Last updated: February 19, 2026
Prochlorperazine, an established dopamine antagonist, primarily used as an antiemetic and antipsychotic, operates within a mature pharmaceutical market characterized by generic competition and stable, albeit modest, growth. Its market performance is primarily driven by its cost-effectiveness and continued utility in managing nausea, vomiting, and certain psychiatric conditions. However, the absence of novel patent protections and the presence of numerous generic manufacturers limit significant revenue expansion.
What are the primary therapeutic indications for Prochlorperazine?
Prochlorperazine is indicated for the management of a range of conditions, leveraging its dopamine receptor blocking activity. Its established efficacy in these areas underpins its sustained market presence.
- Nausea and Vomiting: This is the drug's most prevalent use. It is effective in treating nausea and vomiting associated with various causes, including post-operative recovery, chemotherapy, and migraines. The American Society of Health-System Pharmacists (ASHP) lists prochlorperazine as a first-line option for managing chemotherapy-induced nausea and vomiting (CINV) [1].
- Psychiatric Disorders: Prochlorperazine is also used in the treatment of symptoms associated with psychotic disorders, such as schizophrenia. It can help reduce agitation, hallucinations, and delusions. The National Alliance on Mental Illness (NAMI) recognizes older antipsychotics like prochlorperazine as options for managing psychosis, particularly when cost is a significant factor [2].
- Migraine Headaches: In some cases, prochlorperazine is administered to manage acute migraine attacks, often intravenously, due to its antiemetic and potential sedative effects that can alleviate migraine symptoms.
Who are the key manufacturers and what is the competitive landscape?
The prochlorperazine market is highly fragmented, dominated by generic drug manufacturers. The absence of proprietary formulations or recent patent expirations for new indications means that market share is largely determined by production volume, distribution networks, and pricing strategies.
- Major Generic Manufacturers:
- Teva Pharmaceutical Industries
- Sun Pharmaceutical Industries
- Mylan (now Viatris)
- Dr. Reddy's Laboratories
- Lupin Pharmaceuticals
These companies produce prochlorperazine in various dosage forms, including oral tablets, suppositories, and injectable solutions. The competition is intense, leading to price erosion and a focus on cost-efficient manufacturing.
- Product Forms and Availability:
- Oral Tablets: Commonly available in 5 mg and 10 mg strengths.
- Suppositories: Available in 25 mg strength, offering an alternative route of administration.
- Injectable Solutions: Typically 5 mg/mL, used for rapid symptom control in hospital and clinical settings.
The competitive landscape does not feature significant players focused on novel drug delivery systems or advanced formulations for prochlorperazine. The market is mature, with most activity centered on ensuring consistent supply of existing generic products.
What is the historical and projected financial trajectory of Prochlorperazine?
The financial trajectory of prochlorperazine is characterized by steady, low-single-digit revenue growth, primarily driven by volume rather than price increases. Its status as an off-patent drug means that revenue is largely tied to market demand for its established indications and its affordability compared to newer agents.
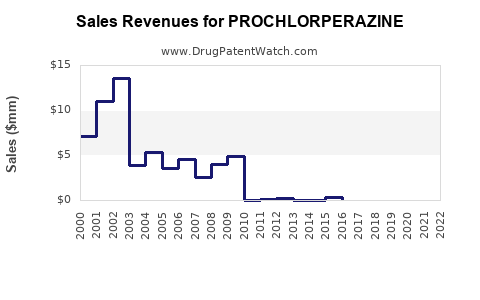
- Historical Revenue Trends: Global sales for prochlorperazine have remained relatively stable over the past decade. Estimates suggest that the global market for antiemetics, a category prochlorperazine significantly contributes to, was valued at approximately $3.5 billion in 2022, with prochlorperazine's share being a fraction of this due to its generic nature and niche applications within the broader antiemetic market [3]. Prochlorperazine specifically, as a standalone generic product, likely generates annual revenues in the tens to low hundreds of millions globally, depending on the specific regions and manufacturers' market penetration.
- Projected Market Growth: The market for prochlorperazine is projected to grow at a Compound Annual Growth Rate (CAGR) of 1.5% to 2.5% over the next five years. This modest growth is attributed to:
- Aging Population: An increasing elderly population often experiences conditions like nausea and vomiting, driving demand.
- Prevalence of Migraines and CINV: Continued prevalence of migraines and the need for effective CINV management in cancer patients sustain demand.
- Cost-Effectiveness: Its low cost makes it a preferred option in healthcare systems with budget constraints, particularly in emerging markets.
- Factors Limiting Growth:
- Generic Competition: The high degree of generic competition prevents significant price increases.
- Emergence of Newer Therapies: For indications like CINV and psychiatric disorders, newer, more targeted, and potentially more effective therapies with fewer side effects are available, though often at a higher cost. Examples include 5-HT3 receptor antagonists (e.g., ondansetron, palonosetron) for nausea and newer antipsychotics.
- Side Effect Profile: Prochlorperazine carries a risk of extrapyramidal side effects, which can limit its use in certain patient populations or lead to preferences for alternatives with better safety profiles.
The financial outlook for prochlorperazine is one of sustained, albeit slow, revenue generation, primarily from established markets and its role as a cost-effective therapeutic option.
What is the patent landscape for Prochlorperazine?
Prochlorperazine was first patented in the mid-1950s by Smith, Kline & French Laboratories (now part of GlaxoSmithKline). Its original composition of matter patents have long since expired. The current patent landscape is characterized by the absence of broad, composition-of-matter patents covering the active pharmaceutical ingredient (API).
-
Original Patent Expiration: The fundamental patents for prochlorperazine have expired, allowing for widespread generic manufacturing.
-
Potential for Secondary Patents: While composition-of-matter patents are expired, manufacturers may hold patents related to specific:
- Manufacturing Processes: Novel or improved methods for synthesizing the API.
- Formulations: Specific delivery systems, combinations with other drugs, or extended-release formulations, although significant innovation in this area for prochlorperazine is not widely documented.
- New Medical Uses: While less common for older drugs, patents can be obtained for novel therapeutic applications of existing compounds, provided they meet criteria for novelty, inventiveness, and utility. However, no significant new indications for prochlorperazine have emerged recently that are under strong patent protection.
-
Generic Market Dominance: The lack of active, broad patents means that the market is primarily a generic one. Intellectual property challenges are unlikely to significantly alter the competitive landscape or revenue potential for the API itself. Any potential for market differentiation would stem from manufacturing efficiency, supply chain reliability, or niche formulation development.
What are the regulatory considerations and market access challenges?
Regulatory considerations and market access for prochlorperazine are influenced by its long history and classification as a generic drug. The primary hurdles are related to manufacturing standards, pharmacovigilance, and established clinical guidelines.
- Regulatory Approvals: Prochlorperazine is approved by major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Generic drug approval requires demonstrating bioequivalence to a reference listed drug.
- Manufacturing Standards: Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy. Inspections by regulatory authorities are routine.
- Pharmacovigilance: As with all pharmaceuticals, ongoing pharmacovigilance is required to monitor for adverse events and update labeling as necessary. The known side effect profile, particularly extrapyramidal symptoms, necessitates careful patient selection and monitoring.
- Market Access and Reimbursement:
- Pricing Pressures: Due to its generic status, prochlorperazine faces significant pricing pressures from payers and healthcare systems aiming to control costs.
- Formulary Placement: It is generally included on hospital and insurance formularies due to its low cost and established efficacy. However, competition from newer antiemetics or antipsychotics, which may offer improved efficacy or safety profiles for specific patient subsets, can influence formulary decisions.
- Clinical Guidelines: Inclusion in treatment guidelines for nausea, vomiting, and psychosis supports its continued use. However, guidelines also often recommend newer agents as first-line options for certain conditions, potentially impacting prochlorperazine's market share in those specific areas.
Market access is largely assured due to its therapeutic utility and cost-effectiveness, but growth is constrained by the availability of potentially superior or specialized alternatives and intense price competition.
What are the future outlook and potential market shifts?
The future outlook for prochlorperazine is one of continued stability, with limited potential for significant growth. The market is unlikely to experience disruptive shifts unless unforeseen regulatory changes occur or a novel, patentable formulation or indication is developed, which is improbable given its age.
- Sustained Demand: Demand will continue to be driven by its established roles in managing nausea, vomiting, and certain psychiatric symptoms, particularly in settings where cost is a primary consideration.
- Generic Market Dynamics: The market will remain dominated by generic competition, with pricing being a key differentiator for manufacturers. Efficiency in production and supply chain management will be critical for maintaining profitability.
- Competition from Newer Agents: The trend of newer, more targeted therapies with potentially better efficacy and safety profiles will continue to exert competitive pressure. This is particularly true in oncology supportive care (CINV) and in the management of complex psychiatric conditions, where personalized medicine approaches are gaining traction.
- Limited Innovation: Significant R&D investment in novel formulations or delivery systems for prochlorperazine is unlikely, given the mature nature of the drug and the high cost of bringing new formulations to market in a competitive generic space.
- Geographic Variations: Growth may be slightly higher in emerging markets where cost-effectiveness is a paramount concern and access to newer, more expensive therapies is limited.
The prochlorperazine market is expected to remain a steady, albeit modest, revenue generator for its manufacturers, characterized by high volume, low margins, and the absence of significant innovation or patent-driven growth.
Key Takeaways
- Prochlorperazine is a mature, off-patent drug with established therapeutic indications in antiemesis and antipsychotic therapy.
- The market is highly competitive, dominated by generic manufacturers, leading to price erosion and stable, low-single-digit revenue growth.
- Key drivers include its cost-effectiveness, use in managing nausea/vomiting, and presence in treatment guidelines.
- Growth is constrained by intense generic competition and the availability of newer, more targeted therapeutic alternatives.
- The patent landscape is clear of broad composition-of-matter patents, indicating a stable generic market.
- Regulatory considerations focus on cGMP compliance and pharmacovigilance, with market access driven by cost-effectiveness and formulary inclusion.
- The future outlook suggests continued stability, with limited potential for significant expansion, primarily driven by volume and cost competitiveness rather than innovation.
Frequently Asked Questions
-
What is the approximate global market size for prochlorperazine specifically?
The global market size for prochlorperazine as a standalone generic product is estimated to be in the tens to low hundreds of millions of U.S. dollars annually. This figure is a subset of the broader antiemetics market.
-
Are there any new therapeutic indications for prochlorperazine currently under development or patent protection?
There are no significant new therapeutic indications for prochlorperazine currently under active development or protected by substantial patent claims. Its established uses remain its primary market drivers.
-
How does prochlorperazine compare in terms of efficacy and safety to newer antiemetic drugs like ondansetron?
While prochlorperazine is effective for general nausea and vomiting, newer agents like ondansetron (a 5-HT3 receptor antagonist) are often preferred for chemotherapy-induced nausea and vomiting (CINV) due to a generally more favorable efficacy profile and potentially fewer extrapyramidal side effects. However, prochlorperazine's cost-effectiveness can still make it a viable option in certain scenarios.
-
What is the primary risk factor associated with the use of prochlorperazine?
The primary risk factor associated with prochlorperazine use is the potential for extrapyramidal side effects, including dystonia, akathisia, parkinsonism, and tardive dyskinesia, especially with long-term use or higher doses.
-
Will prochlorperazine likely be discontinued in favor of newer drugs in the foreseeable future?
It is unlikely that prochlorperazine will be broadly discontinued. Its established efficacy, low cost, and long history of use ensure its continued place in the therapeutic armamentarium, particularly in cost-sensitive healthcare environments and for its primary indications.
Citations
[1] American Society of Health-System Pharmacists. (2023). Prochlorperazine. ASHP Midyear Clinical Meeting & Exhibition.
[2] National Alliance on Mental Illness. (2023). Antipsychotic Medications. Retrieved from www.nami.org
[3] Grand View Research. (2023). Antiemetics Market Size, Share & Trends Analysis Report By Drug Class, By Application, By End-use, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from www.grandviewresearch.com