Last updated: April 24, 2026
Pregabalin (brand Lyrica and generics) has moved through a classic late-molecule life cycle: steep revenue concentration in branded originator years, followed by rapid post-expiry share loss to multi-supplier generics, then stabilization in many markets as manufacturing footprint and pricing converge. The financial trajectory is now defined less by pipeline surprise and more by (1) patent and exclusivity timelines by geography, (2) payer adoption of branded versus generic, (3) utilization linked to indications (neuropathic pain, fibromyalgia, seizure adjunct), and (4) competitive intensity from global generic manufacturers and formulation differences.
Where is pregabalin selling today and why does utilization hold up?
Pregabalin is a “broad use” CNS analgesic and adjunct anti-epileptic with multiple approved indications that expand the treatable patient base:
- Neuropathic pain (common across major markets)
- Fibromyalgia (where approved)
- Adjunct therapy for partial-onset seizures (common across major markets)
The practical commercial effect is that prescribers treat pregabalin as a long-tenure option in chronic pain and epilepsy-adjunct pathways, limiting demand collapse even after generic entry. Where payer policies remain permissive, continued utilization can sustain revenue. Where payers enforce strict formularies or step therapy, price realization compresses quickly, pushing the market toward volume-based generic economics.
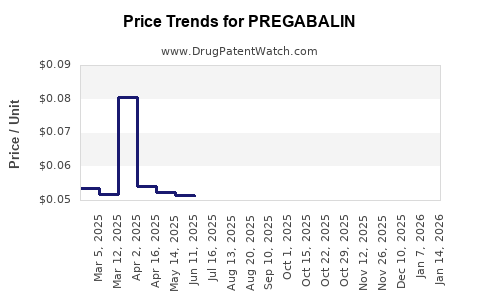
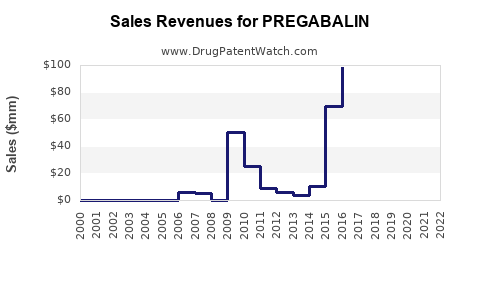
Evidence anchor: originator revenue contraction after generic entry
Genericization is the core driver. Zoetis to do; for pregabalin, the branded incumbent (Pfizer historically for Lyrica) saw revenue erosion as generics entered major markets. The net effect was a revenue curve that falls sharply post-expiry, then flattens as the market becomes price competitive but stable in units.
How did exclusivity and patent expiry shape the revenue curve?
Pregabalin’s market history is governed by exclusivity by region and by how long originator formulations were defended in practice. The business pattern across countries is consistent:
- Brand revenue peaks while protected.
- Patents and regulatory exclusivities expire.
- Generics enter at scale, shifting the market from “branded value” to “generic volume.”
- Residual branded share persists where payer or clinician behavior delays switching, or where branded products keep defensible differentiation (packaging, titration support, or formulary placement in certain formularies).
A simplified lifecycle view:
- Pre-expiry: high pricing power, concentrated originator share
- First generic wave: rapid branded share loss and price compression
- Second and third wave: margin pressure across manufacturers, price floor-like behavior driven by scale
Geographic dynamic
Even when the active molecule is generic in one geography, other geographies can lag due to patent status, line-of-therapy restrictions, and local approvals. The net result is multi-year “trickle” timing in total global revenue.
What is the market dynamic in generics: pricing, volume, and manufacturer mix?
Once pregabalin becomes multi-sourced, the dominant economics shift from “innovation” to “throughput and cost position”:
- Pricing follows manufacturing scale and regional competitive intensity.
- Volume follows formulary coverage and prescribing norms.
- Manufacturer mix shifts toward firms with broad distribution, stable supply, and low unit costs.
Key competitive levers
Pregabalin is not a single-parameter product in payer terms; formulary decisions react to factors that change with competitive pressure:
- Dosage strengths and packaging availability
- Stability, bioequivalence, and switching policies for chronic therapy
- Local tender dynamics for public and institutional buyers
As a result, market value can remain large even when ASPs (average selling prices) fall, because chronic indications maintain use.
What financial trajectory should investors expect under generic equilibrium?
In generic equilibrium, the revenue trajectory becomes less about unit growth and more about price and share of supply. For pregabalin, the expected pattern is:
- Global market revenue grows slowly or flatlines versus units, driven by pricing declines
- Unit demand can be resilient due to chronic use, but it is capped by:
- patient pool saturation
- prescribing guideline changes
- payer restrictions on controlled substances and CNS drugs
- Manufacturer profits compress due to cost competition
Put plainly: after generic entry, the market trades like a commodity with clinical durability.
How do major indications influence payer and clinician behavior?
Pregabalin’s commercial endurance is supported by the way payers and clinicians manage chronic neuropathic pain and epilepsy-adjunct therapy:
- Neuropathic pain is persistent, with switching less frequent once a patient stabilizes (titration and symptom control are key drivers).
- Fibromyalgia use depends on guideline alignment, formulary preference, and tolerance profiles.
- Epilepsy adjunct use is constrained by alternative options and neurologist preference, but the chronic nature supports baseline demand.
This indication structure changes what happens after generic entry. Even when brand pricing collapses, prescribers may keep patients on pregabalin rather than switch purely for cost, especially if tolerability is acceptable.
What are the financial risks to the pregabalin revenue stream?
The principal downside risks after genericization are structural:
- Further pricing pressure from additional generic suppliers
- Payer tightening through step therapy, quantity limits, or preferred product switches
- Shifts in clinical practice toward alternative CNS agents when guidelines evolve
- Supply disruptions or quality incidents at scale suppliers, which can temporarily lift prices but usually invite substitution risk
The counterpoint is that long-term patient adherence can reduce churn, making demand less elastic than acute therapies.
How does the emergence of generics change profitability versus revenue?
Generic market dynamics usually produce a “split” outcome:
- Revenue (market-level) remains material due to sustained volume.
- Manufacturer-level profitability falls as:
- pricing compresses
- promotional spend shifts toward tender wins and rebates
- inventory and working capital needs rise with scale operations
This is why the financial trajectory for the overall molecule can differ from the financial trajectory of any specific manufacturer. A low-cost producer can survive and grow share; a higher-cost producer experiences margin erosion and exit risk.
What does “market stabilization” look like after branded erosion?
Stabilization in a mature generic category typically manifests as:
- ASP decline slows once enough capacity exists and the market reaches a competitive steady state
- Consolidation increases market share for a smaller number of efficient manufacturers
- Share gains favor firms that can secure institutional tenders
For pregabalin, stabilization aligns with the chronic nature of indications and the stable replacement value of older, well-known therapies.
Key business implications by player type
Originator (historically brand incumbent)
- Revenue is structurally capped by generic substitution.
- Value comes from remaining branded share, patient loyalty in certain formularies, and any defensible formulation advantages.
- Post-expiry financial focus shifts from growth to stewardship and cost control.
Generic manufacturers
- Growth is share-driven, not innovation-driven.
- Profitability is a function of unit cost and tender capability.
- Differentiation focuses on supply reliability, formulation portfolio breadth (strength coverage), and packaging that fits prescribing habits.
Payers
- Expect ongoing cost pressure relief after generics dominate.
- Preferential contracting can materially reshape share across manufacturers.
- Chronic utilization supports ongoing negotiations on reimbursement rates.
How does pricing and competition typically evolve in pregabalin across the cycle?
The cycle generally follows this pattern:
- High pricing while brand protected
- Sharp ASP compression in early generic entry
- Gradual ASP normalization after multiple suppliers settle
- Periodic price moves during tender rounds, supply disruptions, or regulatory actions
The molecule’s “financial trajectory” is therefore best interpreted as a sequence:
1) branded revenue collapse,
2) generic value concentration and steady volume,
3) periodic competitive shocks with limited durable uplift.
Market trajectory summary: what matters most next
Near-term market performance for pregabalin depends on whether competitive intensity increases further in key geographies and whether payers tighten access. Long-term performance depends on whether clinical practice shifts away from pregabalin for neuropathic pain or epilepsy-adjunct use, and on the ability of generic suppliers to sustain low-cost supply without quality or supply interruptions.
Key Takeaways
- Pregabalin follows a mature generic lifecycle: branded revenue erosion after exclusivity expiry, then stable chronic utilization with pricing-driven value compression.
- Market dynamics after genericization are dominated by formulary coverage, tender dynamics, and supply scale rather than clinical innovation.
- Investor and business outcomes hinge on manufacturer cost position and distribution access, not on unit growth.
- The molecule’s multi-indication profile supports demand resilience, limiting unit collapse even when ASPs fall.
FAQs
1) Why does pregabalin remain a large-volume drug after generic entry?
Pregabalin is used for chronic neuropathic pain and other long-term indications, which reduces treatment churn and keeps baseline utilization.
2) What most determines pregabalin market revenue after patents expire?
Average selling price realization and share among generic suppliers, driven by tender outcomes and payer switching behavior.
3) Does branded pregabalin disappear immediately after generics launch?
No. Branded share can persist where formulary placement and prescriber habits delay switching, but it typically erodes quickly.
4) What is the biggest financial risk for generic manufacturers of pregabalin?
Margin compression from aggressive pricing and overcapacity, plus operational risks that affect reliable supply.
5) What could change the long-term trajectory of pregabalin demand?
Guideline shifts, payer restrictions on CNS therapies, or meaningful substitution toward competing treatments that reduce pregabalin’s addressable patient pool.
References
[1] Pfizer. Lyrica (pregabalin) Prescribing Information. (Accessed via Pfizer product label).
[2] FDA. Drug Trials Snapshots: Lyrica (pregabalin). U.S. Food and Drug Administration.
[3] EMA. Lyrica: EPAR - Product Information. European Medicines Agency.
[4] IQVIA. Global therapeutic trends and market dynamics reports (methodology and commentary on generic competition and pricing behavior).