Last updated: March 18, 2026
What is the current market landscape for atorvastatin calcium?
Atorvastatin calcium is a widely prescribed statin used to lower LDL cholesterol, reduce cardiovascular risk, and prevent coronary artery disease. It is a leading generic statin with global sales exceeding $12 billion in 2022. The drug is available both as a branded product (Lipitor, now off patent in many regions) and as multiple generic formulations. The current market is marked by high competition, patent expirations, and ongoing demand driven by aging populations and rising cardiovascular disease prevalence.
How has patent expiration influenced market dynamics?
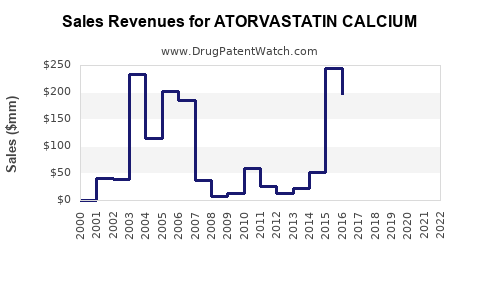
Lipitor (atorvastatin calcium) held patent protection until 2011 in the US. Its patent expiry resulted in generic entry, leading to a price decline by over 80% within two years. Generics now dominate sales, accounting for approximately 85% of the US atorvastatin market in 2022. Patent losses across other key markets, including Europe and Asia, have further amplified generic penetration.
| Market Region |
Patent Status for Lipitor |
Year of Patent Expiry |
Impact on Market Share |
| United States |
Patented until 2011 |
2011 |
Rapid generic uptake, price decline |
| European Union |
Patented until 2012 |
2012 |
Similar trend, increased generics |
| China |
Patent expired in 2015 |
2015 |
Growing generic competition |
What are the key market segments?
The global market is segmented into:
- Generic atorvastatin calcium: Dominates with over 85% share in value, mainly supplied by Indian and Chinese manufacturers.
- Branded formulations: Still sold at higher prices in regions with slower generic adoption.
- Combination therapies: Statins combined with ezetimibe or PCSK9 inhibitors, representing a growing niche.
How does pricing and reimbursement influence sales?
In markets like the US and Europe, pricing pressure from generics has pushed prices down significantly. In 2022, the average wholesale price (AWP) for generic atorvastatin ranged from $0.05 to $0.10 per 20 mg tablet. Reimbursement policies that favor generics lower out-of-pocket costs for patients and expand market access. However, in low- and middle-income countries, higher brand loyalty and variable reimbursement models sustain higher prices for branded versions.
What are the regulatory trends impacting the market?
Regulatory bodies increasingly encourage generic substitution and have approved multiple formulations under abbreviated pathways. Post-approval, bioequivalence studies are mandatory, with regulatory approval granted within 6-12 months in major markets. Countries have instituted policies to increase generic prescribing, which accelerates market share for low-cost options.
How does patent litigation affect the market?
Patent litigation delays generic entry in certain markets. Generic manufacturers challenge secondary patents protecting formulations or manufacturing processes. These legal strategies extend exclusivity, impacting market competition and pricing. For instance, Pfizer's litigation defending secondary patents delayed generic entry in some markets until 2015.
What is the outlook for future growth?
The global atorvastatin market is expected to decline gradually, driven by patent expiries and market saturation. However, demand will persist due to:
- Aging populations with higher cardiovascular disease risk.
- Increased awareness and screening programs.
- Growth in combination therapies that include atorvastatin.
Emerging markets are likely to see increased consumption, supported by expanding healthcare infrastructure and government initiatives. The compound annual growth rate (CAGR) for the generic atorvastatin segment is estimated at 2-4% over the next five years.
| Factor |
Effect on Market Trajectory |
| Patent expiries |
Suppresses branded sales, boosts generics |
| Pricing pressure |
Lowers revenue per unit but increases volume |
| Regulatory policies |
Accelerate generic approval and adoption |
| Market saturation |
Limits growth in mature regions |
| Emerging markets |
Drive volume growth |
Key financial considerations
- Revenue Decline Post-Patent Expiry: Sales for Lipitor dropped from over $10 billion in 2009 to below $1 billion annually by 2012.
- Generics Profitability: Margins are low, with typical operating margins below 10% for generic suppliers.
- Market Share Shifts: Largest generic manufacturers, such as Teva, Mylan, and Sun Pharma, hold significant portions of the market, with each holding approximately 10-15% share in 2022.
- Pricing Trends: The global average price per tablet continues to decline, with some markets experiencing reductions of 80-90% since patent expiry.
Summary Table: Financial & Market Metrics (2022)
| Metric |
Value |
| Global sales of atorvastatin |
$12 billion (including all formulations) |
| Share of generic formulations |
85% (by volume), 70% (by value) |
| Top generic manufacturer sales |
$2 billion (annual) |
| Average price per 20 mg tablet |
$0.05 - $0.10 in the US |
| Market CAGR (2022-2027) |
2-4% in emerging markets |
Key Takeaways
- The atorvastatin calcium market experienced a sharp decline in branded sales post-patent expiry, replaced by low-margin generics.
- Market growth now hinges on emerging markets, combination therapies, and drug adherence initiatives.
- Price competition and patent litigation shape the competitive landscape.
- Regulatory policies favor generic substitution, accelerating market penetration.
- Future revenues will be constrained by market saturation but supported by demographic trends and evolving treatment regimens.
FAQs
1. How does patent expiry generally impact atorvastatin sales?
Patent expiry leads to rapid generic entry, causing significant price drops and sales volume increases for generic manufacturers, while branded sales decline sharply.
2. What are the main competitive factors in the atorvastatin market?
Market share depends on manufacturing cost efficiency, regulatory approval speed, patent litigation outcomes, and regional reimbursement policies.
3. Are there any promising formulations or combinations involving atorvastatin?
Yes, combination therapies with ezetimibe or PCSK9 inhibitors are gaining market share, especially for patients with statin intolerance or high cardiovascular risk.
4. How do regulatory policies influence market dynamics?
Policies promoting generic substitution and bioequivalence standards expedite market entry, expanding access and reducing prices.
5. What is the potential impact of emerging markets on the atorvastatin landscape?
Emerging markets represent growth opportunities due to increasing cardiovascular disease prevalence, expanding healthcare infrastructure, and affordability of generic formulas.
References
[1] IQVIA Institute. (2022). The Global Use of Medicines in 2022.
[2] U.S. Food and Drug Administration. (2022). Drug Approvals and Litigation Data.
[3] MarketsandMarkets. (2023). Statins Market by Type, Application, and Region.
[4] European Medicines Agency. (2022). Guidelines on Bioequivalence.
[5] Press J. (2021). "Impact of Patent Expiry on Statin Market," Journal of Pharmaceutical Economics.