LEVOFLOXACIN - Generic Drug Details
✉ Email this page to a colleague
What are the generic drug sources for levofloxacin and what is the scope of patent protection?
Levofloxacin
is the generic ingredient in six branded drugs marketed by Janssen Pharms, Avet Lifesciences, Baxter Hlthcare Corp, Eugia Pharma, Gland, Hospira, Pharmobedient, Rising, Zydus Pharms, Fresenius Kabi Usa, Hikma Farmaceutica, Inforlife, Knack, Santen, Micro Labs Ltd India, Rubicon Research, Sciegen Pharms, Watson Labs Teva, Lannett Co Inc, Novitium Pharma, Apotex Inc, Aurobindo Pharma Ltd, Chartwell Molecular, Chartwell Rx, Dr Reddys Labs Inc, Glenmark Pharms Ltd, Hec Pharm, Hetero Labs Ltd V, Jubilant Generics, Lupin, Macleods Pharms Ltd, Natco Pharma, Orbion Pharms, Sandoz, Teva, Torrent Pharms, Watson Labs Inc, and Zydus Lifesciences, and is included in forty-eight NDAs. Additional information is available in the individual branded drug profile pages.There are thirty-one drug master file entries for levofloxacin. Forty-three suppliers are listed for this compound. There are four tentative approvals for this compound.
Summary for LEVOFLOXACIN
| US Patents: | 0 |
| Tradenames: | 6 |
| Applicants: | 38 |
| NDAs: | 48 |
| Drug Master File Entries: | 31 |
| Finished Product Suppliers / Packagers: | 43 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 318 |
| Patent Applications: | 5,554 |
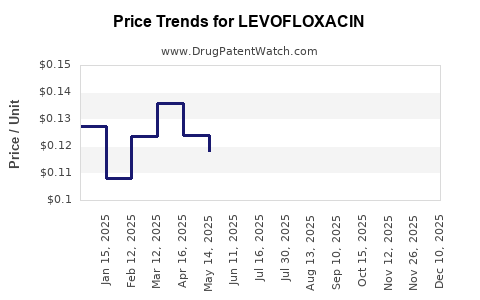
| Drug Prices: | Drug price trends for LEVOFLOXACIN |
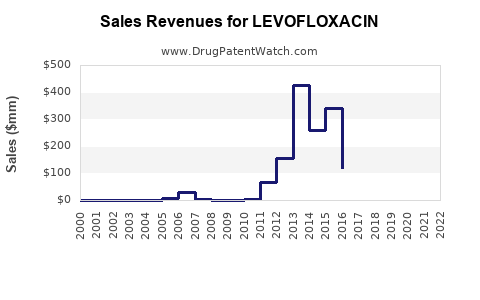
| Drug Sales Revenues: | Drug sales revenues for LEVOFLOXACIN |
| What excipients (inactive ingredients) are in LEVOFLOXACIN? | LEVOFLOXACIN excipients list |
| DailyMed Link: | LEVOFLOXACIN at DailyMed |
Recent Clinical Trials for LEVOFLOXACIN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Institute of Allergy and Infectious Diseases (NIAID) | PHASE3 |
| University of Virginia | PHASE3 |
| Dr. Sardjito Hospital, Yogyakarta | PHASE4 |
Generic filers with tentative approvals for LEVOFLOXACIN
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Start Trial | ⤷ Start Trial | 5MG/ML | INJECTABLE; INJECTION |
| ⤷ Start Trial | ⤷ Start Trial | 750MG | TABLET; ORAL |
| ⤷ Start Trial | ⤷ Start Trial | 500MG | TABLET; ORAL |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Medical Subject Heading (MeSH) Categories for LEVOFLOXACIN
Anatomical Therapeutic Chemical (ATC) Classes for LEVOFLOXACIN
Paragraph IV (Patent) Challenges for LEVOFLOXACIN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LEVAQUIN | Oral Solution | levofloxacin | 25 mg/mL | 021721 | 1 | 2009-07-30 |
US Patents and Regulatory Information for LEVOFLOXACIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Knack | LEVOFLOXACIN IN DEXTROSE 5% IN PLASTIC CONTAINER | levofloxacin | INJECTABLE;INJECTION | 216164-003 | Jan 29, 2024 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Chartwell Molecular | LEVOFLOXACIN | levofloxacin | TABLET;ORAL | 076890-001 | Mar 30, 2012 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Gland | LEVOFLOXACIN | levofloxacin | INJECTABLE;INJECTION | 205540-002 | Apr 22, 2020 | AP | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Teva | LEVOFLOXACIN | levofloxacin | TABLET;ORAL | 076361-002 | Jun 20, 2011 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for LEVOFLOXACIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Santen | QUIXIN | levofloxacin | SOLUTION/DROPS;OPHTHALMIC | 021199-001 | Aug 18, 2000 | 5,053,407 | ⤷ Start Trial |
| Janssen Pharms | LEVAQUIN IN DEXTROSE 5% IN PLASTIC CONTAINER | levofloxacin | INJECTABLE;INJECTION | 020635-003 | Dec 20, 1996 | 4,382,892 | ⤷ Start Trial |
| Janssen Pharms | LEVAQUIN | levofloxacin | INJECTABLE;INJECTION | 020635-001 | Dec 20, 1996 | 4,382,892 | ⤷ Start Trial |
| Janssen Pharms | LEVAQUIN | levofloxacin | TABLET;ORAL | 020634-002 | Dec 20, 1996 | 4,382,892 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for LEVOFLOXACIN
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Chiesi Farmaceutici S.p.A | Quinsair | levofloxacin | EMEA/H/C/002789Quinsair is indicated for the management of chronic pulmonary infections due to Pseudomonas aeruginosa in adult patients with cystic fibrosis.Consideration should be given to official guidance on the appropriate use of antibacterial agents. | Authorised | no | no | no | 2015-03-25 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |