Last updated: February 19, 2026
EXECUTIVE SUMMARY
Sirolimus, an immunosuppressant and mTOR inhibitor, faces a mature market characterized by established generics and ongoing patent expiration. Key patents for the original formulation and indications have lapsed, leading to significant generic competition. However, new patent filings focus on novel formulations, delivery methods, and expanded therapeutic applications, particularly in oncology and rare diseases. This evolving landscape necessitates strategic R&D investment in differentiated formulations and new indications to secure future market share.
PATENT EXPIRATION AND GENERIC COMPETITION
The original U.S. patent for Sirolimus (Rapamune), held by Wyeth (now Pfizer), expired in 2009. [1] This expiration opened the door for multiple generic manufacturers to enter the market. Companies such as Teva Pharmaceuticals, Dr. Reddy's Laboratories, and Mylan (now Viatris) have since launched generic versions of sirolimus.
- Original Patent Expiration: U.S. Patent 4,992,447, covering the compound sirolimus, expired on March 26, 2009. [1]
- Generic Market Entry:
- Teva Pharmaceuticals received FDA approval for its generic sirolimus oral solution in 2009. [2]
- Dr. Reddy's Laboratories launched its generic sirolimus tablets in 2009. [3]
- Mylan introduced its generic sirolimus tablets and oral solution in 2012. [4]
The availability of multiple generic options has driven down prices and increased market penetration for the drug's established indications, primarily organ transplant rejection prophylaxis and treatment of lymphangioleiomyomatosis (LAM).
NEW PATENT FILINGS AND INNOVATION
Despite the generic competition for the original product, pharmaceutical innovation continues through new patent filings. These patents are strategically designed to protect novel aspects of sirolimus, including improved delivery systems, new formulations, and expanded therapeutic uses.
Novel Formulations and Delivery Systems
Research and development efforts are focused on overcoming limitations of oral sirolimus, such as variable bioavailability and gastrointestinal side effects. New formulations aim for improved efficacy, reduced dosing frequency, and better patient compliance.
- Oral Liquid Formulations: Patents have been filed for improved oral liquid formulations that offer enhanced stability and taste masking. For example, patents may describe compositions with specific excipients to improve dissolution profiles or reduce degradation.
- Topical Formulations: Development of topical sirolimus formulations is underway for dermatological applications, potentially targeting conditions like psoriasis or certain types of skin cancer. These patents often detail specific cream or ointment bases and penetration enhancers.
- Injectable Formulations: Research into injectable or depot formulations could offer advantages for patients who have difficulty with oral administration or require sustained release. Patents in this area may cover microparticle encapsulation or nanoparticle-based delivery systems.
Expanded Therapeutic Applications
The most significant area of ongoing patent activity and market expansion for sirolimus lies in its application beyond transplantation. Its mechanism as an mTOR inhibitor makes it a candidate for a range of other diseases, particularly in oncology and rare genetic disorders.
Oncology
Sirolimus's role in inhibiting cell growth and proliferation pathways has led to its investigation and repurposing in various cancers. Patents in this domain often focus on specific cancer types, combination therapies, or novel dosing regimens.
- Hereditary Leiomyomatosis and Renal Cell Cancer (HLRCC): Sirolimus has shown promise in treating HLRCC, a rare genetic syndrome associated with multiple fibroids and an increased risk of renal cell carcinoma. Patents may cover its use in preventing or treating the renal tumors associated with this condition.
- Other Cancers: Investigations are ongoing for its efficacy in other solid tumors and hematological malignancies. Patents might be filed for its use in treating conditions such as:
- Angiomyolipomas (often associated with Tuberous Sclerosis Complex)
- Certain types of lymphomas
- Pancreatic neuroendocrine tumors
Rare Diseases and Genetic Disorders
Sirolimus is emerging as a therapeutic option for several rare genetic conditions due to its ability to modulate cell growth and protein synthesis.
- Tuberous Sclerosis Complex (TSC): Sirolimus has received FDA approval for treating TSC-associated tumors, including subependymal giant cell astrocytomas (SEGAs) and renal angiomyolipomas. [5] Patents related to TSC often cover specific dosing regimens and the management of these tumor types. The approval of sirolimus (as Afinitor by Novartis, which contains everolimus, a related analog, but the principle applies to sirolimus's broad mTOR inhibition) for TSC-related SEGAs in 2009 highlights this therapeutic expansion. [6]
- Pulmonary Lymphangioleiomyomatosis (LAM): While existing patents cover its use in LAM, ongoing research might lead to new formulations or combination therapies protected by subsequent patents.
- Other Potential Indications: Emerging research suggests potential benefits in conditions like Gorlin syndrome or certain rare vascular malformations, which could lead to future patent filings.
INTELLECTUAL PROPERTY STRATEGIES
Pharmaceutical companies are employing various strategies to protect their investment in sirolimus and secure future market exclusivity.
- Composition of Matter Patents: The original composition of matter patent has expired. New patents are not for the molecule itself but for specific crystalline forms, polymorphs, or enantiomers that may exhibit improved properties.
- Method of Use Patents: These patents protect specific therapeutic applications of sirolimus. For example, a patent might claim the method of treating a particular cancer by administering sirolimus at a defined dosage and frequency. These are crucial for expanding the drug's market.
- Formulation and Delivery Patents: As mentioned, patents are filed for novel drug delivery systems, including extended-release formulations, topical preparations, or nanoparticle-based delivery.
- Everolimus and Related Analogs: It is important to distinguish sirolimus from its analogs, such as everolimus, which have their own patent portfolios and market dynamics. Everolimus, for instance, has been successfully developed for various oncology indications (e.g., breast cancer, renal cell carcinoma) with its own distinct patent protection. [6]
MARKET DYNAMICS AND FINANCIAL TRAJECTORY
The market for sirolimus is bifurcated. The established indications face a mature, price-sensitive generic market. The future growth trajectory is contingent on the successful development and patent protection of new indications and formulations.
Established Indications (Transplant, LAM)
- Market Size: The market for sirolimus in transplant rejection prophylaxis is significant but has been largely commoditized due to generic availability.
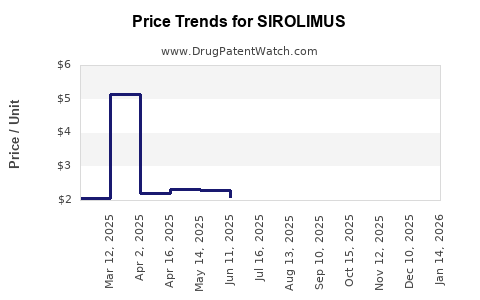
- Pricing: Generic sirolimus pricing is competitive, with significant price erosion since the loss of market exclusivity.
- Growth: Limited growth is expected for these traditional uses, primarily driven by transplant volumes and cost-containment measures by healthcare payers.
Expanding Indications (Oncology, Rare Diseases)
- Growth Potential: This segment represents the primary growth driver for sirolimus and its derivatives. Approvals for conditions like TSC have opened new revenue streams.
- Pricing: Drugs for rare diseases and oncology indications typically command higher prices, allowing for greater revenue generation per patient.
- R&D Investment: Continued investment in clinical trials and patent applications for new indications is essential to unlock this potential.
- Competition: While sirolimus itself may be off-patent for basic use, competition in oncology and rare diseases can come from other mTOR inhibitors, targeted therapies, and immunotherapies.
Key Market Players
- Pfizer (formerly Wyeth): Holds original intellectual property and may still have market share in certain regions or for specific formulations.
- Generic Manufacturers: Teva, Dr. Reddy's, Viatris, and others are major suppliers of generic sirolimus for established indications.
- Specialty Pharma & Biotech: Companies focusing on novel formulations or rare disease applications may hold key patents and target specific patient populations.
- Novartis: A significant player in the mTOR inhibitor space with everolimus, which competes directly in some expanded indications.
FINANCIAL CONSIDERATIONS FOR INVESTORS AND R&D
For R&D departments and investors, the sirolimus market presents both challenges and opportunities.
Challenges
- Generic Price Erosion: For traditional indications, achieving profitability requires high-volume sales and efficient manufacturing to compete on price.
- Long Development Cycles: Bringing new indications to market requires extensive and costly clinical trials, with no guarantee of regulatory approval.
- Patent Litigation: Companies developing new formulations or uses must be prepared for potential patent challenges from competitors.
Opportunities
- Orphan Drug Designation: For rare disease indications, orphan drug status can provide market exclusivity periods (e.g., 7 years in the U.S., 10 years in Europe) beyond patent protection, as well as tax credits and R&D grants. [7]
- Combination Therapies: Developing sirolimus as part of a combination therapy in oncology can create new patentable methods of treatment and potentially synergistic efficacy.
- Differentiated Formulations: Investing in novel delivery systems that offer clear clinical advantages (e.g., improved tolerability, less frequent dosing) can support premium pricing and market differentiation, even in the face of generic competition for the base drug.
- Repurposing: The ongoing exploration of sirolimus for new indications leverages existing safety and efficacy data, potentially reducing development timelines and costs compared to developing entirely new molecular entities.
INTELLECTUAL PROPERTY SUMMARY
| Patent Type |
Status |
Focus |
Market Impact |
| Composition of Matter |
Expired (U.S. Patent 4,992,447, 2009) |
The sirolimus molecule itself. |
Opens market to generic competition for all indications. |
| Method of Use (Transplant) |
Expired or nearing expiration |
Prophylaxis of organ rejection. |
Generic availability and price pressure. |
| Method of Use (LAM) |
Expired or nearing expiration |
Treatment of lymphangioleiomyomatosis. |
Generic availability and price pressure. |
| Method of Use (TSC) |
Active and pending, some approved |
Treatment of TSC-associated tumors (SEGAs, angiomyolipomas). |
Creates distinct, higher-value market segments with potential for exclusivity. |
| Method of Use (Oncology) |
Active and pending, some approved |
Treatment of various cancers (e.g., HLRCC, pancreatic neuroendocrine). |
Potential for new revenue streams, requiring clinical validation and patent robust- |
| Formulation/Delivery |
Active and pending |
Novel oral liquids, topical preparations, injectable formulations. |
Can support premium pricing, improve patient compliance, and extend product life- |
| Polymorphs/Salts |
Active and pending |
Specific crystalline forms or salts with improved properties. |
Can create narrow barriers to entry for competitors on specific forms. |
KEY TAKEAWAYS
- Sirolimus faces widespread generic competition for its original indications, significantly impacting pricing and market dynamics for these uses.
- New patent filings are concentrated on novel formulations, improved delivery systems, and expanded therapeutic applications, particularly in oncology and rare genetic diseases.
- Companies are strategically pursuing method-of-use patents and formulation patents to create new market segments and extend exclusivity.
- The financial trajectory of sirolimus will be increasingly driven by the success of these new indications and differentiated products, which typically command higher pricing and benefit from orphan drug designations.
- Investors and R&D professionals must analyze both the mature generic market and the dynamic, patent-protected segments of expanding therapeutic areas.
FAQS
-
What is the primary driver of sirolimus market value beyond its original indications?
The primary driver is the expansion into new therapeutic areas, particularly oncology and rare genetic diseases like Tuberous Sclerosis Complex (TSC), which are protected by method-of-use patents and can command higher pricing.
-
How does the patent status of sirolimus affect its pricing for transplant patients?
The expiration of key composition-of-matter and method-of-use patents for transplant prophylaxis has led to widespread generic competition, resulting in significant price erosion and competitive pricing for generic sirolimus.
-
What types of innovations are pharmaceutical companies pursuing to extend sirolimus's market life?
Companies are focusing on developing novel formulations (e.g., topical, injectable), improved delivery systems (e.g., extended-release), and seeking regulatory approval for new therapeutic indications through method-of-use patents.
-
What is the competitive landscape for sirolimus in the oncology sector?
In oncology, sirolimus competes with other mTOR inhibitors (like everolimus) and a broad range of targeted therapies and immunotherapies. Patent protection for specific cancer types and combination therapies is critical for market differentiation.
-
How does orphan drug designation impact the commercial strategy for sirolimus in rare diseases?
Orphan drug designation provides market exclusivity for a defined period (e.g., 7 years in the U.S.) beyond patent expiration, offers R&D incentives, and allows for premium pricing, making rare disease indications a key focus for companies seeking to maximize sirolimus's commercial potential.
CITATIONS
[1] U.S. Patent 4,992,447. (1991). Immunosuppressive agents. Wyeth.
[2] U.S. Food & Drug Administration. (2009). Orange Book. Teva Pharmaceuticals.
[3] U.S. Food & Drug Administration. (2009). Orange Book. Dr. Reddy's Laboratories.
[4] U.S. Food & Drug Administration. (2012). Orange Book. Mylan.
[5] U.S. Food & Drug Administration. (n.d.). Drug Approvals & Databases. Retrieved from fda.gov
[6] Novartis. (2009). Afinitor (everolimus) approval. (Press Release).
[7] U.S. Food & Drug Administration. (n.d.). Orphan Drug Act. Retrieved from fda.gov