Last updated: February 19, 2026
Lactulose, a synthetic disaccharide, plays a critical role in managing hepatic encephalopathy and chronic constipation. Its market trajectory is shaped by patent expirations, generic competition, evolving treatment guidelines, and the emergence of novel therapeutic approaches. This analysis details the current market landscape, patent status, competitive environment, and future financial outlook for lactulose.
What is the Current Market Size and Growth Rate for Lactulose?
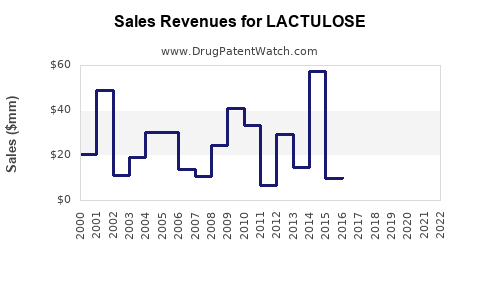
The global lactulose market was valued at approximately $550 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.5% over the next five to seven years, reaching an estimated $700 million to $750 million by 2030. This growth is primarily driven by an aging global population, increasing prevalence of liver diseases such as cirrhosis, and a higher incidence of gastrointestinal disorders. The demand for cost-effective laxatives also contributes to market expansion, particularly in emerging economies.
Market Segmentation
The lactulose market can be segmented by:
- Product Type: Oral solutions, powders, and enemas. Oral solutions represent the largest segment due to ease of administration.
- Indication: Hepatic encephalopathy and chronic constipation are the primary therapeutic areas. Hepatic encephalopathy treatment accounts for a significant portion of the market due to the chronic nature of the condition and the established efficacy of lactulose.
- Distribution Channel: Hospitals, retail pharmacies, and online pharmacies. Retail pharmacies hold the largest market share.
- Region: North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. North America and Europe are the dominant regions due to higher healthcare expenditure and established patient populations. The Asia Pacific region is expected to exhibit the fastest growth due to increasing chronic disease burden and improving healthcare infrastructure.
What is the Patent Landscape for Lactulose?
Lactulose itself is a well-established compound and its original patents have long expired. The key patent activity surrounding lactulose is not on the molecule itself, but on:
- Formulation Patents: These patents protect novel formulations that improve lactulose's taste, stability, or absorption. For example, patents may cover specific flavoring agents, preservatives, or controlled-release mechanisms.
- Manufacturing Process Patents: Innovations in the synthesis or purification of lactulose can be patented, leading to more efficient or cost-effective production methods.
- Combination Therapy Patents: Patents may exist for the use of lactulose in combination with other active pharmaceutical ingredients (APIs) for synergistic therapeutic effects.
- New Indication Patents: While less common for lactulose given its established uses, research into new therapeutic applications could lead to new patent filings.
As of 2024, the landscape is dominated by generic manufacturers. The patent expiry of any significant, novel lactulose formulations or processes would directly impact market exclusivity for the innovator. However, the absence of new blockbuster lactulose formulations means the market is largely open to generic competition. Key patent expirations that occurred in the early 2000s removed major barriers to entry for generic manufacturers, leading to price erosion.
Key Patent Expiration Impacts
- Increased Generic Entry: Following original patent expiries, numerous generic versions of lactulose entered the market.
- Price Reduction: The influx of generics significantly lowered the price of lactulose, making it a more accessible treatment option.
- Market Concentration Shift: The market share shifted from a few branded manufacturers to a larger number of generic producers.
Companies with proprietary formulations or enhanced manufacturing processes may still hold some patent protection, but these are typically limited in scope and duration compared to primary molecule patents.
Who are the Key Players in the Lactulose Market?
The lactulose market is characterized by a mix of established pharmaceutical companies and a significant number of generic manufacturers. The generic segment is highly competitive, with several players offering comparable products.
Major Manufacturers and Suppliers
- Branded/Innovator (Limited Scope): While original patents have expired, companies that have developed novel formulations or specific delivery systems may still have some market presence. These are often smaller players focusing on niche segments or higher-margin formulations.
- Generic Manufacturers: This segment comprises the bulk of the market. Key players include:
- Bayer AG: Historically a significant player, though its focus may have shifted.
- Abbott Laboratories: Offers lactulose products in various markets.
- Fresenius Kabi AG: A prominent supplier of specialty pharmaceuticals, including lactulose for hospital use.
- Dr. Reddy's Laboratories: A major global generic pharmaceutical company with lactulose offerings.
- Teva Pharmaceutical Industries Ltd.: A leading generic manufacturer with a broad portfolio.
- Sun Pharmaceutical Industries Ltd.: Another significant Indian generic company with a global presence.
- Mylan N.V. (now Viatris Inc.): A substantial player in the global generics market.
- Sanofi S.A.: Offers lactulose under various brand names in different regions.
The competitive landscape is dynamic, with mergers and acquisitions frequently reshaping market share. Pricing strategies, supply chain efficiency, and regulatory compliance are critical for success in the generic lactulose market.
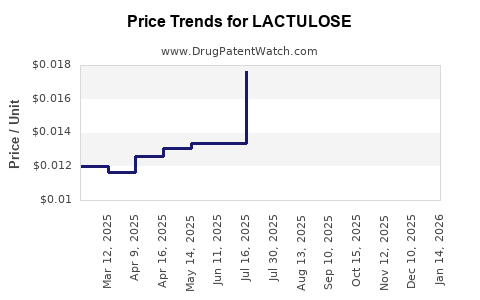
What are the Competitive Dynamics and Pricing Strategies?
The competitive dynamics in the lactulose market are primarily driven by price, product availability, and manufacturing efficiency, especially within the generic segment.
Competitive Factors
- Price Sensitivity: As a mature drug with widespread generic availability, lactulose is highly price-sensitive. Buyers, including pharmacies, hospitals, and government tenders, often negotiate based on cost.
- Quality and Bioequivalence: Generic manufacturers must demonstrate bioequivalence to the reference product. Consistent quality control and adherence to regulatory standards (e.g., FDA, EMA) are paramount.
- Supply Chain Reliability: Ensuring a stable and consistent supply is crucial, particularly for bulk purchases and hospital procurement. Disruptions can lead to loss of contracts.
- Formulation Differentiation: While the core molecule is generic, companies that offer improved formulations (e.g., better taste masking, more convenient packaging) can command a slight premium or gain market preference. However, these advantages are often temporary as competitors replicate or improve upon them.
- Market Access and Distribution: Establishing strong distribution networks and securing formulary placement in hospitals and healthcare systems are vital.
Pricing Strategies
- Cost-Plus Pricing: Generic manufacturers typically employ a cost-plus pricing strategy, factoring in manufacturing costs, regulatory compliance, marketing, and a desired profit margin.
- Competitive Pricing: Prices are heavily influenced by competitor pricing. Manufacturers often align their prices with or slightly undercut competitors to gain market share.
- Tender and Volume-Based Pricing: For large institutional buyers (hospitals, government health programs), significant discounts are offered based on contract volume and duration.
- Geographic Price Variation: Prices vary significantly between regions due to differences in healthcare systems, regulatory environments, local competition, and disposable incomes. Developed markets typically have higher prices than emerging markets.
The sustained price erosion due to generic competition has made lactulose a high-volume, lower-margin product for most manufacturers. Profitability hinges on efficient production and significant sales volumes.
What are the Regulatory Considerations and Market Access Challenges?
Navigating the regulatory landscape and securing market access are critical for lactulose manufacturers, particularly for generic products.
Key Regulatory Aspects
- Abbreviated New Drug Application (ANDA) / Marketing Authorization Application (MAA): Generic manufacturers must submit detailed applications to regulatory agencies (e.g., FDA in the US, EMA in Europe) demonstrating that their product is bioequivalent to the reference listed drug and meets all quality and safety standards.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality, consistency, and safety. Regular inspections by regulatory authorities are common.
- Pharmacopeial Standards: Lactulose products must comply with standards set by official pharmacopoeias, such as the United States Pharmacopeia (USP) and the European Pharmacopoeia (Ph. Eur.), for identity, purity, and strength.
- Labeling and Packaging Requirements: Strict regulations govern product labeling, including indication, dosage, warnings, and contraindications, to ensure patient safety. Packaging must also meet specific standards to maintain product integrity.
Market Access Challenges
- Reimbursement Policies: While lactulose is generally considered a cost-effective treatment, reimbursement policies can vary by country and payer. Securing favorable reimbursement status is important for broad market access.
- Healthcare System Procurement: In many countries, lactulose is procured through government tenders or hospital group purchasing organizations. Manufacturers must navigate these complex procurement processes to secure sales.
- Competition from Other Laxatives: Lactulose competes with a wide range of laxative products, including osmotic laxatives (e.g., polyethylene glycol), stimulant laxatives, and bulk-forming agents. Treatment guidelines and physician preferences can influence product choice.
- Therapeutic Guidelines: Evolving clinical guidelines for managing constipation and hepatic encephalopathy can impact the preferred position of lactulose. Manufacturers must stay abreast of these changes.
- Emergence of Novel Therapies: The development of new drugs for liver diseases or gastrointestinal disorders could potentially displace lactulose in certain treatment algorithms, though its established safety profile and low cost remain advantages.
What is the Future Outlook and Financial Trajectory?
The financial trajectory of lactulose is characterized by steady but modest growth, largely sustained by its established therapeutic value and cost-effectiveness.
Projected Financial Performance
- Revenue Growth: Modest revenue growth is expected, driven by increasing global demand for gastrointestinal and liver disease treatments. The CAGR is projected to remain in the 3.5% to 4.5% range.
- Profitability: Profitability for generic manufacturers will continue to be squeezed by intense price competition. Economies of scale, efficient manufacturing, and stringent cost control are essential for maintaining healthy profit margins.
- Market Consolidation: The highly fragmented generic market may see further consolidation as smaller players struggle to compete. Larger manufacturers with efficient supply chains and broad product portfolios are likely to gain market share.
- Geographic Expansion: Emerging markets in Asia Pacific and Latin America are anticipated to be key growth drivers, offering higher volume potential as healthcare access improves.
- Investment Considerations: For investors, lactulose represents a stable, albeit low-growth, segment of the pharmaceutical market. Companies with strong manufacturing capabilities, robust supply chains, and effective market access strategies are best positioned. The primary risks include ongoing price erosion and the potential, albeit low, for disruptive therapeutic advancements.
Key Growth Drivers
- Aging Population: The increasing proportion of elderly individuals globally leads to a higher prevalence of chronic constipation and liver-related complications.
- Rising Incidence of Chronic Liver Diseases: Conditions like cirrhosis, often linked to fatty liver disease and viral hepatitis, drive demand for lactulose in managing hepatic encephalopathy.
- Cost-Effectiveness: Lactulose remains one of the most affordable treatment options for its primary indications, making it attractive in both developed and developing healthcare systems.
- Physician Preference and Established Efficacy: Decades of clinical use have established lactulose's efficacy and safety profile, leading to continued physician confidence.
The financial future of lactulose is one of stable demand meeting continued competitive pressures. Innovation in formulation or delivery methods could offer marginal upside, but the core market will remain driven by generic competition and volume.
Key Takeaways
- The global lactulose market is projected to grow at a CAGR of 3.5%-4.5%, reaching $700-$750 million by 2030.
- Original lactulose patents have expired, leading to a market dominated by generic competition and significant price sensitivity.
- Key market players are primarily generic manufacturers, including Teva, Dr. Reddy's, and Sun Pharma.
- Pricing strategies are largely driven by cost-plus and competitive dynamics, with significant volume-based discounts for institutional buyers.
- Regulatory hurdles include ANDA/MAA submissions and strict adherence to GMP, while market access is influenced by reimbursement policies and healthcare procurement processes.
- Future financial trajectory points to modest revenue growth with pressure on profit margins due to competition, while emerging markets offer significant expansion potential.
Frequently Asked Questions
1. What are the primary therapeutic indications for lactulose?
Lactulose is primarily used to treat hepatic encephalopathy, a complication of liver disease that affects brain function, and chronic constipation.
2. What is the typical shelf life of lactulose oral solutions?
Lactulose oral solutions generally have a shelf life of 2 to 3 years when stored at room temperature, protected from light. Specific product labeling should always be consulted for precise expiration dates.
3. How does lactulose differ from other osmotic laxatives?
Lactulose is a non-absorbable disaccharide that works by drawing water into the colon, softening stool and increasing bowel movements. Other osmotic laxatives, such as polyethylene glycol (PEG), also draw water into the colon but achieve this through different chemical mechanisms and may have slightly different onset times and efficacy profiles for certain patients.
4. Are there any significant side effects associated with lactulose?
Common side effects include bloating, gas, abdominal discomfort, and diarrhea. These are usually dose-dependent. In patients with hepatic encephalopathy, the dosage is carefully titrated to avoid excessive diarrhea.
5. What is the mechanism of action for lactulose in treating hepatic encephalopathy?
In hepatic encephalopathy, lactulose is thought to work by reducing ammonia absorption in the colon. Gut bacteria metabolize lactulose into organic acids, which lowers the colonic pH. This converts ammonia (NH3) into ammonium (NH4+), a form that is less easily absorbed and is excreted in the stool. Additionally, the osmotic effect of lactulose can accelerate transit time, further reducing ammonia absorption.
Citations
[1] Global Market Insights. (2024). Lactulose Market Size, Share & Trends Analysis Report By Product, By Indication, By Region, And Segment Forecasts, 2024 – 2032.
[2] Grand View Research. (2023). Lactulose Market Size, Share & Trends Analysis Report By Product (Oral Solution, Powder, Enema), By Indication (Hepatic Encephalopathy, Chronic Constipation), By Distribution Channel, By Region, And Segment Forecasts, 2023 – 2030.
[3] Various pharmaceutical company annual reports and investor relations disclosures (e.g., Teva Pharmaceutical Industries Ltd., Dr. Reddy's Laboratories, Sun Pharmaceutical Industries Ltd.).
[4] U.S. Food and Drug Administration (FDA). (n.d.). Guidance for Industry.
[5] European Medicines Agency (EMA). (n.d.). Scientific Guidelines.