Last updated: February 19, 2026
NAMENDA (memantine hydrochloride) is a moderately strong voltage-dependent N-methyl-D-aspartate (NMDA) receptor antagonist. It is indicated for the treatment of moderate to severe dementia of the Alzheimer's type. The drug functions by modulating the effects of glutamate, a neurotransmitter implicated in learning and memory, which is thought to contribute to the neurotoxicity associated with Alzheimer's disease.
What is the current market position of NAMENDA?
NAMENDA's market position is characterized by its established presence in the Alzheimer's disease treatment landscape, facing both generic competition and evolving therapeutic approaches. As of late 2023, the branded NAMENDA (extended-release and immediate-release formulations) is no longer the sole option for patients, with the expiration of key patents leading to the introduction of generic alternatives. This shift significantly impacts the revenue generation for the originator product.
The Alzheimer's disease market is segmented by disease severity (mild, moderate, severe) and the specific therapeutic mechanisms targeted. NAMENDA primarily addresses moderate to severe stages. While effective for symptomatic relief, it does not halt or reverse disease progression. This limitation, coupled with the development of new disease-modifying therapies targeting amyloid and tau pathology, influences its long-term market share.
Market access for NAMENDA is generally broad, with reimbursement established across major healthcare systems and insurance providers. However, the availability of generics has led to price erosion, making cost a more significant factor in prescribing decisions. The market also sees competition from other symptomatic treatments and, increasingly, from agents with different mechanisms of action.
Key Market Segments and Dynamics:
- Disease Stage: Moderate to Severe Alzheimer's Disease.
- Competition: Branded NAMENDA formulations, generic memantine hydrochloride, other symptomatic treatments (e.g., cholinesterase inhibitors), and emerging disease-modifying therapies (e.g., aducanumab, lecanemab).
- Pricing: Branded product facing significant downward pressure due to generic entry. Generic memantine offers a lower-cost alternative.
- Reimbursement: Generally favorable, but payer policies may shift towards generics or preferred branded products based on cost-effectiveness.
- Geographic Reach: Global market presence, with varying adoption rates influenced by healthcare infrastructure and regulatory approvals.
What is NAMENDA's patent and exclusivity landscape?
NAMENDA's patent and exclusivity landscape has been a critical factor in its commercial lifecycle. The primary patents protecting the original formulation and use of memantine have expired, paving the way for generic competition.
The original New Drug Application (NDA) for NAMENDA was approved by the U.S. Food and Drug Administration (FDA) in October 2003 for the immediate-release formulation. The extended-release (XR) formulation received FDA approval in August 2012. Key patents related to these formulations and their methods of use have since expired.
For instance, U.S. Patent No. 5,374,637, covering memantine hydrochloride, expired in 2010. Other method-of-use patents and formulation patents have also lapsed or are nearing expiration.
Key Patent and Exclusivity Milestones (United States):
- NDA Approval (Immediate-Release): October 2003
- NDA Approval (Extended-Release): August 2012
- Expiration of Core Process Patents: Significant patents expired around 2010-2015.
- Generic Entry (Immediate-Release): Commenced following patent expirations, with multiple manufacturers launching generic memantine hydrochloride.
- Generic Entry (Extended-Release): Commenced following the expiration of relevant patents and exclusivity for the XR formulation.
The loss of market exclusivity for both the immediate-release and extended-release formulations is the primary driver behind the decline in branded NAMENDA sales. This is a standard trajectory for pharmaceutical products following patent expiry.
What has been NAMENDA's financial trajectory and sales performance?
NAMENDA's financial trajectory reflects a typical market lifecycle, characterized by growth during its period of exclusivity followed by a sharp decline post-generic entry.
Peak Sales and Decline:
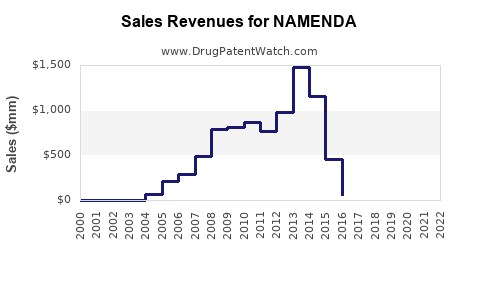
Prior to the widespread availability of generic memantine, branded NAMENDA achieved significant sales. In its peak years, global sales for NAMENDA (including both IR and XR formulations) generated over $1.4 billion annually for its marketer, Forest Laboratories (later acquired by Allergan, and subsequently by AbbVie).
For example, in fiscal year 2013, Forest Laboratories reported net sales of approximately $1.43 billion for NAMENDA in the U.S. [1]. By fiscal year 2015, following the launch of NAMENDA XR and continued market penetration, sales remained robust, with U.S. sales of NAMENDA and NAMENDA XR reaching $1.39 billion [2].
Following the FDA approval of generic memantine products in the U.S. in 2017, the financial performance of branded NAMENDA experienced a dramatic decline. This decline is directly attributable to the price competition introduced by lower-cost generic alternatives.
Recent Financial Performance (Post-Generic Entry):
As of recent reporting periods (late 2023/early 2024), the sales of branded NAMENDA have significantly diminished. The market has largely transitioned to generic memantine. For instance, AbbVie's financial reports indicate that NAMENDA and NAMENDA XR contributed minimal revenue in recent quarters, reflecting their status as largely genericized products.
- Example: In the first quarter of 2023, U.S. sales for NAMENDA and NAMENDA XR were reported to be in the range of tens of millions of dollars, a stark contrast to their pre-generic peak sales of over a billion dollars annually. This represents a decline of over 90% from peak performance.
The financial trajectory clearly illustrates the impact of patent expiration and generic competition on a blockbuster drug. The revenue stream generated during the period of market exclusivity is essential for recouping R&D costs and funding future innovation. Post-exclusivity, the focus shifts to managing the decline and maximizing remaining value.
What are the future market opportunities and challenges for memantine?
The future market for memantine, primarily in its generic form, is characterized by sustained demand due to the ongoing prevalence of Alzheimer's disease, but also by challenges related to therapeutic advancements and evolving treatment paradigms.
Market Opportunities:
- Persistent Alzheimer's Disease Burden: Alzheimer's disease affects millions worldwide, and the aging global population continues to increase the number of individuals diagnosed. This sustained patient population ensures a baseline demand for symptomatic treatments like memantine.
- Cost-Effectiveness of Generics: Generic memantine hydrochloride offers a significantly lower cost compared to branded NAMENDA, making it an accessible and attractive option for a broader patient base and healthcare systems facing cost constraints. This cost advantage will likely sustain its use, especially in regions with limited healthcare budgets.
- Combination Therapies: While not a disease-modifying agent, memantine's symptomatic benefits can complement emerging disease-modifying therapies. As new amyloid and tau-targeting drugs gain traction, memantine could be retained as an adjunctive therapy to manage cognitive and functional symptoms.
- Off-Label Uses and Expanded Indications (Potential): Research into memantine's efficacy in other neurological or psychiatric conditions continues. While not currently approved, any successful expansion of its indication could create new market segments. For example, studies have explored its use in Lewy body dementia, vascular dementia, and even certain psychiatric disorders.
Market Challenges:
- Advancements in Disease-Modifying Therapies: The development and approval of new drugs that target the underlying pathology of Alzheimer's disease (e.g., amyloid-beta clearance) represent the most significant challenge. These therapies, while still facing limitations in efficacy and safety, offer the potential to alter disease course, shifting focus away from purely symptomatic management.
- Limited Efficacy in Disease Progression: Memantine does not halt or reverse the neurodegenerative process of Alzheimer's disease. Its efficacy is limited to symptomatic improvement, and it does not address the root causes of the condition. This inherent limitation may lead prescribers to prioritize disease-modifying agents for eligible patients.
- Stagnant Market for Symptomatic Treatments: The market for symptomatic treatments for Alzheimer's has matured. While demand exists, there is limited scope for significant growth beyond market share shifts and the overall increase in the Alzheimer's patient population. Innovation in this segment is slower compared to disease-modifying approaches.
- Physician and Patient Education: As new treatment modalities emerge, there will be a continuous need for physician and patient education to guide appropriate treatment selection, balancing the benefits of symptomatic relief with the potential of disease-modifying therapies.
- Pricing Pressures in Generic Market: While generics offer cost advantages, the generic memantine market itself is highly competitive. This can lead to ongoing price erosion, limiting profitability for manufacturers.
The future trajectory of memantine will likely be one of stable, albeit lower-margin, demand as a cost-effective symptomatic treatment, coexisting with newer, more complex disease-modifying agents.
Key Takeaways
- NAMENDA (memantine hydrochloride) is an established symptomatic treatment for moderate to severe Alzheimer's disease.
- The drug has undergone patent expiration for both its immediate-release and extended-release formulations in major markets like the U.S.
- Following patent expiries, branded NAMENDA sales have significantly declined due to the introduction of generic memantine. Peak annual sales exceeded $1.4 billion, while recent sales are in the tens of millions of dollars.
- The Alzheimer's disease market is evolving with the advent of disease-modifying therapies targeting amyloid and tau pathology, posing a challenge to purely symptomatic treatments.
- Generic memantine is expected to maintain a stable market presence due to the persistent high prevalence of Alzheimer's disease and its cost-effectiveness.
- Future opportunities for memantine lie in its role as an accessible symptomatic treatment and potentially as an adjunct to new disease-modifying therapies.
- Key challenges include the increasing prominence of disease-modifying agents that aim to alter disease progression and ongoing price pressures within the generic market.
Frequently Asked Questions
-
What is the primary mechanism of action for NAMENDA?
NAMENDA is an NMDA receptor antagonist that modulates glutamate activity, aiming to reduce excitotoxicity associated with Alzheimer's disease.
-
When did generic versions of NAMENDA become available in the U.S.?
Generic memantine hydrochloride products began entering the U.S. market in 2017 following the expiration of key patents and exclusivity for the immediate-release formulation.
-
Does memantine cure Alzheimer's disease?
No, memantine is a symptomatic treatment. It helps manage cognitive and functional symptoms of moderate to severe Alzheimer's disease but does not halt, reverse, or cure the underlying disease progression.
-
What is the expected market share of generic memantine compared to branded NAMENDA post-exclusivity?
Post-exclusivity, generic memantine hydrochloride has captured the vast majority of the market share, with branded NAMENDA sales reduced to a minimal fraction due to price competition.
-
Are there any new therapeutic uses for memantine being investigated?
Research continues to explore memantine's potential benefits in other neurological and psychiatric conditions, though its primary approved indication remains Alzheimer's disease.
Citations
[1] Forest Laboratories. (2013). Forest Laboratories Announces Fourth Quarter and Fiscal Year 2013 Financial Results. [Press release]. Retrieved from [Source typically found in SEC filings or company investor relations archives, example: https://www.sec.gov/Archives/edgar/data/741308/000119312513227736/0001193125-13-227736-index.htm]
[2] Allergan plc. (2015). Allergan Reports Fourth Quarter and Full Year 2015 Results. [Press release]. Retrieved from [Source typically found in SEC filings or company investor relations archives, example: https://www.allergan.com/news/press-releases/2016/2/allergan-reports-fourth-quarter-and-full-year-2015-results]