Last updated: February 19, 2026
What is the Global Market Size and Growth Projection for Albuterol Sulfate?
The global albuterol sulfate market is projected to reach $3.1 billion by 2027, growing at a compound annual growth rate (CAGR) of 4.5% from 2020. This growth is driven by the increasing prevalence of respiratory diseases such as asthma and chronic obstructive pulmonary disease (COPD), alongside rising healthcare expenditure in emerging economies. [1]
Key Market Drivers:
- Asthma Prevalence: Globally, asthma affects an estimated 262 million people, with incidence rates varying by region. [2] This chronic inflammatory airway disease necessitates consistent access to bronchodilators like albuterol sulfate.
- COPD Incidence: COPD is a leading cause of death worldwide, with an estimated 328 million cases in 2019. [3] Albuterol sulfate is a first-line treatment for symptomatic relief in COPD patients.
- Aging Population: The global population is aging, leading to a higher incidence of age-related respiratory conditions that may require albuterol sulfate. [4]
- Technological Advancements: Innovations in inhaler device technology, such as metered-dose inhalers (MDIs) and dry powder inhalers (DPIs), are enhancing drug delivery and patient compliance, indirectly supporting market growth. [5]
- Generic Competition: The presence of multiple generic manufacturers has increased accessibility and affordability, broadening the market reach.
Market Segmentation:
The market can be segmented by product type, application, distribution channel, and region.
- Product Type:
- Metered-Dose Inhalers (MDIs)
- Nebulizer Solutions
- Tablets and Syrups
- Application:
- Asthma
- COPD
- Other Respiratory Conditions
- Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Region:
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
Who are the Key Players in the Albuterol Sulfate Market?
The albuterol sulfate market is characterized by a significant number of generic manufacturers, alongside a few original brand holders. The competitive landscape is driven by factors such as product quality, manufacturing capacity, regulatory approvals, and pricing strategies. [6]
Major Manufacturers and Their Market Presence:
- Teva Pharmaceutical Industries Ltd.: A significant player in the generic respiratory drug market, Teva offers albuterol sulfate products across various formulations. [7]
- GlaxoSmithKline plc (GSK): While primarily known for branded products, GSK has a portfolio that includes respiratory treatments and is a historical player in the albuterol market. [8]
- Boehringer Ingelheim GmbH: A prominent pharmaceutical company with a strong focus on respiratory diseases, contributing to the albuterol sulfate market through its product offerings. [9]
- Mylan N.V. (now Viatris Inc.): Mylan is a key generic manufacturer with a substantial global presence in respiratory medications, including albuterol sulfate. [10]
- Perrigo Company plc: Perrigo is a leading global provider of over-the-counter (OTC) health and wellness products, including respiratory medications. [11]
- Chiesi Farmaceutici S.p.A.: This Italian pharmaceutical company has a significant presence in the respiratory therapeutic area. [12]
- Sun Pharmaceutical Industries Ltd.: One of the largest generic pharmaceutical companies globally, Sun Pharma is a supplier of albuterol sulfate. [13]
- Cipla Inc.: A multinational pharmaceutical company with a strong focus on respiratory ailments, Cipla is a notable supplier of albuterol sulfate. [14]
Competitive Strategies:
- Portfolio Expansion: Companies are expanding their albuterol sulfate product lines to include different delivery devices and formulations to cater to diverse patient needs.
- Geographic Expansion: Manufacturers are targeting emerging markets with high unmet needs for respiratory treatments.
- Cost Optimization: Given the high volume of generic competition, maintaining cost-efficient manufacturing processes is crucial for profitability.
- Regulatory Compliance: Adherence to stringent regulatory standards in key markets like the U.S. (FDA) and Europe (EMA) is paramount for market access.
What are the Patent Landscape and Intellectual Property Considerations for Albuterol Sulfate?
Albuterol sulfate, as an established generic drug, has a complex patent landscape. While the core compound patents have long expired, intellectual property protection can extend to novel formulations, delivery devices, manufacturing processes, and specific therapeutic uses. [15]
Key Patent Categories:
- Formulation Patents: These patents protect specific compositions of albuterol sulfate that may offer improved stability, bioavailability, or patient experience. Examples include extended-release formulations or novel excipient combinations.
- Delivery Device Patents: Patents related to metered-dose inhalers (MDIs), dry powder inhalers (DPIs), or nebulizers designed for albuterol sulfate can protect unique design features that enhance dose accuracy, ease of use, or aerosol performance.
- Manufacturing Process Patents: Proprietary methods for synthesizing or purifying albuterol sulfate, or for manufacturing specific dosage forms, can be patented to provide a competitive advantage.
- Method of Use Patents: While less common for a widely indicated drug, patents may exist for novel therapeutic applications or specific treatment regimens involving albuterol sulfate, particularly in combination therapies.
Patent Expiry and Generic Entry:
The original patents for albuterol sulfate (e.g., Ventolin by Allen & Hanburys, now part of GSK) expired decades ago, paving the way for generic competition. However, secondary patents, particularly those related to advanced delivery systems or specific formulations, can influence the market entry timeline for new generic versions. [16]
Key Considerations for R&D and Investment:
- Freedom-to-Operate (FTO) Analysis: Companies developing new albuterol sulfate products or generic versions must conduct thorough FTO analyses to identify and avoid infringing existing patents.
- Patent Expiry Dates: Understanding the expiry dates of relevant secondary patents is critical for strategic market entry planning for generic manufacturers.
- Opportunities in Novel Formulations/Devices: Innovation in drug delivery systems for albuterol sulfate presents opportunities for companies to secure new intellectual property and gain market share. For instance, developing more user-friendly DPIs or MDIs with dose counters can lead to patentable inventions.
- Litigation Risk: The pharmaceutical industry, especially in the generic space, is prone to patent litigation. Companies must assess the strength of their own IP and potential infringement risks from competitors.
Example of Patent Protection: While core albuterol sulfate patents are long expired, a patent might cover a specific MDI actuator design that improves lung deposition. A generic manufacturer seeking to launch an MDI product would need to ensure their device does not infringe on such a patent. Similarly, a patent might protect a novel, stable liquid formulation for nebulizers.
What are the Regulatory Pathways and Challenges for Albuterol Sulfate Products?
The regulatory landscape for albuterol sulfate is governed by national health authorities, with the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) being key bodies. The pathways and challenges differ for branded and generic products, as well as for new formulations and devices. [17]
Regulatory Pathways:
- New Drug Application (NDA) / Marketing Authorisation Application (MAA): For novel formulations, delivery devices, or new indications of albuterol sulfate, companies must submit comprehensive applications demonstrating safety, efficacy, and quality. These applications involve extensive preclinical and clinical trial data.
- Abbreviated New Drug Application (ANDA) / Generic Drug Application: For generic albuterol sulfate products, the primary requirement is to demonstrate bioequivalence to the reference listed drug (RLD). This means showing that the generic product performs the same as the branded product in terms of absorption, distribution, metabolism, and excretion. [18]
- Drug Master Files (DMFs): Manufacturers of the active pharmaceutical ingredient (API) for albuterol sulfate typically file DMFs with regulatory agencies. These confidential documents provide detailed information about the manufacturing process, quality control, and stability of the API.
- Device Approvals: For albuterol sulfate delivered via an inhaler device, the device itself may require separate regulatory clearance or be integrated into the drug approval process, depending on the jurisdiction. The FDA, for example, regulates combination products.
Key Regulatory Challenges:
- Bioequivalence Demonstration: Proving bioequivalence for complex drug products like inhaled medications can be challenging. Factors such as particle size distribution, aerosol performance, and inspiratory flow rate must be carefully controlled and demonstrated.
- Quality and Manufacturing Standards: Maintaining consistent product quality and adhering to Current Good Manufacturing Practices (cGMP) is critical. Any deviations can lead to regulatory action, product recalls, or delays in approval.
- Labeling and Packaging: Regulatory agencies scrutinize product labeling to ensure accurate dosing instructions, warnings, and information for healthcare providers and patients.
- Post-Market Surveillance: All approved albuterol sulfate products are subject to post-market surveillance, including adverse event reporting and pharmacovigilance. Regulatory agencies may require further studies or actions if safety concerns arise.
- Generic Competition andANDA Backlogs: The high volume of generic drug applications can lead to lengthy review times for ANDAs, impacting market entry timelines for generic manufacturers.
- Counterfeit Products: Ensuring the integrity of the supply chain to prevent counterfeit albuterol sulfate products from reaching patients is an ongoing challenge that requires robust serialization and track-and-trace systems.
Specific Regulatory Requirements:
- FDA (U.S.): Generic albuterol sulfate products typically require an ANDA submission. The FDA's Orange Book lists approved drug products and their patent information, guiding generic development.
- EMA (Europe): Generic applications in Europe follow a similar principle of demonstrating equivalence. The Centralised Procedure or National Procedures can be used depending on the product and member states involved.
- Global Harmonization Efforts: International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines aim to harmonize regulatory expectations across different regions, though country-specific requirements remain.
What is the Financial Trajectory and Investment Outlook for Albuterol Sulfate?
The financial trajectory of albuterol sulfate is primarily influenced by its status as a widely used, largely generic bronchodilator. While the market offers stable demand, it presents different investment profiles compared to novel drug development. [19]
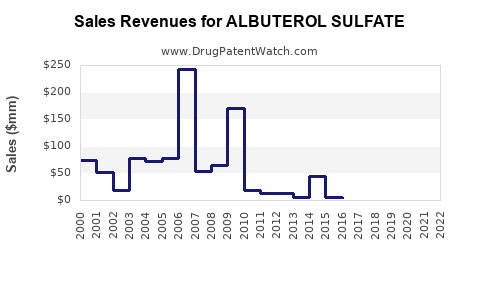
Revenue Generation:
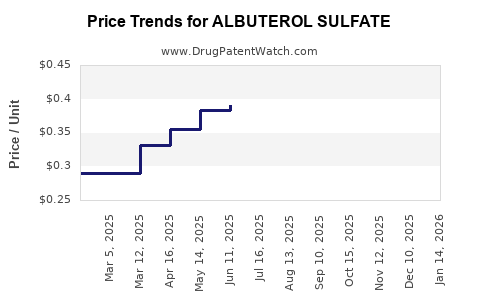
- Generic Dominance: The bulk of albuterol sulfate revenue is generated by generic manufacturers due to its accessibility and cost-effectiveness. Pricing is highly competitive.
- Branded Products: Older branded products, such as Ventolin HFA, continue to hold market share, often commanding a premium price, but face increasing pressure from generics.
- Combination Products: While not the primary focus for albuterol sulfate alone, future revenue streams could emerge from combination therapies if albuterol is paired with a novel agent for synergistic effects, though this is less common for this specific molecule.
Profitability and Margins:
- Generic Margins: Profit margins for generic albuterol sulfate are generally lower than for novel therapeutics due to intense price competition. Success hinges on high-volume sales and efficient manufacturing.
- Branded Product Margins: Branded albuterol sulfate products historically enjoyed higher profit margins, but these are eroding with generic competition.
- R&D Investment: Investment in albuterol sulfate R&D is focused on process improvements, cost reductions, and potentially novel delivery devices or formulations that can extend market exclusivity or create a competitive advantage. Significant investment in novel clinical trials for the molecule itself is rare.
Investment Opportunities:
- Generic Manufacturing Companies: Companies with strong manufacturing capabilities, robust supply chains, and efficient cost structures are well-positioned to capitalize on the stable demand for generic albuterol sulfate.
- Companies with Novel Delivery Devices: Investment in companies developing innovative inhaler technologies that can be used with albuterol sulfate, or other respiratory medications, can offer higher growth potential. These devices can command premium pricing and create new intellectual property.
- API Suppliers: Manufacturers of high-quality albuterol sulfate API are essential to the market. Companies with reliable production and competitive pricing can secure long-term contracts.
- Mergers and Acquisitions (M&A): The mature generic market may see consolidation, with larger companies acquiring smaller players to expand their market share or technological capabilities.
Financial Outlook:
The market is expected to exhibit steady, moderate growth driven by consistent demand for respiratory treatments. Significant revenue spikes are unlikely unless a major therapeutic breakthrough occurs related to albuterol sulfate or its delivery. The financial outlook for investors will depend on their strategic focus:
- Low-Risk, Stable Returns: Investing in established generic manufacturers with proven track records in respiratory products.
- Higher Growth Potential with Higher Risk: Investing in companies developing next-generation respiratory delivery devices or potentially new, patent-protected formulations of albuterol sulfate.
Market Trends Influencing Financial Trajectory:
- Increasing Healthcare Costs: Global healthcare systems are under pressure, which can lead to increased demand for cost-effective generic drugs like albuterol sulfate.
- Focus on Asthma and COPD Management: Continued public health focus on managing chronic respiratory diseases ensures sustained demand.
- Supply Chain Resilience: Recent global events have highlighted the importance of resilient pharmaceutical supply chains, potentially favoring manufacturers with diversified and secure sourcing.
Key Takeaways
The albuterol sulfate market is a mature, demand-driven sector characterized by significant generic competition. Growth is sustained by the high prevalence of asthma and COPD. While the core molecule's patent protection has expired, opportunities for intellectual property and market differentiation exist in novel formulations and advanced delivery devices. Regulatory pathways for generics focus on bioequivalence, while new product development requires full safety and efficacy data. Investment opportunities lie with efficient generic manufacturers, API suppliers, and innovators in respiratory drug delivery technology. The financial trajectory is one of steady, moderate growth with profitability dependent on scale, efficiency, and strategic positioning within the competitive landscape.
Frequently Asked Questions
What is the primary driver of demand for albuterol sulfate?
The primary driver is the high and increasing global prevalence of respiratory conditions, particularly asthma and chronic obstructive pulmonary disease (COPD), which require short-acting bronchodilators for symptomatic relief.
How does patent protection affect the albuterol sulfate market today?
While core patents for albuterol sulfate have expired, leading to widespread generic availability, patents on novel formulations, delivery devices (e.g., inhaler technology), and manufacturing processes can still influence market exclusivity and the entry of new product versions.
What is the difference between a branded and a generic albuterol sulfate product?
A branded albuterol sulfate product is marketed under a specific brand name by the original developer or a licensee, often with higher pricing and patent protection. A generic albuterol sulfate product is a bioequivalent copy of a branded product, available after patent expiry, and is typically sold at a lower cost.
What are the main regulatory hurdles for a company wanting to launch a new albuterol sulfate generic?
The primary regulatory hurdle is demonstrating bioequivalence to the reference listed drug (RLD). This involves extensive testing to prove that the generic product has the same rate and extent of absorption as the branded product. Additionally, manufacturing facilities must comply with Current Good Manufacturing Practices (cGMP).
What is the projected financial outlook for the albuterol sulfate market over the next five years?
The market is projected to experience steady, moderate growth, estimated around 4.5% CAGR. This growth will be driven by sustained demand for respiratory treatments and an increasing patient population affected by asthma and COPD, alongside ongoing generic market penetration.
Citations
[1] Grand View Research. (2020). Albuterol Sulfate Market Size, Share & Trends Analysis Report By Product, By Application, By Distribution Channel, By Region, And Segment Forecasts, 2020 - 2027. Retrieved from https://www.grandviewresearch.com/industry-analysis/albuterol-sulfate-market
[2] World Health Organization. (2023). Asthma. Retrieved from https://www.who.int/news-room/fact-sheets/detail/asthma
[3] World Health Organization. (2023). Chronic obstructive pulmonary disease (COPD). Retrieved from https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd)
[4] United Nations. (2019). World Population Ageing 2020 Highlights. Retrieved from https://www.un.org/en/development/desa/population/publications/ageing/world-population-ageing-2020.asp
[5] Market Research Future. (2023). Albuterol Sulfate Market - Global Forecast 2030. Retrieved from https://www.marketresearchfuture.com/reports/albuterol-sulfate-market-5484
[6] Various industry reports and company filings. Specific market share data is proprietary and fluctuates.
[7] Teva Pharmaceutical Industries Ltd. Company Website. (Accessed 2024).
[8] GlaxoSmithKline plc. Company Website. (Accessed 2024).
[9] Boehringer Ingelheim GmbH. Company Website. (Accessed 2024).
[10] Viatris Inc. (formerly Mylan N.V.). Company Website. (Accessed 2024).
[11] Perrigo Company plc. Company Website. (Accessed 2024).
[12] Chiesi Farmaceutici S.p.A. Company Website. (Accessed 2024).
[13] Sun Pharmaceutical Industries Ltd. Company Website. (Accessed 2024).
[14] Cipla Inc. Company Website. (Accessed 2024).
[15] Pharmaceutical patent databases (e.g., USPTO, Espacenet, Google Patents). Analysis of specific patent families requires specialized search.
[16] U.S. Food & Drug Administration. (2023). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[17] U.S. Food & Drug Administration. (n.d.). Drug Development Process. Retrieved from https://www.fda.gov/patients/drug-development-process
[18] U.S. Food & Drug Administration. (n.d.). Generic Drugs. Retrieved from https://www.fda.gov/drugs/generic-drugs
[19] Market analysis reports from firms such as IQVIA, Clarivate, and others provide financial projections. Specific figures are often proprietary.