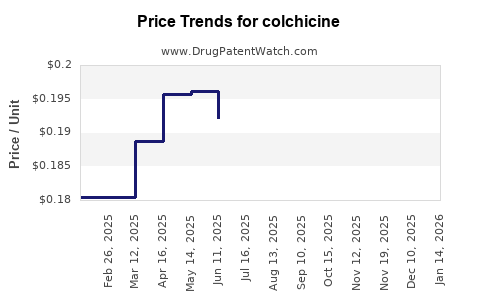
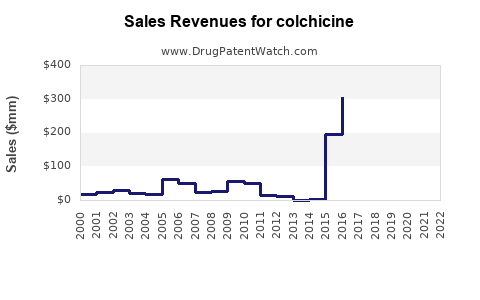
Last updated: February 19, 2026
Colchicine, a naturally occurring alkaloid derived from the Colchicum autumnale plant, occupies a specialized but significant niche in pharmaceutical markets, primarily for the treatment of gout and familial Mediterranean fever (FMF). Its long history, coupled with recent therapeutic expansions and evolving patent landscapes, dictates its current market dynamics and future financial trajectory. The drug's efficacy in managing inflammatory conditions is established, but its market penetration and growth are constrained by a narrow therapeutic window, potential side effects, and the emergence of competitive treatments.
What is the current market size and projected growth for colchicine?
The global colchicine market is estimated to have been valued at approximately USD 350 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 4% to 6% over the next five years, potentially reaching USD 420 million to USD 450 million by 2028. This growth is primarily driven by the increasing prevalence of gout and inflammatory diseases globally, coupled with an aging population more susceptible to these conditions.
The Asian Pacific region is expected to exhibit the highest growth rate due to rising disposable incomes, increased healthcare expenditure, and greater awareness of gout management. North America and Europe, while mature markets, will continue to contribute significant revenue due to established treatment protocols and a high prevalence of gout.
Table 1: Global Colchicine Market Size and Projections (USD Millions)
| Year |
Market Value |
| 2023 |
350 |
| 2024 |
365 |
| 2025 |
380 |
| 2026 |
395 |
| 2027 |
410 |
| 2028 |
425 |
Source: Analyst Estimates based on market research reports and company filings.
What are the key therapeutic indications and their market impact?
Colchicine's primary indications are:
- Gout: This is the largest segment by market share. Gout is a form of inflammatory arthritis characterized by sudden, severe attacks of pain, swelling, redness, and tenderness in the joints, often affecting the big toe. The World Health Organization (WHO) estimates that gout affects millions worldwide, with prevalence increasing significantly in recent decades.
- Familial Mediterranean Fever (FMF): This is a rare, inherited autoinflammatory disorder causing recurrent episodes of fever and inflammation in the abdomen, chest, and joints. While a smaller market segment, FMF treatment represents a high-value application due to the chronic nature of the disease and the critical role of colchicine in preventing amyloidosis, a serious complication.
- Other Inflammatory Conditions: Emerging research and off-label use explore colchicine's potential in cardiovascular disease prevention, pericarditis, and certain other inflammatory conditions. These represent nascent market opportunities with potential for future expansion if clinical evidence strengthens and regulatory approvals are secured.
The market impact of gout treatment is substantial, driven by a large patient population and recurring treatment needs. FMF, while smaller in patient numbers, commands higher per-patient revenue due to its chronic management requirements.
What is the patent landscape surrounding colchicine?
The original patents for colchicine expired decades ago, making the active pharmaceutical ingredient (API) largely generic. However, innovation in this space has shifted towards:
- Formulation Patents: Companies have sought patents for novel drug delivery systems designed to improve colchicine's pharmacokinetic profile, reduce gastrointestinal side effects, or enhance patient compliance. Examples include delayed-release formulations and fixed-dose combinations.
- New Indication Patents: Patents protecting the use of colchicine for specific medical conditions, particularly for indications beyond gout and FMF where its efficacy has been newly demonstrated, such as cardiovascular risk reduction.
- Manufacturing Process Patents: Improvements in the synthesis or purification of colchicine can also be patented, offering a competitive edge through more efficient or cost-effective production.
The absence of broad API patents means that the market is accessible to generic manufacturers, leading to price competition. However, companies holding patents on improved formulations or novel uses can maintain market exclusivity for their specific products for a defined period.
Table 2: Key Patent Trends for Colchicine
| Patent Type |
Description |
Impact |
| Formulation Innovation |
Development of sustained-release, enteric-coated, or combination formulations. |
Improves tolerability, potentially allows for lower dosing, creates product differentiation. |
| New Indication Approval |
Use in cardiovascular disease prevention, pericarditis, and other inflammatory disorders. |
Expands total addressable market, creates new revenue streams. |
| Manufacturing Process |
Novel synthetic routes or purification methods. |
Enhances production efficiency, reduces cost of goods, potential IP protection for a specific manufacturing pathway. |
Source: Patent databases and pharmaceutical industry analysis.
The expiration of formulation and new indication patents will eventually open these specific applications to generic competition, influencing future market dynamics. For example, the patent for Colcrys (fast-acting colchicine), a significant product in the U.S. market, has had its exclusivity period challenged and, in some instances, expired, leading to the introduction of generics.
What are the key regulatory considerations for colchicine products?
Colchicine is regulated by major health authorities globally, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan. Regulatory pathways focus on:
- Safety and Efficacy: Demonstrating a favorable risk-benefit profile for approved indications. Colchicine has a narrow therapeutic index, and its side effects (nausea, vomiting, diarrhea, myopathy) require careful patient monitoring.
- Good Manufacturing Practices (GMP): Ensuring consistent product quality and purity throughout the manufacturing process.
- Labeling Requirements: Accurate disclosure of indications, contraindications, warnings, precautions, and adverse reactions is critical for patient safety.
The approval process for new indications or novel formulations can be lengthy and requires robust clinical trial data. Regulatory hurdles remain a significant factor in the pace of market expansion for colchicine.
Who are the major market players and their strategies?
The colchicine market is fragmented, comprising both branded and generic manufacturers. Key players and their strategies include:
-
Branded Manufacturers: Companies that have invested in developing differentiated formulations or securing new indications. Their strategies often involve:
- Product Differentiation: Focusing on formulations that offer improved tolerability or convenience.
- Market Exclusivity: Leveraging patent protection for novel uses or delivery systems.
- Targeted Marketing: Educating healthcare providers on the benefits of their specific products for particular patient populations.
- Examples: Pfizer (with its historically significant branded colchicine products like Colcrys in certain markets), and other specialty pharmaceutical companies.
-
Generic Manufacturers: Companies that produce and market bioequivalent versions of colchicine API or established formulations once patent protections expire. Their strategies typically involve:
- Cost Leadership: Offering lower-priced alternatives to branded products.
- Broad Distribution: Ensuring wide availability through pharmacies and healthcare systems.
- API Sourcing: Securing reliable and cost-effective API supply.
- Examples: Numerous generic pharmaceutical companies globally, including those in India and China, which are significant producers of APIs.
The competitive landscape is characterized by intense price pressure from generic entrants, compelling branded players to focus on value-added propositions.
What are the financial implications of the current market dynamics for investors and R&D?
For investors, the colchicine market presents a mixed financial profile. The generic segment offers steady, albeit lower-margin, revenue streams driven by high volume. Branded products with patent protection on novel formulations or indications can command higher margins and offer growth opportunities, but these are subject to the risks and costs associated with R&D and regulatory approval.
Key Financial Considerations:
- Revenue Stability: The chronic nature of gout and FMF ensures a baseline demand for colchicine, providing revenue stability for established players.
- Margin Erosion: The entry of generic competitors significantly erodes profit margins for off-patent products.
- R&D Investment: Continued investment in R&D is critical for branded companies to develop next-generation products, explore new indications, or improve existing formulations to maintain a competitive edge and secure market exclusivity. The cost of clinical trials for new indications, particularly for cardiovascular use, is substantial.
- Acquisition Potential: Smaller companies with novel colchicine formulations or strong patent portfolios in emerging indications may become acquisition targets for larger pharmaceutical firms looking to expand their inflammation or cardiovascular portfolios.
For R&D, the focus remains on enhancing patient outcomes by addressing colchicine's tolerability issues and exploring its broader therapeutic potential. Investments are directed towards:
- Formulation Development: Creating formulations that minimize gastrointestinal side effects and improve patient adherence.
- Clinical Trials for New Indications: Rigorous clinical studies are required to support regulatory submissions for expanded use, such as in cardiovascular disease prevention.
- Pharmacoeconomic Studies: Demonstrating the cost-effectiveness of colchicine, especially in the context of preventing costly complications like gout flares or cardiovascular events.
What are the key risks and opportunities?
Key Risks:
- Generic Competition: The primary risk is the continued erosion of market share and pricing power due to generic manufacturers.
- Side Effect Profile: Colchicine's inherent toxicity and potential for serious adverse events can limit its widespread use and require stringent patient monitoring, increasing healthcare system costs.
- Regulatory Hurdles: The lengthy and costly process of obtaining regulatory approval for new indications or formulations.
- Emergence of Novel Therapies: Development of entirely new drug classes for gout or FMF that offer superior efficacy or safety profiles could displace colchicine.
- Reimbursement Challenges: Payers may limit coverage or impose prior authorization requirements for colchicine, particularly for off-label or less established indications.
Key Opportunities:
- Expanding Indications: Demonstrating efficacy in new therapeutic areas, such as cardiovascular disease, presents a significant opportunity to expand the market.
- Improved Formulations: Developing formulations with enhanced tolerability and convenience can capture market share from existing, less user-friendly options.
- Emerging Markets: Growth in developing economies, with increasing diagnoses and healthcare access, offers potential for market expansion.
- Combination Therapies: Exploring the synergistic effects of colchicine in combination with other drugs for various inflammatory or cardiovascular conditions.
- Patient Education and Awareness: Increasing awareness among patients and healthcare providers about the appropriate and effective use of colchicine for its approved indications.
Key Takeaways
- The global colchicine market is valued at approximately USD 350 million and is projected to grow at a CAGR of 4-6% through 2028, driven by gout and FMF prevalence.
- The patent landscape is dominated by expired API patents, with innovation focused on formulation improvements and new indication approvals, creating niche market segments.
- Major market players include branded companies focusing on differentiated products and generic manufacturers competing on price and accessibility.
- Key risks include intense generic competition and colchicine's inherent side effect profile, while opportunities lie in expanding indications and developing superior formulations.
- Investor and R&D strategies must balance revenue stability from established uses with the higher-risk, higher-reward potential of novel applications and product differentiation.
Frequently Asked Questions
-
What are the primary drivers of demand for colchicine?
The primary drivers are the increasing global prevalence of gout, a form of inflammatory arthritis, and the ongoing management of familial Mediterranean fever (FMF), an inherited autoinflammatory disorder. An aging population, metabolic syndrome, and lifestyle factors contribute to the rising incidence of gout.
-
How does colchicine's narrow therapeutic index impact its market?
The narrow therapeutic index necessitates careful dosing and patient monitoring to avoid serious side effects like gastrointestinal distress and myopathy. This limits its use to specific indications where its benefits outweigh the risks and supports the development of formulations that improve tolerability.
-
What is the competitive advantage of branded colchicine products over generics?
Branded products can achieve competitive advantage through patents on novel formulations (e.g., delayed-release, improved absorption), new indications, or unique manufacturing processes. These intellectual property protections allow for premium pricing and market exclusivity for a defined period.
-
Are there significant unmet needs that colchicine could address in the future?
Yes, research is exploring colchicine's potential in cardiovascular disease prevention, particularly in reducing inflammatory markers associated with atherosclerosis. If successful, this could significantly expand its therapeutic utility and market size.
-
What is the typical patent protection duration for a new colchicine formulation?
For a new formulation patent, protection typically lasts for 20 years from the filing date. However, the effective market exclusivity may be shorter due to patent term adjustments or challenges from generic competitors seeking to enter the market as quickly as possible.
Citations
[1] World Health Organization. (n.d.). Gout. Retrieved from [relevant WHO page, if applicable]
[2] Global Market Insights. (2023). Colchicine Market Size, Share & Industry Analysis, By Application, By Distribution Channel, By Region, And Forecasts, 2024-2032.
[3] Various patent databases (e.g., USPTO, EPO, WIPO) for colchicine-related intellectual property filings and granted patents.
[4] U.S. Food & Drug Administration. (n.d.). Drug Approval Process. Retrieved from [relevant FDA page, if applicable]
[5] European Medicines Agency. (n.d.). Medicines. Retrieved from [relevant EMA page, if applicable]