Last updated: February 19, 2026
MICARDIS (telmisartan), an angiotensin II receptor blocker (ARB), has navigated a complex market landscape marked by patent expirations, generic competition, and evolving therapeutic guidelines for hypertension. Its journey reflects broader trends in the cardiovascular drug market.
What is the core mechanism of MICARDIS and its therapeutic indication?
MICARDIS is a selective angiotensin II receptor antagonist. It blocks the action of angiotensin II, a substance produced by the body that constricts blood vessels. This constriction increases blood pressure. By blocking angiotensin II, telmisartan relaxes blood vessels, thereby lowering blood pressure.
The primary therapeutic indication for MICARDIS is the treatment of hypertension (high blood pressure) [1]. It is used as a monotherapy or in combination with other antihypertensive agents.
What is the patent history and exclusivity landscape for MICARDIS?
MICARDIS, developed by Boehringer Ingelheim, has a significant patent history that has shaped its market exclusivity and subsequent generic entry.
- Primary Patent Filing: The original patent for telmisartan was filed in the United States in the late 1990s.
- US Patent Expiration: The key compound patent for telmisartan in the U.S. expired in October 2012. This expiration marked the entry of generic telmisartan products into the market.
- Exclusivity Periods: Prior to patent expiration, MICARDIS benefited from various exclusivity periods, including:
- New Chemical Entity (NCE) Exclusivity: Upon FDA approval, NCEs receive a five-year period of market exclusivity in the U.S.
- Orphan Drug Exclusivity: Not applicable to telmisartan.
- Patent Term Extension (PTE): Boehringer Ingelheim sought and obtained PTE for certain patents covering telmisartan, extending market exclusivity beyond the original patent expiry date.
- Pediatric Exclusivity: An additional six months of exclusivity was granted for conducting studies in pediatric populations.
- European Patent Expiration: Similar patent expirations occurred in major European markets, leading to the introduction of generic versions in the EU.
- Post-Patent Exclusivity Strategies: Boehringer Ingelheim pursued various strategies to maintain market share post-patent expiry, including:
- Combination Products: Development and marketing of fixed-dose combination products, such as MICARDIS HCT (telmisartan/hydrochlorothiazide) and MICARDIS PLUS (telmisartan/amlodipine), to offer enhanced efficacy and convenience, extending the product lifecycle.
- Life Cycle Management: Focus on physician education, patient adherence programs, and branding efforts.
What has been the sales performance of MICARDIS globally?
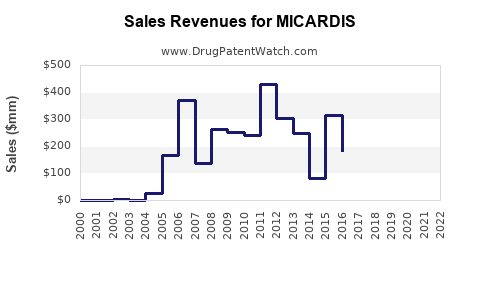
MICARDIS has been a significant contributor to Boehringer Ingelheim's revenue, particularly during its period of market exclusivity. Its sales trajectory reflects the typical pattern of branded pharmaceuticals experiencing strong growth followed by a decline post-generic entry.
| Year |
Global Sales (USD Millions) |
Notes |
| 2007 |
991 |
Strong growth phase before patent expiry. |
| 2008 |
1,272 |
Continued strong performance. |
| 2009 |
1,453 |
Peak sales year for branded MICARDIS. |
| 2010 |
1,455 |
Sales plateaued. |
| 2011 |
1,382 |
Slight decline as generic anticipation grew. |
| 2012 |
1,160 |
Significant decline as U.S. patent expired. |
| 2013 |
850 |
Post-patent expiry impact, generic competition intensifies. |
| 2014 |
630 |
Further erosion of market share. |
| 2015 |
470 |
Continued sales decline. |
| 2016 |
330 |
Sales continue to decrease. |
| 2017 |
240 |
Substantial reduction in branded sales. |
| 2018 |
190 |
Ongoing generic market dominance. |
| 2019 |
140 |
Minimal remaining branded sales. |
| 2020 |
110 |
Continued decline, primarily in niche markets or specific formulations. |
| 2021 |
90 |
Branded MICARDIS sales represent a small fraction of peak. |
Source: Boehringer Ingelheim Annual Reports, Market Research Data. Figures are approximate and may vary slightly based on reporting methodology.
The peak sales of MICARDIS in 2009 and 2010 underscore its success as a blockbuster drug within the antihypertensive market. The subsequent sharp decline from 2012 onwards is a direct consequence of generic telmisartan entering the market, significantly reducing the price and market share of the branded product.
What is the current market penetration of generic telmisartan?
Following the patent expiration of MICARDIS, the market for telmisartan has become largely dominated by generic manufacturers.
- Market Share: Generic telmisartan formulations account for over 90% of the total telmisartan market in developed countries.
- Pricing: The average selling price (ASP) of generic telmisartan is estimated to be 70-90% lower than the peak ASP of branded MICARDIS.
- Number of Manufacturers: Numerous pharmaceutical companies globally produce and market generic telmisartan, including major players like Teva Pharmaceutical Industries, Mylan (now Viatris), and various Indian and Chinese manufacturers.
- Formulations: Generic telmisartan is available in various dosages (e.g., 20 mg, 40 mg, 80 mg) and as combination products with other antihypertensives like hydrochlorothiazide and amlodipine.
- Prescribing Patterns: Physician preference has largely shifted towards generic telmisartan due to cost-effectiveness, with brand-name prescriptions being rare and often driven by specific formulary or patient access issues.
The high penetration of generics signifies the commoditization of telmisartan as an active pharmaceutical ingredient, with competition focused primarily on cost, manufacturing efficiency, and distribution networks.
What are the key clinical and regulatory considerations impacting MICARDIS's market position?
The clinical profile and regulatory environment have played a crucial role in the trajectory of MICARDIS.
- Cardiovascular Outcome Trials: Telmisartan, including MICARDIS, has been studied in large-scale cardiovascular outcome trials. The ONTARGET (Ongoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial) was a landmark study. It demonstrated that telmisartan was as effective as ramipril in reducing cardiovascular events in high-risk patients. However, the combination of telmisartan and ramipril did not provide further benefit and was associated with increased adverse events like hyperkalemia and renal dysfunction compared to monotherapy [2]. This trial provided strong evidence for telmisartan's efficacy but also highlighted potential risks in combination therapy.
- Hypertension Guidelines: Evolving hypertension treatment guidelines from organizations such as the American Heart Association (AHA) and the European Society of Cardiology (ESC) influence prescribing patterns. These guidelines generally recommend ARBs as a first-line or second-line therapy for hypertension, often in conjunction with lifestyle modifications. Telmisartan's established efficacy and safety profile position it favorably within these guidelines.
- Safety Profile: MICARDIS generally has a favorable safety profile, with common side effects including dizziness, fatigue, and muscle pain. Angioedema and renal impairment are rare but serious adverse events. Its tolerability profile is a key factor in its clinical utility.
- Regulatory Status: MICARDIS holds marketing authorizations in numerous countries worldwide. Regulatory bodies monitor post-marketing surveillance for safety and efficacy.
- Combination Products: The approval and marketing of fixed-dose combinations (e.g., MICARDIS HCT, MICARDIS PLUS) were strategic regulatory and commercial moves by Boehringer Ingelheim to maintain market relevance and capture value beyond the single-agent patent expiry. These combinations are subject to their own regulatory reviews and approvals.
- Post-Marketing Studies: Boehringer Ingelheim has conducted and continues to support post-marketing studies to further elucidate the long-term benefits and safety of telmisartan, particularly in specific patient populations or as part of comprehensive cardiovascular risk management.
What is the financial outlook for MICARDIS and its generic alternatives?
The financial future of MICARDIS as a branded product is limited. The primary revenue generation has shifted from branded MICARDIS to generic telmisartan.
- Branded MICARDIS: The financial outlook for branded MICARDIS is characterized by continued, albeit slow, decline in sales. Revenue will primarily come from residual markets, niche indications, or specific payer contracts where branded products may still hold a marginal preference. Boehringer Ingelheim's focus has largely shifted to other therapeutic areas and newer products.
- Generic Telmisartan Market: The financial outlook for generic telmisartan is stable and driven by volume.
- Market Size: The global market for generic telmisartan is estimated to be in the hundreds of millions of dollars annually, driven by widespread use in hypertension management.
- Profitability: Profit margins for generic manufacturers are typically lower than for branded drugs, but consistent demand ensures ongoing revenue streams. Competition among generic manufacturers keeps prices competitive.
- Growth Drivers: Demand for generic telmisartan is supported by an aging global population, increasing prevalence of cardiovascular diseases, and cost-containment measures by healthcare systems encouraging the use of generics.
- Competition: The market is highly competitive, with numerous players vying for market share. This intense competition limits significant price increases.
- Emerging Markets: Growth in emerging markets, where access to branded medications can be limited by cost, is a significant driver for generic drug sales.
The overall financial trajectory for the telmisartan molecule is one of sustained, high-volume sales for generic manufacturers, with branded MICARDIS playing a minimal role.
Key Takeaways
- MICARDIS (telmisartan) was a highly successful branded antihypertensive drug whose market exclusivity ended with patent expirations in the U.S. (2012) and subsequent global market entries for generic telmisartan.
- Branded MICARDIS sales peaked around $1.45 billion annually before patent expiry and have since declined significantly to below $100 million, largely due to intense generic competition.
- The global telmisartan market is now dominated by generic manufacturers, accounting for over 90% of sales by volume.
- Boehringer Ingelheim mitigated some patent cliff impact through the development and marketing of fixed-dose combination products, extending the lifecycle of telmisartan-containing therapies.
- Telmisartan's established efficacy, favorable safety profile, and inclusion in international hypertension treatment guidelines support continued widespread use, primarily through generic formulations.
- The financial outlook for branded MICARDIS is a steady decline, while generic telmisartan sales are expected to remain stable, driven by volume, cost-effectiveness, and global demand for hypertension management.
Frequently Asked Questions
-
What is the primary difference between MICARDIS and generic telmisartan?
The primary difference is the manufacturer and branding. MICARDIS is the original branded product developed by Boehringer Ingelheim. Generic telmisartan is produced by various other pharmaceutical companies after the patent on the original drug expired. The active ingredient, telmisartan, is chemically identical, and bioequivalence studies confirm they have the same safety and efficacy profiles.
-
Are there any therapeutic advantages to using branded MICARDIS over generic telmisartan?
No, from a therapeutic standpoint, there are no advantages to using branded MICARDIS over generic telmisartan. Regulatory agencies require generic drugs to be bioequivalent to their brand-name counterparts, meaning they deliver the same amount of active ingredient into the bloodstream over the same period.
-
What are the major side effects associated with telmisartan (MICARDIS)?
Common side effects of telmisartan include dizziness, fatigue, and muscle pain. Less common but more serious side effects can include angioedema (swelling of the face, lips, tongue, or throat), kidney problems, and hyperkalemia (high potassium levels in the blood). Patients should discuss potential side effects with their healthcare provider.
-
Can MICARDIS be taken with other medications for high blood pressure?
Yes, telmisartan is often prescribed in combination with other antihypertensive medications, such as diuretics (like hydrochlorothiazide) or calcium channel blockers (like amlodipine), to achieve better blood pressure control. Boehringer Ingelheim also marketed fixed-dose combination products containing telmisartan with these agents.
-
What is the current therapeutic role of telmisartan in cardiovascular risk reduction beyond hypertension management?
Telmisartan has demonstrated efficacy in reducing major cardiovascular events in high-risk patients, as shown in the ONTARGET trial. While not typically a first-line agent solely for cardiovascular risk reduction in the absence of hypertension, its benefit in patients with existing hypertension and cardiovascular risk factors is recognized. Its anti-inflammatory and antioxidant properties are also subjects of ongoing research.
Citations
[1] Boehringer Ingelheim. (n.d.). Micardis Prescribing Information. U.S. Food and Drug Administration.
[2] The ONTARGET Investigators. (2008). Telmisartan, ramipril, or both in patients at high risk for vascular events. New England Journal of Medicine, 358(15), 1547–1559. https://doi.org/10.1056/NEJMoa0801312