PREGABALIN - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for pregabalin and what is the scope of patent protection?
Pregabalin
is the generic ingredient in three branded drugs marketed by Upjohn, Actavis Elizabeth, Adaptis, Alembic, Alkem Labs Ltd, Amneal Pharms Co, Apotex, Aurobindo Pharma, Cadila Pharms Ltd, Changzhou Pharm, Chartwell Rx, Cipla, Creekwood Pharms, Dr Reddys, Eskayef, Hetero Labs Ltd Iii, Invagen Pharms, Jubilant Generics, Lupin Ltd, Macleods Pharms Ltd, MSN, Pharmobedient, Prinston Inc, Regcon Holdings, Rising, Sciegen Pharms, Somerset Theraps Llc, Strides Pharma, Sun Pharm, Teva Pharms, Torrent, Yiling, Zydus Pharms, Anda Repository, Aiping Pharm Inc, Alvogen, Epic Pharma Llc, and Rubicon Research, and is included in fifty-three NDAs. There are three patents protecting this compound and six Paragraph IV challenges. Additional information is available in the individual branded drug profile pages.Pregabalin has thirty-seven patent family members in thirty-three countries.
There are forty-one drug master file entries for pregabalin. Fifty-five suppliers are listed for this compound. There are six tentative approvals for this compound.
Summary for PREGABALIN
| International Patents: | 37 |
| US Patents: | 3 |
| Tradenames: | 3 |
| Applicants: | 38 |
| NDAs: | 53 |
| Drug Master File Entries: | 41 |
| Finished Product Suppliers / Packagers: | 55 |
| Raw Ingredient (Bulk) Api Vendors: | 69 |
| Clinical Trials: | 577 |
| Patent Applications: | 6,942 |
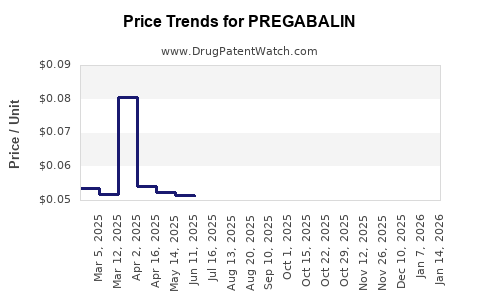
| Drug Prices: | Drug price trends for PREGABALIN |
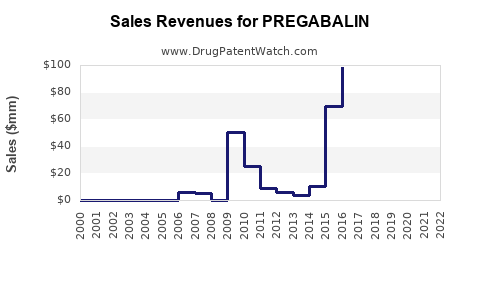
| Drug Sales Revenues: | Drug sales revenues for PREGABALIN |
| What excipients (inactive ingredients) are in PREGABALIN? | PREGABALIN excipients list |
| DailyMed Link: | PREGABALIN at DailyMed |
Recent Clinical Trials for PREGABALIN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| McMaster University | PHASE4 |
| Eurofarma Laboratorios S.A. | PHASE3 |
| Tanta University | NA |
Generic filers with tentative approvals for PREGABALIN
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Start Trial | ⤷ Start Trial | 300MG | CAPSULE;ORAL |
| ⤷ Start Trial | ⤷ Start Trial | 225MG | CAPSULE;ORAL |
| ⤷ Start Trial | ⤷ Start Trial | 200MG | CAPSULE;ORAL |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Medical Subject Heading (MeSH) Categories for PREGABALIN
Anatomical Therapeutic Chemical (ATC) Classes for PREGABALIN
Paragraph IV (Patent) Challenges for PREGABALIN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LYRICA CR | Extended-release Tablets | pregabalin | 82.5 mg and 165 mg | 209501 | 1 | 2018-02-02 |
| LYRICA CR | Extended-release Tablets | pregabalin | 330 mg | 209501 | 1 | 2018-01-29 |
| LYRICA | Oral Solution | pregabalin | 20 mg/mL | 022488 | 1 | 2010-05-19 |
| LYRICA | Capsules | pregabalin | 25 mg, 50 mg, 75 mg, 100 mg, 150 mg, 200 mg, 225 mg and 300 mg | 021446 | 8 | 2008-12-30 |
US Patents and Regulatory Information for PREGABALIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Msn | PREGABALIN | pregabalin | CAPSULE;ORAL | 209357-008 | Jul 19, 2019 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Jubilant Generics | PREGABALIN | pregabalin | CAPSULE;ORAL | 209530-003 | May 20, 2025 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Apotex | PREGABALIN | pregabalin | CAPSULE;ORAL | 211685-006 | Jul 7, 2021 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Somerset Theraps Llc | PREGABALIN | pregabalin | CAPSULE;ORAL | 210585-003 | Dec 26, 2019 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Anda Repository | PREGABALIN | pregabalin | SOLUTION;ORAL | 212604-001 | Feb 18, 2022 | AA | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Hetero Labs Ltd Iii | PREGABALIN | pregabalin | CAPSULE;ORAL | 206912-007 | Oct 8, 2019 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Sciegen Pharms | PREGABALIN | pregabalin | TABLET, EXTENDED RELEASE;ORAL | 215675-002 | Sep 14, 2022 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for PREGABALIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Upjohn | LYRICA | pregabalin | SOLUTION;ORAL | 022488-001 | Jan 4, 2010 | 5,563,175 | ⤷ Start Trial |
| Upjohn | LYRICA | pregabalin | CAPSULE;ORAL | 021446-001 | Dec 30, 2004 | 5,563,175 | ⤷ Start Trial |
| Upjohn | LYRICA | pregabalin | CAPSULE;ORAL | 021446-005 | Dec 30, 2004 | 5,563,175 | ⤷ Start Trial |
| Upjohn | LYRICA | pregabalin | CAPSULE;ORAL | 021446-008 | Dec 30, 2004 | 5,563,175 | ⤷ Start Trial |
| Upjohn | LYRICA | pregabalin | CAPSULE;ORAL | 021446-006 | Dec 30, 2004 | 5,563,175 | ⤷ Start Trial |
| Upjohn | LYRICA | pregabalin | CAPSULE;ORAL | 021446-007 | Dec 30, 2004 | 5,563,175 | ⤷ Start Trial |
| Upjohn | LYRICA | pregabalin | CAPSULE;ORAL | 021446-002 | Dec 30, 2004 | 5,563,175 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for PREGABALIN
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Zentiva k.s. | Pregabalin Zentiva k.s. | pregabalin | EMEA/H/C/004277Neuropathic painPregabalin Zentiva k.s. is indicated for the treatment of peripheral and central neuropathic pain in adults.EpilepsyPregabalin Zentiva k.s. is indicated as adjunctive therapy in adults with partial seizures with or without secondary generalisation.Generalised anxiety disorderPregabalin Zentiva k.s. is indicated for the treatment of generalised anxiety disorder (GAD) in adults. | Withdrawn | yes | no | no | 2017-02-27 | |
| Zentiva, k.s. | Pregabalin Zentiva | pregabalin | EMEA/H/C/003900Neuropathic pain, , , Pregabalin Zentiva is indicated for the treatment of peripheral and central neuropathic pain in adults., , , Epilepsy, , , Pregabalin Zentiva is indicated as adjunctive therapy in adults with partial seizures with or without secondary generalisation., , , Generalised anxiety disorder, , , Pregabalin Zentiva is indicated for the treatment of generalised anxiety disorder (GAD) in adults., , | Authorised | yes | no | no | 2015-07-17 | |
| Mylan S.A.S. | Pregabalin Mylan Pharma | pregabalin | EMEA/H/C/003962EpilepsyPregabalin Mylan Pharma is indicated as adjunctive therapy in adults with partial seizures with or without secondary generalisation.Generalised Anxiety DisorderPregabalin Mylan Pharma is indicated for the treatment of Generalised Anxiety Disorder (GAD) in adults. | Withdrawn | yes | no | no | 2015-06-25 | |
| Upjohn EESV | Pregabalin Pfizer | pregabalin | EMEA/H/C/003880Neuropathic painPregabalin Pfizer is indicated for the treatment of peripheral and central neuropathic pain in adults.EpilepsyPregabalin Pfizer is indicated as adjunctive therapy in adults with partial seizures with or without secondary generalisation.Generalised Anxiety DisorderPregabalin Pfizer is indicated for the treatment of Generalised Anxiety Disorder (GAD) in adults. | Authorised | no | no | no | 2014-04-10 | |
| Upjohn EESV | Lyrica | pregabalin | EMEA/H/C/000546Neuropathic painLyrica is indicated for the treatment of peripheral and central neuropathic pain in adults.EpilepsyLyrica is indicated as adjunctive therapy in adults with partial seizures with or without secondary generalisation.Generalised anxiety disorderLyrica is indicated for the treatment of generalised anxiety disorder (GAD) in adults. | Authorised | no | no | no | 2004-07-05 | |
| Sandoz GmbH | Pregabalin Sandoz GmbH | pregabalin | EMEA/H/C/004070EpilepsyPregabalin Sandoz GmbH is indicated as adjunctive therapy in adults with partial seizures with or without secondary generalisation.Generalised Anxiety DisorderPregabalin Sandoz GmbH is indicated for the treatment of Generalised Anxiety Disorder (GAD) in adults. | Withdrawn | yes | no | no | 2015-06-19 | |
| Accord Healthcare S.L.U. | Pregabalin Accord | pregabalin | EMEA/H/C/004024EpilepsyPregabalin Accord is indicated as adjunctive therapy in adults with partial seizures with or without secondary generalisation.Generalised Anxiety DisorderPregabalin Accord is indicated for the treatment of Generalised Anxiety Disorder (GAD) in adults. | Authorised | yes | no | no | 2015-08-28 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for PREGABALIN
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Taiwan | 200803831 | Solid pharmaceutical compositions containing pregabalin | ⤷ Start Trial |
| Ecuador | SP088422 | COMPOSICIONES FARMACÉUTICAS SÓLIDAS QUE CONTIENEN PREGABALINA | ⤷ Start Trial |
| Serbia | 20080181 | ČVRSTE FARMACEUTSKE KOMPOZICIJE KOJE SADRŽE PREGABALIN SREDSTVO ZA FORMIRANJE MATRIKSA I SREDSTVO ZA BUBRENJE (SOLID PHARMACEUTICAL COMPOSITIONS CONTAINING PREGABALIN, A MATRIX FORMING AGENT AND A SWELLING AGENT) | ⤷ Start Trial |
| Dominican Republic | P2006000241 | COMPOSICIONES FARMACÉUTICAS SÓLIDAS QUE CONTIENEN PREGABALINA | ⤷ Start Trial |
| Montenegro | 00482 | ČVRSTE FARMACEUTSKE KOMPOZICIJE KOJE SADRŽE PREGABALIN SREDSTVO ZA FORMIRANJE MATRIKSA I SREDSTVO ZA BUBRENJE (SOLID ORAL PHARMACEUTICAL COMPOSITIONS FOR ONCE DAILY DOSING CONTAINING PREGABALIN, A MATRIX FORMING AGENT AND A SWELLING AGENT) | ⤷ Start Trial |
| Norway | 20081816 | ⤷ Start Trial | |
| Japan | 4334610 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for PREGABALIN
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0641330 | 27/2004 | Austria | ⤷ Start Trial | PRODUCT NAME: PREGABALIN, GEGEBENENFALLS IN FORM EINES PHARMAZEUTISCH ANNEHMBAREN SALZES; REGISTRATION NO/DATE: EU/1/04/279/001 - EU/1/04/279/025 20040706 |
| 0641330 | SPC/GB04/034 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: PREGABALIN (S-(+)-4-AMINO-3(2-METHYLPROPYL)BUTANOIC ACID) OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACTIVE SALT.; REGISTERED: UK EU/1/04/279/001 20040706; UK EU/1/04/279/002 20040706; UK EU/1/04/279/003 20040706; UK EU/1/04/279/004 20040706; UK EU/1/04/279/005 20040706; UK EU/1/04/279/006 20040706; UK EU/1/04/279/025 20040706; UK EU/1/04/279/019 20040706; UK EU/1/04/279/020 20040706; UK EU/1/04/279/021 20040706; UK EU/1/04/279/022 20040706; UK EU/1/04/279/023 20040706; UK EU/1/04/279/024 20040706; UK EU/1/04/279/013 20040706; UK EU/1/04/279/014 20040706; UK EU/1/04/279/015 20040706; UK EU/1/04/279/016 20040706; UK EU/1/04/279/017 20040706; UK EU/1/04/279/018 20040706; UK EU/ |
| 0934061 | PA2004017,C0934061 | Lithuania | ⤷ Start Trial | PRODUCT NAME: PREGABALINUM ((S)-3-(AMINOMETIL)-5-METILHEKSANO RUGSTIS); REGISTRATION NO/DATE: EU/1/04/279/001-025 20040725 |
| 0934061 | PA2004017 | Lithuania | ⤷ Start Trial | PRODUCT NAME: PREGABALINUM ((S)-3-(AMINOMETIL)-5-METILHEKSANO RûGðTIS) |
| 0641330 | CR 2004 00036 | Denmark | ⤷ Start Trial | PRODUCT NAME: PREGABALIN ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/04/279/001-025 20040706 |
| 0641330 | C300164 | Netherlands | ⤷ Start Trial | PRODUCT NAME: PREGABALINE, DESGEWENST IN DE; REGISTRATION NO/DATE: EU/1/04/279/001 20040706 |
| 0641330 | 2004C/022 | Belgium | ⤷ Start Trial | PRODUCT NAME: PREGABALIN; REGISTRATION NO/DATE: EU/1/04/279/001 20040708 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for Pregabalin
More… ↓