Last updated: February 19, 2026
Gabapentin is an antiepileptic drug used to treat partial seizures and neuropathic pain. Its market trajectory is shaped by patent expirations, generic competition, evolving therapeutic guidelines, and increasing use in off-label indications.
What are Gabapentin's Primary Approved Indications?
Gabapentin is approved by the U.S. Food and Drug Administration (FDA) for the following uses:
- Epilepsy: Adjunctive therapy in the treatment of partial seizures in patients 3 years of age and older.
- Postherpetic Neuralgia: Treatment of postherpetic neuralgia in adults.
What is the History of Gabapentin's Market Exclusivity?
Gabapentin was first marketed by Pfizer under the brand name Neurontin. Key dates regarding its market exclusivity include:
- 1993: FDA approval of Neurontin for epilepsy.
- 2000: FDA approval of Neurontin for postherpetic neuralgia.
- 2004: Expiration of key patents for Neurontin, paving the way for generic entry.
How Has Generic Competition Impacted Gabapentin's Market Share and Pricing?
The expiration of Neurontin's patents led to significant generic competition, fundamentally altering the market landscape:
- Market Share: Generic gabapentin formulations now hold the dominant market share. By 2010, over 90% of gabapentin prescriptions were for generic versions.
- Pricing: The introduction of generics resulted in substantial price erosion. Average wholesale prices (AWP) for gabapentin plummeted by over 80% within two years of the first generic approvals. For example, a 300mg capsule, which might have cost several dollars under brand name, can now be purchased for a fraction of that amount as a generic.
- Manufacturer Landscape: Multiple pharmaceutical companies, including Teva Pharmaceuticals, Mylan (now Viatris), and Sandoz, are major producers of generic gabapentin. This fragmented manufacturing base contributes to competitive pricing.
What are the Key Market Drivers for Gabapentin?
Despite being off-patent for nearly two decades, several factors continue to drive gabapentin's market demand:
- Off-Label Prescribing: A significant portion of gabapentin prescriptions are for off-label uses, including:
- Anxiety disorders
- Insomnia
- Restless legs syndrome
- Diabetic neuropathy (though gabapentin's efficacy in this specific indication has been debated and is often overshadowed by newer agents or guidelines)
- Fibromyalgia
- Migraine prophylaxis
- Aging Population: The increasing prevalence of age-related conditions, such as neuropathic pain and seizure disorders, supports continued demand.
- Cost-Effectiveness: As a generic drug, gabapentin remains a cost-effective option for many patients and healthcare systems compared to newer, branded therapies for some conditions.
- Established Treatment Paradigm: For certain conditions, gabapentin is an established part of treatment algorithms, making it a frequently prescribed option.
What are the Challenges and Restraints Affecting Gabapentin's Market Growth?
Several challenges impact gabapentin's market trajectory:
- Stricter Prescribing Guidelines: Regulatory bodies and healthcare organizations are increasingly scrutinizing off-label use of gabapentin, particularly due to concerns about its abuse potential and side effects. This can lead to:
- Controlled Substance Scheduling: Some jurisdictions have begun classifying gabapentin as a controlled substance or requiring monitoring, impacting prescribing habits. For instance, as of August 2022, 18 U.S. states had added gabapentin to their prescription drug monitoring programs (PDMPs).
- Evidence-Based Prescribing: A greater emphasis on evidence-based medicine encourages the use of drugs with robust clinical trial data for specific indications, potentially limiting gabapentin's use in off-label areas where evidence is weaker.
- Abuse and Diversion Potential: Gabapentin has demonstrated potential for abuse, particularly when combined with opioids. This has led to increased awareness and efforts to curb non-medical use.
- Emergence of Newer Therapies: For specific indications, newer drugs with more targeted mechanisms of action or improved safety profiles have been developed, offering alternatives to gabapentin.
- Side Effect Profile: Common side effects include dizziness, somnolence, ataxia, and peripheral edema, which can limit its use in certain patient populations.
What is the Global Market Size and Financial Trajectory of Gabapentin?
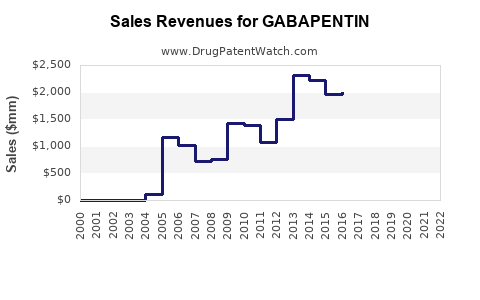
The global gabapentin market, primarily driven by generic sales, is substantial and shows a trajectory characterized by steady demand rather than rapid growth.
- Market Size: While precise, up-to-the-minute market size figures fluctuate based on reporting methodologies, the global gabapentin market is estimated to be in the hundreds of millions to low billions of U.S. dollars annually. For example, estimates from various market research firms place the market between $500 million and $1.5 billion USD in recent years.
- Sales Trends: Sales revenue for gabapentin has stabilized following the initial sharp decline after patent expiry. The revenue is largely generated by generic manufacturers. Brand sales (Neurontin) represent a minimal fraction of the overall market.
- Volume vs. Value: The market is characterized by high prescription volumes due to its broad use. However, the low price per unit of generic gabapentin means that the market's value is constrained compared to branded or newer specialty drugs.
- Geographic Distribution: North America and Europe represent the largest markets, driven by high healthcare spending and established prescribing patterns. Asia-Pacific is a growing market due to increasing access to healthcare and rising prevalence of neurological disorders.
Gabapentin Market Drivers and Restraints Summary
| Category |
Key Factors |
| Drivers |
Off-label prescribing (anxiety, insomnia, fibromyalgia), aging population, cost-effectiveness, established treatment use |
| Restraints |
Stricter prescribing guidelines, abuse potential, emergence of newer therapies, side effect profile |
What are the Key Companies Involved in Gabapentin Manufacturing and Sales?
The gabapentin market is dominated by generic manufacturers. Key players include:
- Teva Pharmaceutical Industries Ltd.: A leading global generic pharmaceutical company.
- Viatris Inc. (formerly Mylan N.V.): Another major producer of generic pharmaceuticals.
- Sun Pharmaceutical Industries Ltd.: An Indian multinational pharmaceutical company.
- Aurobindo Pharma Limited: A global pharmaceutical company.
- Hikma Pharmaceuticals PLC: A multinational pharmaceutical company.
These companies compete on price, supply chain reliability, and manufacturing capacity.
What is the Future Outlook for Gabapentin?
The future outlook for gabapentin is one of continued steady demand, tempered by regulatory scrutiny and evolving clinical practices.
- Sustained Off-Label Use: While some off-label uses may decline due to newer alternatives or stricter guidelines, other indications like anxiety and fibromyalgia are likely to sustain demand.
- Increased Regulatory Oversight: Expect continued efforts to monitor and potentially restrict the prescribing of gabapentin, especially in combination with other substances or for indications with weaker evidence.
- Market Competition: The generic market will remain highly competitive, with pricing pressures likely to persist. Profitability for manufacturers will depend on efficient production and large-scale distribution.
- Niche Applications: Gabapentin may continue to find utility in specific patient populations or as an add-on therapy where its established safety and efficacy profile, combined with low cost, remains advantageous.
Key Takeaways
Gabapentin's market is mature, characterized by widespread generic availability and significant price erosion since patent expiry. Off-label prescribing is a primary driver of sustained demand, although this is increasingly balanced by regulatory scrutiny and concerns over abuse potential. The market is highly competitive among generic manufacturers, with ongoing price pressures and a focus on high-volume production. Future growth will be modest, shaped by evolving clinical guidelines and the ongoing evaluation of its risk-benefit profile across various indications.
Frequently Asked Questions
- Has gabapentin been repurposed for new indications recently?
Recent market activity has focused more on the monitoring and regulation of existing uses, rather than significant new approved indications for gabapentin. Efforts have been made to clarify its role in pain management and neurological disorders based on updated clinical evidence.
- What is the typical prescription volume for gabapentin globally?
Global prescription volumes for gabapentin are in the tens of millions annually, reflecting its broad use in both approved and off-label indications. Precise figures vary but indicate substantial patient access.
- Are there any specific patent challenges or litigation related to gabapentin that are currently active?
Given that the primary patents for Neurontin expired over a decade ago, there are no major active patent litigations or challenges concerning its core composition of matter or initial uses. Disputes may arise regarding manufacturing processes or specific formulations, but these are distinct from the foundational exclusivity.
- How do gabapentin's sales compare to other antiepileptic drugs (AEDs)?
Gabapentin's total sales revenue is lower than many newer, branded AEDs due to its generic status and low price. However, in terms of prescription volume, it remains one of the most frequently prescribed AEDs, often due to its utility in off-label pain management.
- What is the estimated market share of brand-name Neurontin compared to generic gabapentin?
The market share of brand-name Neurontin is negligible, estimated to be well below 1% of the total gabapentin market. Generic versions dominate prescription volume and sales.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Prescription Drug Information. Retrieved from [FDA Website (general reference for drug approvals)]
[2] Pfizer Inc. (various years). Annual Reports and SEC Filings. (Internal company data not publicly accessible for specific year-over-year Neurontin sales after generic entry).
[3] Various Market Research Reports on the Global Gabapentin Market (e.g., Reports by Grand View Research, Mordor Intelligence, etc.). (Specific dates and report titles vary).
[4] National Institute on Drug Abuse. (2020). Gabapentin. DrugFacts.
[5] Prescription Drug Monitoring Programs. (various states and dates of inclusion). Information accessed through state health department websites and legislative records.