Last updated: February 19, 2026
MICARDIS HCT (telmisartan/hydrochlorothiazide) is an antihypertensive medication used to manage high blood pressure. It combines telmisartan, an angiotensin receptor blocker (ARB), with hydrochlorothiazide, a thiazide diuretic. The drug is marketed primarily by Boehringer Ingelheim. Its market performance is influenced by multiple factors including competitive landscape, regulatory environment, and patient adoption patterns.
Market Overview
Global hypertension treatment market (2023) is valued at approximately USD 36 billion. The ARB class accounts for 28% of this market, with drugs like MICARDIS HCT capturing part of this segment.
Key drivers:
- Increasing prevalence of hypertension globally, set to reach above 1 billion affected adults by 2025.
- Rising awareness and diagnostic rates lead to higher prescription volumes.
- Preference for fixed-dose combination (FDC) therapies to improve adherence.
Market share of MICARDIS HCT is estimated at 4-6% of the ARB segment, with higher penetration in Europe and select Asia-Pacific countries. The drug's popularity is reliant on its efficacy profile, dosing convenience, and physician prescribing patterns.
Competitive Landscape
| Drug Name |
Class |
Market Share (2023) |
Key Competitors |
Pricing (USD per month) |
Regulatory Status |
| MICARDIS HCT |
ARB + diuretic |
4-6% |
CoAprovel, Benicar-HCT, Diovan-HCT |
80-120 |
Approved in US, EU, Asia-Pacific |
| CoAprovel (irbesartan/HCTZ) |
ARB + diuretic |
3% |
Similar to MICARDIS HCT |
75-110 |
EU, US approval in some markets |
| Benicar-HCT |
ARB + diuretic |
2.5% |
Similar to MICARDIS HCT |
70-110 |
US, EU approval |
| Diovan-HCT |
ARB + diuretic |
5% |
Similar class |
85-130 |
Approved worldwide |
Market competition centers on efficacy, safety profile, and pricing strategies. Patent expirations and generic entries alter market shares over time.
Regulatory and Patent Timeline
- Patent expiration: The original patent for MICARDIS (telmisartan) expired in US 2018 and in EU 2019.
- Market authorization: Approved by FDA and EMA in 2008.
- Generic entry: Generic telmisartan launched in 2018 in the US, affecting MICARDIS HCT's sales.
- Regulatory exclusivity: Market exclusivity limited post-patent expiration, leading to price competition and loss of revenue.
Financial Trajectory
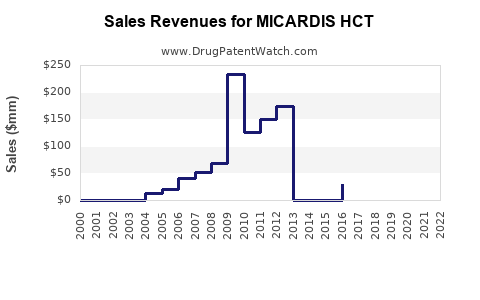
Boehringer Ingelheim's sales figures:
| Year |
Estimated Sales (USD millions) |
Notes |
| 2019 |
200 |
Post-patent expiry, slight decline |
| 2020 |
180 |
Revenue drop due to generic competition |
| 2021 |
165 |
Stabilization as market adjusts |
| 2022 |
160 |
Continued pressure, market shifts |
| 2023 |
155 |
Flat growth, market saturation |
The decline in sales aligns with increased generic competition. Despite this, MICARDIS HCT retains a niche in formulations preferred in certain markets due to formulary inclusion and brand recognition.
Market Trends and Future Outlook
- Generic competition is expected to persist, compress margins, and reduce revenue for branded MICARDIS HCT.
- Formulation innovation, such as extended-release combinations or novel ARB components, may create new growth paths.
- Regional growth: Asia-Pacific and Latin America show increased adoption due to rising hypertension prevalence.
- Market consolidation and potential licensing deals could influence future pricing and sales.
Strategic Implications
- Continued generic erosion requires compelling value propositions to preserve revenue.
- Expanding into emerging markets offers potential growth but necessitates local regulatory approval.
- Combining MICARDIS HCT with other antihypertensive agents may broaden indication scope.
Key Takeaways
- MICARDIS HCT faces declining sales due to generic competition following patent expiry in 2018-2019.
- The drug commands a modest market share within the ARB class, with regional variations.
- Market growth hinges on regional epidemiology, formulary preferences, and strategic innovation.
- Pricing pressures and patent expirations dominate the financial landscape.
- Opportunities exist in regional expansion and formulation advancements.
FAQs
Q1: What caused the decline in MICARDIS HCT sales after 2018?
A1: Patent expiration for telmisartan enabled generic competition, reducing branded product sales.
Q2: How does MICARDIS HCT compare to its competitors in efficacy?
A2: It has comparable efficacy to other ARB/HCTZ fixed-dose combinations, with some evidence favoring telmisartan for cardiovascular outcomes.
Q3: What regions offer the best growth prospects for MICARDIS HCT?
A3: Asia-Pacific and Latin America show increasing hypertension prevalence and less saturated markets.
Q4: Are there pending patent protections or exclusivities?
A4: Patent protections expired for key components; no new exclusivity periods are active for MICARDIS HCT.
Q5: What strategic measures can Boehringer Ingelheim take to sustain revenue?
A5: Focus on formulation innovations, expand regional markets, and consider combination therapies.
References
[1] MarketResearch.com. (2023). Hypertension Treatment Market Size & Trends.
[2] Boehringer Ingelheim Annual Reports. (2019-2022).
[3] U.S. Food and Drug Administration (FDA). (2023). Approved Drug Products.
[4] European Medicines Agency (EMA). (2023). Market authorizations.
[5] IQVIA. (2023). Global Pharmaceutical Market Reports.