MICARDIS Drug Patent Profile
✉ Email this page to a colleague
When do Micardis patents expire, and when can generic versions of Micardis launch?
Micardis is a drug marketed by Boehringer Ingelheim and is included in two NDAs.
The generic ingredient in MICARDIS is hydrochlorothiazide; telmisartan. There are thirty-two drug master file entries for this compound. Nine suppliers are listed for this compound. Additional details are available on the hydrochlorothiazide; telmisartan profile page.
Summary for MICARDIS
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 151 |
| Clinical Trials: | 71 |
| Patent Applications: | 4,727 |
| Formulation / Manufacturing: | see details |
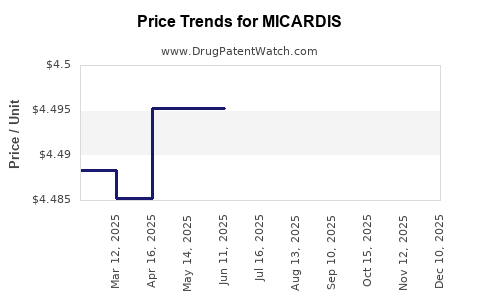
| Drug Prices: | Drug price information for MICARDIS |
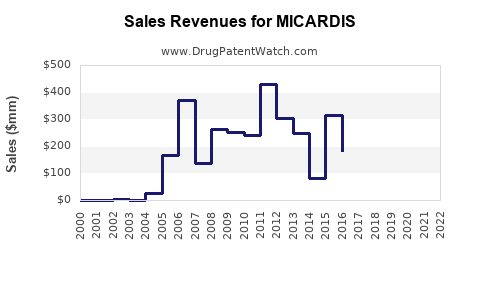
| Drug Sales Revenues: | Drug sales revenues for MICARDIS |
| What excipients (inactive ingredients) are in MICARDIS? | MICARDIS excipients list |
| DailyMed Link: | MICARDIS at DailyMed |
Recent Clinical Trials for MICARDIS
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Queens Medical Center | Phase 2 |
| University of Hawaii | Phase 2 |
| HK inno.N Corporation | Phase 1 |
Pharmacology for MICARDIS
| Drug Class | Angiotensin 2 Receptor Blocker |
| Mechanism of Action | Angiotensin 2 Receptor Antagonists |
Anatomical Therapeutic Chemical (ATC) Classes for MICARDIS
Paragraph IV (Patent) Challenges for MICARDIS
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| MICARDIS | Tablets | telmisartan | 20 mg, 40 mg and 80 mg | 020850 | 1 | 2006-12-26 |
US Patents and Regulatory Information for MICARDIS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-003 | Apr 4, 2000 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Boehringer Ingelheim | MICARDIS HCT | hydrochlorothiazide; telmisartan | TABLET;ORAL | 021162-001 | Nov 17, 2000 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-001 | Nov 10, 1998 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-002 | Nov 10, 1998 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Boehringer Ingelheim | MICARDIS HCT | hydrochlorothiazide; telmisartan | TABLET;ORAL | 021162-003 | Apr 19, 2004 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Boehringer Ingelheim | MICARDIS HCT | hydrochlorothiazide; telmisartan | TABLET;ORAL | 021162-002 | Nov 17, 2000 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for MICARDIS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-001 | Nov 10, 1998 | ⤷ Sign Up | ⤷ Sign Up |
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-003 | Apr 4, 2000 | ⤷ Sign Up | ⤷ Sign Up |
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-002 | Nov 10, 1998 | ⤷ Sign Up | ⤷ Sign Up |
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-002 | Nov 10, 1998 | ⤷ Sign Up | ⤷ Sign Up |
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-002 | Nov 10, 1998 | ⤷ Sign Up | ⤷ Sign Up |
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-001 | Nov 10, 1998 | ⤷ Sign Up | ⤷ Sign Up |
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-003 | Apr 4, 2000 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for MICARDIS
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim International GmbH | Micardis | telmisartan | EMEA/H/C/000209 HypertensionTreatment of essential hypertension in adults.Cardiovascular preventionReduction of cardiovascular morbidity in patients with:manifest atherothrombotic cardiovascular disease (history of coronary heart disease, stroke, or peripheral arterial disease) or;type-2 diabetes mellitus with documented target-organ damage. |
Authorised | no | no | no | 1998-12-16 | |
| Krka, d.d., Novo mesto | Tolura | telmisartan | EMEA/H/C/001196 HypertensionTreatment of essential hypertension in adults.Cardiovascular preventionReduction of cardiovascular morbidity in patients with:manifest atherothrombotic cardiovascular disease (history of coronary heart disease or peripheral arterial disease) or;type 2 diabetes mellitus with documented target organ damage. |
Authorised | yes | no | no | 2010-06-04 | |
| Teva B.V. | Telmisartan Teva Pharma | telmisartan | EMEA/H/C/002511 Treatment of essential hypertension in adults. |
Authorised | yes | no | no | 2011-10-03 | |
| Teva B.V. | Telmisartan Teva | telmisartan | EMEA/H/C/001146 Treatment of essential hypertension in adults |
Withdrawn | yes | no | no | 2010-01-25 | |
| Actavis Group PTC ehf | Telmisartan Actavis | telmisartan | EMEA/H/C/001168 HypertensionTreatment of essential hypertension in adults.Cardiovascular preventionReduction of cardiovascular morbidity in patients with:manifest atherothrombotic cardiovascular disease (history of coronary heart disease, stroke, or peripheral arterial disease) or;type 2 diabetes mellitus with documented target organ damage. |
Authorised | yes | no | no | 2010-09-29 | |
| Bayer AG | Kinzalmono (previously Telmisartan Boehringer Ingelheim Pharma KG) | telmisartan | EMEA/H/C/000211 HypertensionTreatment of essential hypertension in adults.Cardiovascular preventionReduction of cardiovascular morbidity in patients with:manifest atherothrombotic cardiovascular disease (history of coronary heart disease, stroke, or peripheral arterial disease) or;type-2 diabetes mellitus with documented target-organ damage. |
Authorised | no | no | no | 1998-12-16 | |
| Bayer AG | Pritor | telmisartan | EMEA/H/C/000210 HypertensionTreatment of essential hypertension in adults.Cardiovascular preventionReduction of cardiovascular morbidity in patients with:manifest atherothrombotic cardiovascular disease (history of coronary heart disease, stroke, or peripheral arterial disease) or;type-2 diabetes mellitus with documented target-organ damage. |
Authorised | no | no | no | 1998-12-11 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for MICARDIS
See the table below for patents covering MICARDIS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Austria | 294579 | ⤷ Sign Up | |
| Poland | 197164 | ⤷ Sign Up | |
| Hong Kong | 1041485 | ⤷ Sign Up | |
| Argentina | 035475 | FORMA B POLIMORFA DEL TELMISARTAN, LAS MEZCLAS DE LOS POLIMORFOS DEL TELMISARTAN, UN PROCEDIMIENTO PARA LA PREPARACION DEL TELMISARTAN QUE CONTIENE LA FORMA B POLIMORFA, Y EL USO DEL TELMISARTAN PARA PREPARAR UN MEDICAMENTO | ⤷ Sign Up |
| China | 1050122 | ⤷ Sign Up | |
| South Africa | 9302521 | ⤷ Sign Up | |
| New Zealand | 513528 | Telmisartan polymorphs and their use as angiotensin antagonists | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for MICARDIS
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0502314 | C300095 | Netherlands | ⤷ Sign Up | PRODCUT NAME: TELMISARTAN, DESGEWENST IN DE VORM VAN EEN FYSIOLOGISCH VERDRAAGBAAR ZOUT, EN HYDROCHLOROTHIAZIDE; REGISTRATION NO/DATE: EU/1/02/213/001-010 20020419 |
| 0502314 | C300478 | Netherlands | ⤷ Sign Up | PRODUCT NAME: TELMISARTAN, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, EN AMLODIPINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER AMLODIPINEBESILAAT; REGISTRATION NO/DATE: EU/1/10/648/001-028 20101007 |
| 0502314 | 24/2002 | Austria | ⤷ Sign Up | PRODUCT NAME: TELMISARTAN, GEGEBENENFALLS IN FORM SEINER PHARMAZEUTISCH ANNEHMBAREN SALZE, UND HYDROCHLOROTHIAZID; REGISTRATION NO/DATE: EU/1/02/213/001- EU/1/02/213/010 20020419 |
| 0502314 | C990007 | Netherlands | ⤷ Sign Up | PRODUCT NAME: TELMISARTAN, DESGEWENST IN DE VORM VAN EEN FYSIOLOGISCH VERDRAAGBAAR ZOUT; REGISTRATION NO/DATE: EU/1/98/089/001-EU/1/98/089/010 19981211 |
| 0502314 | 3/2011 | Austria | ⤷ Sign Up | PRODUCT NAME: TELMISARTAN, GGF. IN FORM SEINER PHARMAZEUTISCH ANNEHMBAREN SALZE UND AMLODIPIN, GGF. IN FORM SEINER PHARMAZEUTISCH ANNEHMBAREN SALZE, INSBESONDERE AMLODIPINBESILAT; REGISTRATION NO/DATE: EU/1/10/648/001 - EU/1/10/648/028 20101007 |
| 0502314 | 99C0014 | Belgium | ⤷ Sign Up | PRODUCT NAME: TELMISARTAN REGISTRATION NO/DATE: EU/1/98/089/001 / 19981211 |
| 0502314 | 23/1999 | Austria | ⤷ Sign Up | PRODUCT NAME: TELMISARTAN, GEGEBENENFALLS IN FORM EINES PHARMAZEUTISCH ANNEHMBAREN SALZES; NAT. REGISTRATION NO/DATE: EU/1/98/089/001-010 19981211; FIRST REGISTRATION: BE K (1998) 4288-DE 19981211 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


