Last updated: April 24, 2026
Cyclosporine is a mature immunosuppressant with a long commercial history across transplant medicine and a smaller set of chronic autoimmune indications. Market dynamics are dominated by (1) patent and life-cycle events across branded and generic formulations, (2) oral formulation substitution (including improved-release formats), (3) channel and reimbursement behavior in transplant centers, and (4) price erosion in geographies where generics hold share.
Cyclosporine’s financial trajectory reflects a late-stage molecule with ongoing volume demand but diminishing net pricing power in many markets. The main upside levers are formulation durability (brand switches tied to tolerability and adherence), new patient capture within existing indications, and value-prescribing behavior where payers reward lower dosing variability or fewer monitoring events.
How does the cyclosporine market size behave across indications?
Cyclosporine is used most heavily in transplant care:
- Solid organ transplant (SOT): kidney, liver, heart (with center-specific protocols)
- Prevention of graft rejection after transplantation
- Adjunct immunosuppression in combination regimens
Outside classic transplantation, cyclosporine has significant but smaller chronic use:
- Dermatology (notably atopic dermatitis in certain populations; indication access varies by formulation and jurisdiction)
- Ocular surface disease in select markets (availability depends on formulation and regulatory approvals)
The market behaves like a “chronic procedural” segment: volumes track transplant volumes and maintenance immunosuppression adherence, not acute episodic cycles. That creates relatively stable baseline demand even as prices compress.
What are the main demand drivers and constraints?
Demand drivers
- Transplant volume and survival improvements: higher survival increases long-run prevalence of patients maintained on immunosuppression.
- Protocol durability: many transplant regimens are conservative and slow to switch when new options do not materially improve outcomes.
- Formulation tolerance: oral cyclosporine formulations differ in absorption profiles and food interaction; clinicians and patients often adopt a regimen that reduces variability.
Constraints
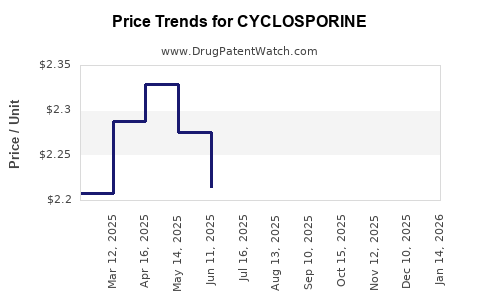
- Price pressure from generics: cyclosporine is off-patent in most major markets; generic competition typically drives margin compression.
- Narrow therapeutic index and monitoring: stability requirements can slow switching between products in practice.
- Safety and drug-drug interactions: the product is dependent on therapeutic drug monitoring (TDM) and interacts with CYP3A and transporters; this creates friction for rapid substitution.
- Payer preference for lower unit cost: formularies and reimbursement frequently push toward least-cost options once switching risk is acceptable.
How do formulation and life-cycle dynamics shape competitive outcomes?
Cyclosporine has multiple commercially important oral formats, and the switch dynamics differ by therapeutic setting:
The financial consequence is a two-speed market:
- Volume resilience for the total cyclosporine class in transplant.
- Margin decline at the product level as generic penetration expands.
Where does cyclosporine sit in the broader immunosuppressant landscape?
Cyclosporine competes against:
- Calcineurin inhibitors (CNIs): tacrolimus is typically the closest comparator for many transplant indications.
- Other immunosuppressants in combination regimens: mycophenolate, mTOR inhibitors, corticosteroids, and biologics in selected settings.
This matters financially because:
- Protocol choices affect net share. Even if cyclosporine remains clinically relevant, centers that shift to tacrolimus can reduce cyclosporine’s addressable share.
- Combination regimen economics: payer dynamics can steer toward lower-cost CNI options, impacting net revenue for cyclosporine even when overall transplant volume grows.
What does the financial trajectory look like: pricing vs. unit growth?
Cyclosporine’s commercial math usually splits into:
- Units (stable to mildly growing) driven by transplant prevalence and chronic use.
- Net sales (flat to declining) driven by pricing compression from generics and by competitive CNI substitution.
The shape of net sales usually follows a pattern common to mature off-patent specialty drugs:
- After major branded periods, net price declines accelerate with generic penetration.
- Share shifts can occur around formulation preference, but over time economic substitution dominates.
For investors and R&D planners, this means:
- Growth is harder to generate by incremental clinical benefits unless they unlock payer-relevant advantages (monitoring burden reduction, adherence, or demonstrable outcome improvements).
- Return profiles often depend on protecting formulation share rather than expecting molecular novelty.
How do market channels and reimbursement influence revenue?
Cyclosporine distribution is typically characterized by:
- Hospital and transplant center purchasing for initiation regimens
- Long-term maintenance dispensing through specialty pharmacy networks or hospital outpatient programs
Reimbursement behavior often results in:
- Formulary tiering that favors least-cost equivalents once clinical interchangeability is accepted.
- Prior authorization and step therapy in some markets for branded modified-release formulations.
- Therapeutic monitoring requirements that can increase administrative burden, but also create a place for brand loyalty if centers standardize monitoring workflows.
Net effect:
- Branded products can retain share where switching costs (monitoring and stability) outweigh pricing differences.
- Generics tend to win once health systems harmonize monitoring and adopt interchangeability protocols.
What are the likely swing factors for cyclosporine net sales going forward?
Key variables that can move cyclosporine’s revenue:
- Generic competition intensity in specific jurisdictions (how many competitors enter and when)
- Modified-release uptake (conversion rate from immediate-release and generic equivalence)
- Transplant volume trends by region
- CNI protocol shifts between cyclosporine and tacrolimus
- Safety monitoring practices that affect interchangeability
Which products and jurisdictions matter most to the financial trajectory?
Cyclosporine is a global molecule, but financial outcomes depend heavily on:
- Patent and formulation life-cycle per region
- Generic adoption speed
- Regulatory approvals for specific formulations in dermatology/ocular indications
- Tendering and hospital procurement frameworks in Europe and parts of Asia
Because cyclosporine is mature and widely generic, regional differences in pricing and formulary access often produce the biggest observable divergence in revenue trend.
What is the overall investment and R&D implication for cyclosporine?
Given the mature molecule status, the commercially rational R&D posture tends to focus on:
- Formulation differentiation that changes real-world exposure variability or adherence
- Patient-relevant safety outcomes linked to therapeutic monitoring practices
- Indication expansion only where regulatory pathways and market access support premium pricing
For existing cyclosporine suppliers:
- Defending share is more feasible through formulation continuity and clinical workflow integration than through breakthrough claims.
- Revenue upside is mainly a function of retaining higher-margin branded or authorized products against generic substitution.
Key financial timeline signals to track (business checklist)
These are the measurable events that typically drive cyclosporine’s financial trajectory:
- Generic entry announcements for each key formulation and market
- Formulary changes by major payers and hospital groups
- Tender outcomes for transplant pharmacy supply contracts
- Switch guidelines for therapeutic drug monitoring and dose conversion
- Regulatory updates for dermatology/ocular indications by formulation
A consistent pattern emerges: each generic entry resets the pricing floor; uptake of modified-release formats can slow decline but usually does not stop it in fully generic markets.
Key Takeaways
- Cyclosporine demand is structurally stable due to transplant maintenance and chronic immunosuppression, but financial performance is dominated by pricing erosion once generics penetrate.
- Market dynamics hinge on formulation substitution, therapeutic drug monitoring friction, and protocol-level CNI selection (notably competition with tacrolimus).
- Cyclosporine net sales typically show unit resilience with margin compression, with regional differences driven by generic adoption speed and formulary behavior.
- The highest-leverage commercial defenses are formulation durability and real-world workflow fit, not new molecular differentiation.
- The most important forward-looking signals are generic entry timing, hospital tender outcomes, and payer formulary tiering for branded versus authorized generics.
FAQs
-
Is cyclosporine’s market growth driven by new patients or by prevalence?
It is primarily driven by prevalence in transplant maintenance rather than rapid new patient creation in most mature markets.
-
Why does therapeutic drug monitoring slow product switching for cyclosporine?
Switching can require dose recalibration and tighter monitoring to maintain exposure within the therapeutic range.
-
What formulation factors most affect competitive positioning?
Oral absorption profile consistency, dosing convenience, food interaction behavior, and center-level adherence practices.
-
How does cyclosporine compete with tacrolimus financially?
Financially, centers and payers decide CNI choice based on protocol outcomes and total regimen economics, with pricing and formulary tiering often shifting share.
-
What are the most common events that rebase cyclosporine revenue?
Generic entry waves, authorized generic launches, and formulary or tender contract renewals that shift supply to least-cost options.
References
[1] European Medicines Agency. Sandimmun (cyclosporin) and related products: product information and assessment materials. (Accessed via EMA product pages). https://www.ema.europa.eu/
[2] U.S. Food and Drug Administration. Prescribing information and drug label history for cyclosporine products (as applicable by brand and formulation). https://www.accessdata.fda.gov/
[3] NICE (UK). Immunosuppression and transplant medicine guidance reflecting CNI usage context. https://www.nice.org.uk/
[4] American Society of Transplantation (AST) and transplant clinical resources on CNI regimens and monitoring practices. https://www.myast.org/