Last updated: April 23, 2026
What is the market footing for divalproex sodium?
Divalproex sodium is an established oral anticonvulsant and mood-stabilizing agent used for conditions that include epilepsy and bipolar disorder. Commercial dynamics are shaped by (1) long treatment duration, (2) strong generic penetration, and (3) pricing pressure typical of mature, off-patent small-molecule products.
Commercial positioning (practical reality)
- Generic-dominated market structure: Divalproex sodium is marketed as divalproex sodium delayed-release tablets and divalproex sodium extended-release tablets, with multiple generic entrants. In mature therapeutic classes, generic substitution drives volume share but compresses net prices.
- Formulation split matters: Extended-release products often carry different pricing and payer preferences versus delayed-release, influencing net revenue mix even when total molecule demand is stable.
Formulation identifiers used in commerce
- Delayed-release: Divalproex sodium delayed-release tablets.
- Extended-release: Divalproex sodium extended-release tablets.
How do patent and exclusivity status shape pricing power?
Divalproex sodium is not a new chemical entity. Its market pricing and profit pool are constrained by the absence of meaningful, ongoing exclusivity for the branded molecule in most major markets, with generics capturing share.
Exclusivity constraint profile
- Generic erosion: Once main patent protection ended, manufacturers shifted to abbreviated new drug applications (ANDA) and marketed generics at substantial discounts.
- Reformulation does not restart a broad monopoly: Any incremental protections typically do not prevent generic competition on the active moiety at scale, limiting long-lived brand premium.
What market dynamics drive demand for divalproex sodium?
Demand is less about “new patient starts” and more about continuation therapy and substitution patterns.
Key demand drivers
- Chronic use profile: Many patients remain on valproate-based therapy for long durations, supporting base demand even in the face of generic pricing pressure.
- Payer controls and step therapy: Formularies and therapeutic substitutions influence net unit realizations. When generics are preferred, switching reduces brand-like pricing.
- Safety and tolerability management: Dosing schedules (delayed vs extended release) and monitoring practices influence real-world adherence, which in turn impacts persistence and refill volumes.
Key competitive dynamics
- Interchange and switching: When generics have equivalent FDA-rated performance, pharmacy-level switching is common, pressuring realized price.
- Inventory cycles and supply stability: Mature products experience periodic volume swings tied to procurement behavior and generic supply adjustments.
How has financial performance typically evolved in mature divalproex markets?
For a mature, generic-heavy CNS small molecule, the financial trajectory typically follows a sequence:
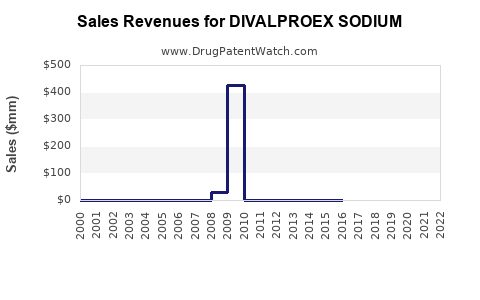
- Early growth (pre-generic): Brand-led revenues with premium pricing.
- Rapid revenue compression (post-generic launch): Unit volumes may remain stable while price per unit drops materially.
- Platform stabilization: Revenues become more dependent on total prescription volume, inventory demand timing, and product mix (delayed vs extended release).
- Periodic share movements: Shifts among generic manufacturers can alter revenue distribution without changing the underlying total category demand.
This trajectory aligns with how mature oral anticonvulsants behave across large markets, including the recurring pattern of lower net pricing with steady prescriptions.
What is the category impact on investor and business decisions?
The molecule’s economics favor strategies that improve execution in a price-compressed environment.
Business levers that matter most
- Cost position and manufacturing capacity: Gross margin is determined by production economics and supply stability.
- Product mix: Extended-release vs delayed-release mix can improve revenue stability when one format maintains relatively stronger payer preferences.
- Contract performance: Bulk and wholesaler contracting drives realized pricing more than list price.
Where does the financial story concentrate across the value chain?
Manufacturer economics
- Revenue depends on prescription volume and net price, with net price heavily influenced by generic competition intensity.
- Margin is driven by manufacturing cost, regulatory compliance overhead, and channel contracting.
Distributor and payer economics
- Payer and PBM contracting uses competitive tenders among generics, typically causing a race to the bottom on net unit pricing.
- Reimbursement policies influence which labeled formulation wins at the point of dispensing.
What regulatory and listing mechanisms shape competitive behavior?
Market participants rely on FDA and payer infrastructure to sustain throughput and maintain inventory reliability.
FDA system participation
- NDA/ANDA framework: Generic competition occurs through ANDAs for delayed-release and extended-release versions.
- Orange Book driven substitution: Bioequivalence and listed products influence pharmacy substitution and payer coverage decisions.
Is there a measurable “molecule vs product” divergence in economics?
Yes. Even when total demand is molecule-stable, revenues can diverge by:
- Release profile (extended vs delayed)
- Dosage strength distribution
- Package size and channel contracts
- Manufacturer market share changes after pricing renegotiations
For divalproex sodium, the extended-release segment can carry different payer and patient preference patterns, which can affect net revenue per script even as the molecule remains generic.
What scenario map best fits divalproex sodium’s financial trajectory?
Given the mature and competitive nature of the market, the most actionable scenario map is built around competitive pricing rather than innovation cycles.
Base case (most consistent with mature generic dynamics)
- Stable or slow-growth prescription volume
- Continued price compression
- Periodic share reallocation among generic suppliers
Downside case
- Intensified generic competition (more entrants or aggressive contracting)
- Supply disruptions leading to short-term price spikes then normalization
Upside case
- Favorable payer contracting for specific manufacturers
- Better-than-category persistence due to mix improvements (extended-release gains)
What are the practical implications for R&D and pipeline strategy?
For businesses evaluating divalproex-related opportunities, the molecule’s market structure implies:
- New entrants need either clinical differentiation that meaningfully changes prescribing behavior or execution differentiation that improves market share and net pricing without relying on brand-style premium.
- Development that targets the existing generic equivalent without a clear clinical or access advantage typically faces limited willingness-to-pay.
Key Takeaways
- Divalproex sodium is a mature, generic-dominated CNS product where pricing pressure outweighs innovation-led upside.
- Financial trajectory follows a standard pattern: post-generic revenue compression, then stabilization driven by volume, mix, and contracting.
- Competitive advantage concentrates in manufacturing cost, supply reliability, and product mix (extended vs delayed) rather than new exclusivity.
- Business value in this category comes from execution in a contract-driven market, not from long-duration brand-style pricing.
FAQs
How does generic competition typically change divalproex sodium revenues?
It lowers net price per prescription while total volumes often remain supported by chronic use. Revenue outcomes then depend on market share and formulation mix (extended vs delayed).
Does extended-release divalproex change the financial profile?
Yes. Extended-release can have different payer preferences and prescribing patterns, which can shift net revenue per unit even if molecule demand is stable.
What is the main determinant of margins for generic divalproex suppliers?
Manufacturing economics and channel contracting, including the ability to maintain supply continuity and compete on net pricing.
What market events most often move divalproex category performance in the short term?
Wholesale and payer contract renegotiations, shifts in generic manufacturer share, and any temporary supply constraints.
Is divalproex sodium an innovation-led market?
Not in the way patented, brand-differentiated therapies are. Value creation is primarily operational or access-driven unless a product introduces clear clinical or formulary-impacting differentiation.
References
[1] FDA. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm