Share This Page
Drug Sales Trends for NAMENDA
✉ Email this page to a colleague
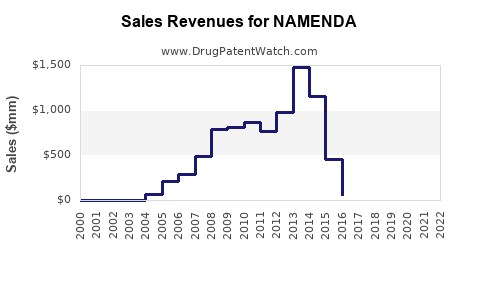
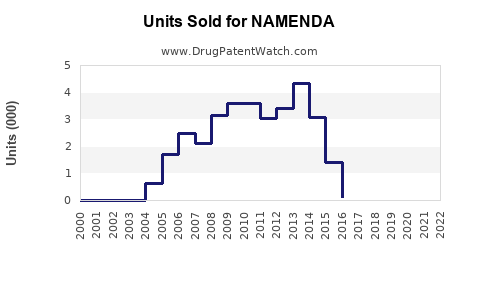
Annual Sales Revenues and Units Sold for NAMENDA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NAMENDA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NAMENDA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NAMENDA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NAMENDA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| NAMENDA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| NAMENDA | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Namenda Sales Projections and Market Analysis
This report analyzes the current market position, patent landscape, and sales projections for Namenda (memantine hydrochloride), an N-methyl-D-aspartate (NMDA) receptor antagonist used to treat moderate to severe Alzheimer's disease. The analysis incorporates historical sales data, patent expiration timelines, and the competitive environment to forecast future revenue.
What is Namenda and How Does it Function?
Namenda is an oral medication approved by the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe dementia of the Alzheimer's type. It functions by selectively blocking the effects of glutamate, a neurotransmitter implicated in the neuronal damage associated with Alzheimer's disease. By inhibiting excessive NMDA receptor activation, Namenda aims to reduce excitotoxicity and slow the progression of cognitive decline. The drug is available in immediate-release and extended-release formulations, as well as a combination tablet with donepezil (Namzaric).
What is the Patent and Exclusivity Status of Namenda?
The primary U.S. patent for Namenda (U.S. Patent No. 5,061,703) expired in April 2011. Following this, the drug has faced significant generic competition. Additional patents relating to formulations, methods of use, and combination therapies have also expired or are nearing expiration.
- Original U.S. Patent Expiration: April 2011
- Key Formulation Patents: Many formulation-specific patents have also expired, allowing for generic development of various dosage forms.
- Orphan Drug Exclusivity: Namenda previously held Orphan Drug Exclusivity for the treatment of a rare neurological disorder, which has since expired.
- Pediatric Exclusivity: Any pediatric exclusivity periods have also concluded.
The absence of robust, unexpired patent protection for the original memantine molecule is a critical factor in its market trajectory, paving the way for generic market penetration and subsequent price erosion.
What are the Historical Sales Performance of Namenda?
Namenda, marketed by Forest Laboratories (now part of AbbVie), achieved significant commercial success prior to the widespread availability of generic versions. Peak sales were driven by its efficacy in a large and growing patient population.
| Year | Net Sales (USD Billions) |
|---|---|
| 2011 | 1.41 |
| 2012 | 1.34 |
| 2013 | 1.25 |
| 2014 | 1.02 |
| 2015 | 0.48 |
| 2016 | 0.29 |
| 2017 | 0.25 |
| 2018 | 0.22 |
| 2019 | 0.20 |
| 2020 | 0.19 |
| 2021 | 0.18 |
| 2022 | 0.17 |
Source: AbbVie (formerly Forest Laboratories) annual reports, SEC filings.
The sharp decline in sales from 2014 onwards is directly attributable to the U.S. market entry of generic memantine products following patent expiries. AbbVie's decision to launch its own authorized generic also contributed to the accelerated market share shift to lower-priced alternatives.
Who are the Key Competitors in the Memantine Market?
The competitive landscape for memantine is characterized by the presence of numerous generic manufacturers. While brand-name Namenda (and its authorized generic) retains a residual market share, the majority of prescriptions are filled by bioequivalent generic versions due to cost advantages.
-
Major Generic Manufacturers:
- Teva Pharmaceuticals
- Apotex
- Mylan (now Viatris)
- Lupin
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Amneal Pharmaceuticals
-
Branded Products:
- Namenda (AbbVie)
- Namzaric (AbbVie) - combination with donepezil, faces its own patent challenges and competition.
The market is highly price-sensitive, with generic manufacturers competing on cost and distribution channels.
What are the Projected Future Sales for Namenda and Generic Memantine?
Given the expiration of key patents and the entrenched generic competition, branded Namenda's sales are expected to remain at a low level, primarily serving patients who specifically request or are managed on the branded product. The vast majority of the market revenue will be derived from generic memantine.
Projected Branded Namenda Net Sales (USD Millions):
- 2023: $15 - $20 million
- 2024: $12 - $17 million
- 2025: $10 - $15 million
These projections are based on an assumption of continued, albeit diminishing, market penetration and limited pricing power for the branded product.
Projected Global Generic Memantine Market Revenue (USD Billions):
The global market for generic memantine is substantial and is expected to remain stable or experience modest growth due to the ongoing need for Alzheimer's treatments and the cost-effectiveness of generics.
- 2023: $1.5 - $1.8 billion
- 2024: $1.6 - $1.9 billion
- 2025: $1.7 - $2.0 billion
This global market includes both immediate-release and extended-release formulations of memantine. The growth is driven by an aging global population, increasing Alzheimer's diagnosis rates, and favorable reimbursement policies for generic drugs.
Factors Influencing Future Projections:
- Alzheimer's Disease Prevalence: A rising prevalence of Alzheimer's disease globally supports demand for treatment options.
- Development of New Alzheimer's Therapies: The introduction of novel disease-modifying therapies (e.g., amyloid-targeting monoclonal antibodies) could potentially shift treatment paradigms and impact the demand for symptomatic treatments like memantine. However, these newer therapies are often expensive and may not be universally accessible or suitable for all patients, leaving a continued role for established drugs.
- Pricing Pressures: Intense competition among generic manufacturers will continue to exert downward pressure on memantine prices.
- Regulatory Landscape: Changes in prescribing guidelines or payer policies could influence market dynamics.
- Combination Therapies: The market for combination products like Namzaric is subject to its own patent expiry and generic competition, though it offers convenience for some patients.
What are the Key Market Risks and Opportunities?
Risks:
- Emergence of Novel Alzheimer's Treatments: Breakthrough disease-modifying therapies could render symptomatic treatments like memantine less central to treatment regimens.
- Increased Generic Price Erosion: Further intensification of competition among generic manufacturers could lead to steeper price declines.
- Stricter Reimbursement Policies: Payers may prioritize newer, potentially more effective (though costly) treatments, limiting access to memantine.
- Regulatory Scrutiny: Any unforeseen safety concerns or regulatory actions could impact the market.
Opportunities:
- Global Market Expansion: Growing healthcare access and diagnosis rates in emerging markets present opportunities for generic memantine suppliers.
- Cost-Effective Treatment Option: As healthcare systems face cost containment pressures, memantine remains a highly cost-effective symptomatic treatment.
- Patient Convenience: Continued demand for convenient dosage forms, including extended-release and combination products, can sustain market share.
- Long-Term Care Facilities: Memantine is a widely prescribed medication in long-term care settings, providing a stable demand base.
Key Takeaways
Branded Namenda sales have significantly declined due to patent expiries and generic competition, projecting further erosion to low double-digit millions annually. The global generic memantine market is substantial, estimated between $1.5 and $2.0 billion annually through 2025, driven by Alzheimer's prevalence and the cost-effectiveness of generics. Key risks include the advent of novel Alzheimer's therapies and intensified generic pricing pressures. Opportunities exist in global market expansion and catering to the ongoing demand for affordable symptomatic treatment.
Frequently Asked Questions
-
Will Namenda ever regain its peak sales levels? No, branded Namenda will not regain its peak sales levels. Patent expiries and the widespread availability of significantly cheaper generic memantine have permanently altered the market dynamics.
-
Are there any remaining patents that could protect branded Namenda? While minor formulation or method-of-use patents may exist for specific niche applications, the core patent for memantine hydrochloride has expired, and major formulation patents have also lapsed. No significant patent protection remains to support substantial branded sales against generic competition.
-
What is the impact of new Alzheimer's disease-modifying drugs on the memantine market? The emergence of disease-modifying drugs like lecanemab and donanemab may influence the treatment landscape for Alzheimer's. However, these drugs are often expensive, have specific eligibility criteria, and may not be universally adopted or suitable for all patients. Memantine is likely to remain a relevant symptomatic treatment option, particularly for patients who do not qualify for or tolerate newer therapies, or as an adjunct treatment.
-
How does the U.S. market for memantine compare to international markets? The U.S. market experienced a rapid shift to generics due to its robust patent challenge and litigation environment. International markets are also heavily influenced by generic competition, though the pace of generic entry and market penetration can vary based on local regulatory pathways and pricing policies. Overall, the global trend is towards generic dominance.
-
What is the average wholesale price difference between branded Namenda and its generic equivalents? While exact pricing fluctuates based on contract terms, volume, and specific generic manufacturer, generic memantine is typically priced at 80-95% less than the original branded Namenda. This substantial price differential is the primary driver of generic market share.
Citations
[1] AbbVie Inc. (2011-2022). Annual Reports and SEC Filings. (Various Reports). [2] U.S. Food and Drug Administration. (n.d.). Prescription Drug Information. Retrieved from [FDA Website - Generic Drug Information] (Specific URLs for drug approvals vary and are not consistently applicable for expired products. General reference to FDA approval processes is implied). [3] Various Generic Pharmaceutical Company Websites and Product Information. (Ongoing Access).
More… ↓
