success in this space depends on efficient manufacturing, strong distribution networks, and competitive pricing.
- Formulation Development: While RENVELA's primary formulations (tablets and powder) are well-established, there may be limited opportunities for novel formulations or delivery systems that offer a clinical advantage, though the barrier to entry for such innovation in a genericized market is high.
- Geographic Expansion: For generic manufacturers, expanding access to sevelamer carbonate in emerging markets where branded options may still hold sway or where access is limited presents an opportunity.
Lifecycle Management Strategies (Limited for Branded RENVELA)
For branded RENVELA, traditional lifecycle management strategies are largely exhausted due to patent expiry.
- Combination Therapies: Exploring fixed-dose combinations with other CKD management drugs is a common strategy, but unlikely to be pursued for a drug nearing the end of its commercial lifecycle as a brand.
- New Indications: Seeking new indications for sevelamer is a possibility, but the clinical development and regulatory hurdles for a well-established, off-patent drug make this a less probable or cost-effective strategy for the branded product.
Investment Considerations
For investors, the focus shifts from the branded RENVELA product to companies involved in the generic sevelamer carbonate market.
- Generic Manufacturers: Companies with robust ANDA pipelines and efficient manufacturing for sevelamer carbonate are potential investment targets.
- API Suppliers: Suppliers of sevelamer API who can provide high-quality, cost-effective material to generic drug manufacturers will also be critical players.
- Market Analysis: Investors should monitor the pricing trends, competitive intensity, and regulatory landscape within the broader phosphate binder market.
The future for branded RENVELA is one of diminishing revenue. The financial opportunity lies with the generic producers and suppliers who will capitalize on the demand for this essential medication at a lower cost.
Key Takeaways
- RENVELA's patent protection, specifically U.S. Patent 7,781,516, expired in 2021, marking the end of its primary market exclusivity.
- The U.S. market for sevelamer carbonate is now dominated by generic versions, leading to significant price erosion and a sharp decline in branded RENVELA's market share and revenue.
- RENVELA achieved substantial revenue during its exclusivity period, with combined sevelamer product sales reaching approximately $1.3 billion globally in 2014.
- Generic entry has led to competitive pricing and a shift in prescribing towards lower-cost alternatives.
- Future opportunities are primarily in the generic sevelamer carbonate market, with potential for generic manufacturers and API suppliers, rather than in the branded RENVELA product.
FAQs
-
When did the key patent for RENVELA expire in the U.S.?
The primary U.S. patent protecting sevelamer carbonate, U.S. Patent 7,781,516, expired in 2021.
-
What is the primary impact of patent expiry on RENVELA's sales?
Patent expiry has led to the introduction of generic sevelamer carbonate, causing significant price erosion and a substantial decrease in branded RENVELA's market share and revenue.
-
Which companies are currently marketing generic versions of sevelamer carbonate?
Several pharmaceutical companies, including Teva Pharmaceuticals, Apotex, and Mylan (now Viatris), have launched generic versions of sevelamer carbonate in the U.S.
-
Does RENVELA have any remaining market exclusivity in major markets?
In the U.S. and most major European markets, RENVELA no longer has patent-driven market exclusivity. Its market presence is now that of a branded product competing against generics.
-
Are there any new indications being pursued for branded RENVELA?
Given the patent expiry and the commoditized nature of the market, significant investment in pursuing new indications for branded RENVELA is unlikely. The focus has shifted to the generic market.
Citations
[1] U.S. Food & Drug Administration. (2009, May 12). Drug Approval Package: Renvela (sevelamer carbonate) Tablets. [Press release].
[2] U.S. Patent and Trademark Office. (1999). Patent No. US 5,958,907. Retrieved from USPTO database.
[3] U.S. Patent and Trademark Office. (2010). Patent No. US 7,781,516. Retrieved from USPTO database.
[4] U.S. Food & Drug Administration. (2019, October 28). FDA Tentative Approval List. [Internal document, may not be publicly accessible in its original form].
[5] Sanofi. (2015). Sanofi Annual Report 2014. Retrieved from Sanofi Investor Relations website.