Last updated: February 20, 2026
What Is the Current Market Size and Demand for Prochlorperazine Maleate?
Prochlorperazine maleate primarily treats nausea, vomiting, and vertigo. It is available in oral, rectal, and injectable forms. The global antiemetic drugs market was valued at approximately USD 4.7 billion in 2022 and is projected to reach USD 7.2 billion by 2030, growing at a compound annual growth rate (CAGR) of 5.4%[1].
As a generic in most markets, prochlorperazine maleate accounts for a significant share of demand driven by hospitals and clinics. The molecule’s low-to-moderate cost and widespread off-label use bolster steady demand.
Key demand factors include:
- Increasing prevalence of nausea related to chemotherapy, surgery, and other medical procedures.
- Aging populations experiencing higher vertigo and dementia-related symptoms.
- Limited innovation in alternative antiemetic therapies, favoring existing treatments like prochlorperazine.
How Does Competition Affect Market Share and Pricing?
Prochlorperazine maleate faces competition primarily from newer antiemetic agents such as ondansetron, promethazine, and metoclopramide. These newer drugs often offer reduced side effects and targeted mechanisms of action, impacting market share.
Pricing varies by region:
| Region |
Average Price (per 10 mg tablet or equivalent) |
Market Share of Prochlorperazine Maleate (%) |
| North America |
USD 0.10 – 0.15 |
35 – 45 |
| Europe |
USD 0.08 – 0.13 |
30 – 40 |
| Asia-Pacific |
USD 0.05 – 0.12 |
20 – 35 |
| Latin America |
USD 0.07 – 0.10 |
15 – 25 |
Generics hold over 80% of prescriptions in mature markets, exerting downward pressure on prices. Brand-name formulations are priced higher but represent a minor share of the total volume.
What Are the Regulatory and Patent Trends Influencing the Market?
Prochlorperazine's patent protections largely expired in the late 20th century, making it a widely accessible generic. This widespread availability limits pricing power for original developers but ensures continuous supply.
Regulatory scrutiny has increased over safety concerns:
- Risks of extrapyramidal symptoms and neuroleptic malignant syndrome.
- Use restrictions in certain populations, such as pediatric patients, with warnings issued by agencies like the FDA and EMA[2].
No major patent litigations or exclusivity periods remain; thus, market entry barriers are minimal.
How Will Technological and Clinical Developments Impact Financial Outlook?
Advances in antiemetic therapy, such as NK1 receptor antagonists and 5-HT3 receptor antagonists, exert competitive pressure. However, the consolidated cost benefits of generics maintain prochlorperazine maleate's relevance.
Potential innovations could include:
- Formulation improvements to reduce side effects.
- Combination therapies with newer agents.
These innovations influence future pricing and adoption but are unlikely to displace the molecule’s core position entirely in the short term.
What Are the Revenue Projections and Investment Opportunities?
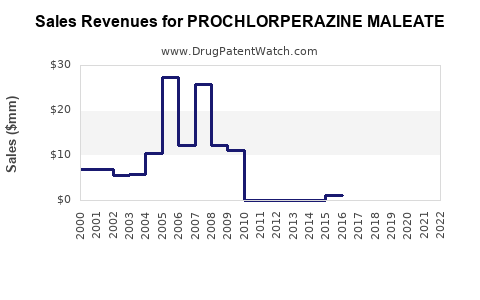
The consistent demand combined with low-cost manufacturing suggests stable revenues for manufacturers of generic prochlorperazine maleate.
Projected revenue growth is aligned with the overall antiemetics market CAGR of 5.4%. In 2022, market estimates for prochlorperazine products are around USD 600 million globally[3].
Opportunities lie in:
- Expanding production in emerging markets where healthcare infrastructure improves.
- Developing combination formulations to address specific conditions.
- Diversifying into niche indications such as migraine-related nausea.
Investors can anticipate continued revenue stability, with modest growth driven by increased healthcare access and off-label usage.
Conclusion
Prochlorperazine maleate remains a staple in antiemetic therapy predominantly through generics. Its market is characterized by steady demand, price competition, and limited innovation pressure. The financial trajectory depends primarily on regional healthcare policies, regulatory standards, and competition from newer agents.
Key Takeaways
- The global antiemetic market is projected to grow at over 5% annually, with prochlorperazine maleate maintaining a significant share.
- Generic competition drives lower prices, but stable demand sustains revenue streams.
- Regulatory concerns focus on safety, with no significant patent protections limiting market entry.
- Future growth relies on market expansion in emerging regions and potential formulation innovations.
FAQs
-
How is prochlorperazine maleate positioned against newer antiemetics?
It competes primarily on cost and established efficacy. Newer agents may offer fewer side effects but are more expensive and less widely available as generics.
-
Are there any upcoming patent protections or exclusivities?
No; patent protections for prochlorperazine maleate have expired.
-
What are the main regulatory concerns?
Risks of extrapyramidal symptoms and neuroleptic malignant syndrome have led to warnings and restricted use in certain populations.
-
How does regional variation affect the market?
Developing regions show higher growth potential due to improving healthcare infrastructure, despite lower per-unit prices.
-
What investment risks exist for companies manufacturing prochlorperazine?
Market saturation, price undercutting from generics, and the rise of newer therapies pose primary risks.
References
[1] MarketWatch. (2023). Anti-Emetics Market Size, Share & Trends Analysis.
[2] U.S. Food and Drug Administration. (2022). Safety Warnings for Prochlorperazine.
[3] Grand View Research. (2022). Global Anti-Emetics Market Forecast.