Last updated: February 19, 2026
Desonide, a topical corticosteroid, has maintained a consistent presence in the dermatological market. Its market trajectory is shaped by established therapeutic use, generic competition, and evolving regulatory and reimbursement landscapes. This analysis examines key market drivers, competitive positioning, and financial performance indicators for desonide.
What is the Current Market Size and Growth Projection for Desonide?
The global market for desonide is primarily driven by its established efficacy in treating inflammatory skin conditions like eczema, psoriasis, and dermatitis. While precise, real-time market size figures for individual topical corticosteroids can be proprietary, industry analysis indicates a stable, albeit modest, growth rate for the overall topical corticosteroid market. This segment is projected to grow at a compound annual growth rate (CAGR) of approximately 3% to 5% over the next five years, driven by increasing skin disease prevalence and an aging global population [1]. Desonide's share within this market is sustained by its favorable safety profile for various potencies and its availability in multiple dosage forms (creams, ointments, lotions, gels).
Who are the Key Manufacturers and Competitors in the Desonide Market?
The desonide market is characterized by a significant number of generic manufacturers, reflecting the drug's patent expiration and widespread availability. Major players include:
- Anacor Pharmaceuticals (now Pfizer): While Anacor developed crisaborole (Eucrisa), a non-steroidal anti-inflammatory, its acquisition by Pfizer places it within the broader eczema treatment landscape, indirectly impacting desonide market share through alternative treatment options.
- Galderma: A significant player in dermatology, Galderma offers a range of topical treatments, including generic desonide formulations.
- Bausch Health Companies: This company manufactures and markets various dermatological products, including generic desonide.
- Sun Pharmaceutical Industries: A global pharmaceutical giant, Sun Pharma is a key producer of generic desonide.
- Various Smaller Generic Manufacturers: Numerous smaller companies globally produce and distribute desonide, contributing to competitive pricing.
Competition in the desonide market is largely based on pricing, distribution channels, and product differentiation within generic formulations (e.g., specific excipients, concentrations). The landscape is intensely competitive, with pricing pressures common due to the high number of generic suppliers [2].
What are the Primary Therapeutic Indications and Efficacy of Desonide?
Desonide is a mid-potency topical corticosteroid indicated for the relief of inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. Its efficacy stems from its anti-inflammatory, antipruritic, and vasoconstrictive properties. Key indications include:
- Atopic Dermatitis (Eczema): Desonide effectively reduces redness, itching, and inflammation associated with eczema [3].
- Psoriasis: It provides relief from the plaques and inflammation of mild to moderate psoriasis.
- Contact Dermatitis: Used to manage allergic and irritant contact dermatitis.
- Seborrheic Dermatitis: Helps control inflammation and scaling in seborrheic dermatitis.
- Other Corticosteroid-Responsive Dermatoses: Applicable to a range of inflammatory skin conditions responsive to topical steroids.
Desonide is available in several formulations, including 0.05% cream, ointment, lotion, and gel. The choice of formulation often depends on the affected skin area and the nature of the dermatosis (e.g., ointments are more occlusive and potent for dry, scaly lesions; lotions are lighter for hairy areas) [4].
What is the Patent Landscape and Generic Entry Impact for Desonide?
Desonide's original patents have long expired, leading to a robust generic market. The drug was first approved by the U.S. Food and Drug Administration (FDA) in the 1970s. The lack of ongoing patent protection means that the market is open to generic competition, which significantly impacts pricing and profit margins for manufacturers.
The genericization of desonide has resulted in:
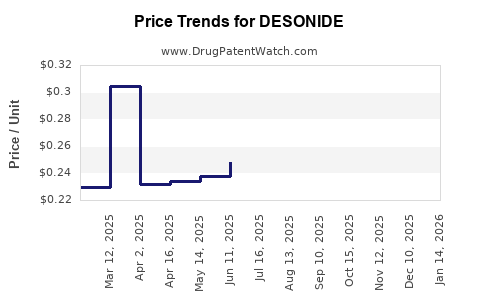
- Price Erosion: Competition among multiple generic manufacturers drives down the average selling price of desonide products.
- Increased Accessibility: Lower prices make desonide more affordable and accessible to a broader patient population and healthcare systems.
- Market Saturation: The market is saturated with various generic brands, requiring manufacturers to compete on cost and distribution.
While there are no active composition-of-matter patents for the original desonide molecule, companies may hold patents related to specific formulations, delivery systems, or manufacturing processes. However, these are generally less impactful than primary drug patents [5].
What are the Regulatory and Reimbursement Considerations for Desonide?
Desonide is regulated by health authorities such as the FDA in the United States and the European Medicines Agency (EMA) in Europe. As a well-established generic drug, its regulatory pathways primarily involve post-market surveillance and adherence to Good Manufacturing Practices (GMP).
Reimbursement for desonide varies by payer and geography.
- United States: Desonide is generally covered by most private health insurance plans and government programs like Medicare and Medicaid when deemed medically necessary. Co-pays and deductibles apply. The Centers for Medicare & Medicaid Services (CMS) has established reimbursement rates for topical corticosteroids, with generic desonide typically falling within established drug pricing frameworks.
- Europe: National health services and private insurers cover desonide, with pricing and reimbursement decisions often made at the country level.
The Affordable Care Act (ACA) in the U.S. and similar healthcare reforms in other regions have aimed to increase insurance coverage, potentially expanding access to treatments like desonide. However, formulary restrictions and prior authorization requirements can still influence prescribing patterns and access [6].
What are the Key Market Drivers and Challenges for Desonide?
Market Drivers:
- Prevalence of Skin Diseases: The increasing incidence of dermatological conditions such as eczema, psoriasis, and dermatitis globally directly fuels demand for effective topical treatments like desonide.
- Established Efficacy and Safety: Decades of clinical use have established desonide as a reliable and generally safe option for managing inflammatory skin conditions, particularly for mild to moderate cases.
- Cost-Effectiveness: As a generic medication, desonide offers a cost-effective alternative to newer, branded therapies, making it a preferred choice in price-sensitive markets and healthcare systems.
- Availability of Multiple Formulations: The availability of desonide in creams, ointments, lotions, and gels allows for tailored treatment based on skin type and lesion location, enhancing its utility.
- Aging Population: Older individuals are more prone to various skin conditions, contributing to sustained demand for dermatological treatments.
Market Challenges:
- Intense Generic Competition: The presence of numerous generic manufacturers leads to significant price erosion and limited opportunities for substantial profit growth.
- Emergence of Novel Therapies: Development of new, non-steroidal anti-inflammatory agents and biologics for dermatological conditions can divert market share from traditional corticosteroids. Examples include crisaborole (Eucrisa) and biologics for psoriasis.
- Concerns Regarding Long-Term Corticosteroid Use: While desonide is considered mid-potency, potential side effects associated with prolonged or inappropriate use of corticosteroids (e.g., skin thinning, striae, potential systemic absorption) can lead to prescriber caution and a preference for alternative treatments in certain scenarios.
- Reimbursement Pressures and Formulary Restrictions: Healthcare payers may implement restrictions or prefer lower-cost alternatives, impacting market access for desonide.
- Limited Innovation in Generic Formulations: Without strong patent protection, there is less incentive for significant investment in novel formulations or delivery systems for desonide compared to newer, patented drugs.
What is the Financial Trajectory and Outlook for Desonide?
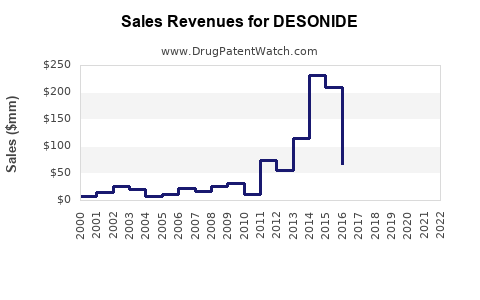
The financial trajectory for desonide is characterized by stable, low-single-digit revenue growth driven by volume. Profitability is heavily influenced by manufacturing efficiency and distribution agreements, rather than novel product development.
- Revenue: Revenue is primarily generated through sales volumes in the generic market. While overall prescription volumes for topical corticosteroids remain high, the price per unit is low due to competition.
- Profit Margins: Margins are typically thin in the generic drug sector. Manufacturers focus on optimizing production costs and supply chain management to maintain profitability.
- Investment Focus: Investment in desonide is primarily focused on manufacturing capacity, quality control, and market access through distribution networks, rather than R&D for new indications or formulations.
The outlook for desonide is one of continued stability. It will remain a foundational treatment for many common dermatological conditions due to its affordability and efficacy. However, significant revenue growth is unlikely. Future market dynamics will be shaped by the overall growth of the dermatology market, the pricing strategies of generic manufacturers, and the continued introduction of novel therapeutic alternatives [7].
Key Takeaways
- Desonide operates within a mature, genericized market characterized by price competition and stable demand.
- Its established efficacy and cost-effectiveness ensure continued relevance in treating common inflammatory dermatoses.
- Market growth is projected at a modest CAGR of 3-5%, mirroring the broader topical corticosteroid segment.
- The competitive landscape is dominated by numerous generic manufacturers, limiting significant profit expansion.
- While regulatory pathways are well-defined, reimbursement strategies and evolving payer preferences can influence market access.
- The emergence of novel therapies presents a challenge to desonide's market share, though its affordability maintains a strong position for first-line or adjunctive therapy.
Frequently Asked Questions
1. What is the difference in potency between desonide and other topical corticosteroids?
Desonide is classified as a mid-potency topical corticosteroid. Other classes range from very low potency (e.g., hydrocortisone 1%) to ultra-high potency (e.g., clobetasol propionate). This categorization influences its suitability for different skin conditions and body areas, with mid-potency agents generally considered appropriate for sensitive areas and less severe dermatoses [8].
2. How does desonide compare in terms of side effects to newer dermatological treatments?
Compared to newer non-steroidal treatments or biologics, desonide, as a corticosteroid, carries a risk of steroid-specific side effects with prolonged or extensive use, including skin atrophy, striae, and telangiectasias. Newer agents often aim to mitigate these specific corticosteroid-related adverse events, although they may have their own distinct safety profiles and efficacy considerations [9].
3. What is the typical duration of treatment for conditions treated with desonide?
Treatment duration varies significantly based on the specific dermatosis, its severity, and the individual patient's response. Typically, topical corticosteroids like desonide are used for the shortest duration necessary to control symptoms. For acute flare-ups, treatment might last a few days to a couple of weeks. Chronic conditions may involve intermittent use or use for a defined period followed by reassessment [10].
4. Are there any specific patient populations for whom desonide is contraindicated or used with caution?
Desonide is generally avoided or used with extreme caution in patients with known hypersensitivity to corticosteroids, untreated bacterial, fungal, or viral skin infections, and in individuals with severe or widespread dermatoses where systemic absorption could be a concern. Caution is also advised in infants and young children due to increased risk of systemic absorption and potential for adverse effects [4].
5. What are the main economic factors driving the price of generic desonide?
The price of generic desonide is primarily driven by manufacturing costs (raw materials, production, quality control), economies of scale achieved by manufacturers, competitive pricing strategies among multiple generic suppliers, and distribution channel markups. The absence of R&D recoupment and marketing expenses associated with branded drugs also contributes to lower generic pricing [2].
Citations
[1] Global Market Insights. (2023). Topical Corticosteroids Market Size, Share & Industry Analysis, By Product Type, By Formulation, By Application, By Distribution Channel, By Region, And Forecast 2023 - 2032.
[2] Grand View Research. (2023). Topical Corticosteroids Market Size, Share & Trends Analysis Report By Product (Low, Medium, High, Ultra-High Potency), By Formulation (Cream, Ointment, Lotion, Gel), By Application (Eczema, Psoriasis, Dermatitis, Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2023 - 2030.
[3] National Eczema Association. (n.d.). Treatments. Retrieved from https://nationaleczema.org/treatment/
[4] U.S. Food & Drug Administration. (n.d.). Prescribing Information: Desonide Cream, Ointment, Lotion, Gel. (Specific manufacturer labeling should be referenced for precise details).
[5] Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. (n.d.). U.S. Food & Drug Administration. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/
[6] Centers for Medicare & Medicaid Services. (n.d.). Medicare Drug Spending. Retrieved from https://www.cms.gov/ (General information on Medicare coverage and drug pricing frameworks).
[7] EvaluatePharma. (Annual Reports and Market Analyses). (Proprietary data on market trends and financial projections for pharmaceutical segments).
[8] Habif, T. F. (2016). Clinical Dermatology: A Color Guide to Diagnosis and Therapy (6th ed.). Elsevier.
[9] Lebwohl, M., & Alexis, A. F. (2019). Emerging Therapies for Atopic Dermatitis. Journal of Allergy and Clinical Immunology: In Practice, 7(6), 1798-1809.
[10] American Academy of Dermatology Association. (n.d.). Common Skin Conditions. Retrieved from https://www.aad.org/public/diseases/skin-conditions