Last updated: February 19, 2026
Levofloxacin, marketed as LEVAQUIN by Johnson & Johnson, is a broad-spectrum fluoroquinolone antibiotic. Its market performance has been shaped by patent expirations, generic competition, and evolving clinical guidelines. This analysis examines LEVAQUIN's historical market penetration, patent landscape, and projected financial trajectory.
What is Levofloxacin's Therapeutic Class and Mechanism of Action?
Levofloxacin is a synthetic chemotherapeutic antibacterial agent. It is the L-isomer of ofloxacin. Levofloxacin acts by inhibiting bacterial DNA gyrase and topoisomerase IV, enzymes essential for bacterial DNA replication, transcription, repair, and recombination. This mechanism leads to rapid bacterial cell death.
Levofloxacin is indicated for the treatment of various bacterial infections, including:
- Pneumonia (community-acquired, hospital-acquired)
- Acute bacterial sinusitis
- Acute bacterial exacerbations of chronic bronchitis
- Urinary tract infections (complicated and uncomplicated)
- Chronic bacterial prostatitis
- Skin and skin structure infections (complicated and uncomplicated)
- Inhalation anthrax (post-exposure prophylaxis)
- Plague (treatment and prophylaxis) [1, 2]
What is LEVAQUIN's Patent and Exclusivity Landscape?
The original composition of matter patent for levofloxacin has long expired, enabling generic competition. Key patent and exclusivity milestones relevant to LEVAQUIN include:
- Original Composition of Matter Patent: Granted in the 1980s, this patent provided the initial market exclusivity.
- Orphan Drug Exclusivity: Not applicable to LEVAQUIN as it was not developed for a rare disease.
- New Chemical Entity (NCE) Exclusivity: Levofloxacin, as a distinct entity from ofloxacin, may have received NCE exclusivity upon its initial approval.
- Pediatric Exclusivity: An extension of 6 months was granted for studies conducted in pediatric populations, extending exclusivity until the early 2000s.
- Method of Use Patents: Johnson & Johnson pursued patents covering specific uses, dosages, or formulations of levofloxacin. These patents faced numerous legal challenges and expirations.
The primary patent protection for the core molecule expired in the early 2000s. Subsequent patents related to specific formulations or methods of treatment also faced expiration or were invalidated through litigation. This opened the door for significant generic market entry.
How Has LEVAQUIN's Market Penetration Evolved?
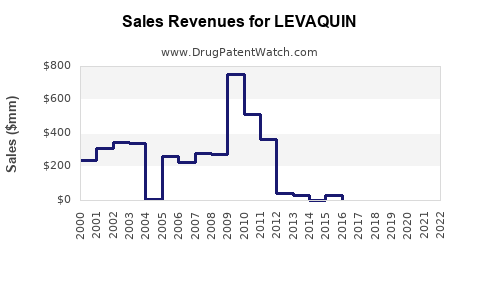
LEVAQUIN achieved substantial market penetration following its introduction in 2000. Its broad spectrum of activity and favorable pharmacokinetic profile made it a preferred agent for numerous bacterial infections.
- Peak Sales: LEVAQUIN’s peak sales were in the mid-to-late 2000s, exceeding $1 billion annually. In 2010, it generated approximately $1.3 billion in revenue for Johnson & Johnson. [3]
- Market Share: At its peak, LEVAQUIN held a significant share within the fluoroquinolone market and the broader antibiotic market for its approved indications.
- Generic Entry Impact: Following patent expirations around 2008-2010, multiple generic versions of levofloxacin entered the market. This led to a rapid decline in LEVAQUIN's market share and average selling price.
Comparative Market Performance: LEVAQUIN vs. Other Fluoroquinolones (Peak Year Example)
| Drug |
Peak Annual Sales (Approx.) |
Year of Peak |
Primary Indication |
| LEVAQUIN |
$1.3 billion |
2010 |
Pneumonia, UTIs, Sinusitis, Skin Infections |
| Cipro (Ciprofloxacin) |
$1.5 billion |
Early 2000s |
UTIs, Anthrax, Bacterial Gastroenteritis |
| Avelox (Moxifloxacin) |
$800 million |
Mid-2010s |
Pneumonia, Sinusitis, Skin Infections |
Note: Peak sales figures are approximate and can vary based on reporting sources and market fluctuations.
What Are the Key Market Drivers and Restraints for Levofloxacin?
Market Drivers:
- Efficacy Against Resistant Organisms: Levofloxacin demonstrates activity against a range of Gram-positive and Gram-negative bacteria, including some strains resistant to older antibiotics.
- Broad Spectrum of Activity: Its broad spectrum simplifies treatment regimens for complicated infections where the causative pathogen is not yet identified.
- Availability of Generic Versions: The presence of affordable generic levofloxacin ensures continued accessibility for healthcare providers and patients.
- Established Clinical Use: Decades of clinical experience and integration into treatment guidelines contribute to its continued use.
Market Restraints:
- Safety Concerns and Black Box Warnings: Fluoroquinolones, including levofloxacin, carry significant safety warnings regarding serious adverse events, such as tendonitis and tendon rupture, peripheral neuropathy, central nervous system effects (including CNS depression and toxicity), and exacerbation of myasthenia gravis. The U.S. Food and Drug Administration (FDA) has strengthened these warnings over time. [4, 5]
- Emergence of Resistance: Like all antibiotics, overuse and misuse of levofloxacin contribute to the development of antibiotic resistance, limiting its efficacy in certain settings.
- Stricter Prescribing Guidelines: Regulatory bodies and professional organizations recommend limiting fluoroquinolone use, particularly for uncomplicated infections, due to the risk of serious side effects and the availability of safer alternatives.
- Competition from Newer Antibiotics: Development of new antibiotic classes and agents with potentially improved safety profiles or activity against resistant pathogens presents competitive pressure.
- Declining Brand Value: Genericization significantly erodes the premium pricing and market dominance previously held by LEVAQUIN.
What is the Current and Projected Financial Trajectory of Levofloxacin?
The financial trajectory of levofloxacin has shifted from blockbuster brand sales to a mature generic market.
- Generic Market Dominance: The market is now dominated by generic levofloxacin. Brand-name LEVAQUIN sales have drastically diminished.
- Price Erosion: Generic competition has led to substantial price erosion, with levofloxacin tablets and solutions available at a fraction of the original brand price.
- Market Volume: While dollar sales have decreased significantly, the overall volume of levofloxacin prescribed and dispensed remains substantial due to its affordability and continued clinical utility for specific indications.
- Projected Revenue: Projections for levofloxacin as a global market focus on the aggregate sales of all levofloxacin generics. The total market value is expected to remain relatively stable or see modest growth, driven by volume in emerging markets and specific treatment needs, rather than significant price appreciation. The market size for levofloxacin generics is estimated to be in the hundreds of millions of U.S. dollars annually, a stark contrast to LEVAQUIN's peak brand revenue. [6]
Key Financial Indicators (Post-Patent Expiration)
| Indicator |
Description |
Trend |
| Brand Sales (LEVAQUIN) |
Revenue generated by Johnson & Johnson from the branded product. |
Drastically declined post-patent expiration. |
| Generic Levofloxacin Sales |
Aggregate revenue from all manufacturers producing generic levofloxacin. |
Primary revenue driver for the molecule. |
| Average Selling Price (ASP) |
The average price at which levofloxacin is sold, considering both branded and generic products. |
Significantly decreased due to generic competition. |
| Market Volume (Units) |
The total number of prescriptions or units dispensed for levofloxacin. |
Remains substantial, supported by generic availability. |
| Market Share |
LEVAQUIN's share of the fluoroquinolone market. |
Minimal for branded LEVAQUIN, high for generic levofloxacin. |
What are the Regulatory and Clinical Landscape Considerations?
Regulatory actions and evolving clinical practices profoundly impact levofloxacin's market position.
- FDA Safety Communications: The FDA has issued multiple safety alerts and strengthened black box warnings for fluoroquinolones due to serious adverse events. These include warnings about disabling and potentially permanent side effects involving tendons, muscles, joints, nerves, and the central nervous system. [4, 5]
- Restrictions on Use: In 2018, the FDA advised against using fluoroquinolones for patients with uncomplicated sinusitis, acute bacterial exacerbations of chronic bronchitis, and uncomplicated urinary tract infections when other treatment options are available. [5] This recommendation aims to reduce the risk of serious side effects.
- Antimicrobial Stewardship Programs: Healthcare institutions increasingly implement antimicrobial stewardship programs that promote the judicious use of antibiotics, including fluoroquinolones, favoring agents with better safety profiles or narrower spectra of activity when appropriate.
- International Regulations: Similar scrutiny and evolving guidance exist in other major markets, influencing prescribing patterns and market access.
Key Takeaways
- LEVAQUIN (levofloxacin) transitioned from a blockbuster branded antibiotic to a mature generic product following patent expirations in the early 2000s.
- Generic competition has led to significant price erosion, with aggregate global sales of levofloxacin generics now in the hundreds of millions of dollars annually.
- Significant safety concerns, including black box warnings for serious adverse events, have led to restricted prescribing guidelines by regulatory bodies and a trend towards using levofloxacin only when other treatment options are inadequate.
- The continued market presence of levofloxacin is driven by its efficacy against a broad range of pathogens and the availability of affordable generic formulations, but its future growth is constrained by safety considerations and the emergence of newer therapeutic alternatives.
Frequently Asked Questions
-
What is the current U.S. market status for branded LEVAQUIN versus generic levofloxacin?
Branded LEVAQUIN has minimal market presence in the U.S. The market is overwhelmingly dominated by generic levofloxacin products.
-
What are the primary reasons for the decline in LEVAQUIN's brand value?
The primary reasons are patent expiration, leading to intense generic competition, and the associated significant price erosion.
-
How do the FDA's safety warnings affect the current prescribing of levofloxacin?
The FDA's warnings, particularly the black box warnings, advise against using fluoroquinolones like levofloxacin for uncomplicated infections when alternative treatments exist, thereby limiting its use to more severe or complicated conditions.
-
Are there any new patents being filed or considered for levofloxacin that could impact its market exclusivity?
Given the age of the core compound, new composition of matter patents are highly unlikely. Any future patent filings would likely pertain to novel formulations, delivery systems, or specific combination therapies, which would require significant innovation to gain market exclusivity and would face a high bar for approval.
-
What is the projected outlook for the global levofloxacin market in the next five years?
The global levofloxacin market, comprised of generics, is projected to remain relatively stable, with potential for modest growth driven by increased volume in developing markets and its continued use in specific indications. However, significant price increases are not anticipated due to established generic competition and ongoing safety scrutiny.
Citations
[1] Product Information: LEVAQUIN® (levofloxacin) Tablets. Ortho-McNeil Pharmaceutical, Inc. Revision Date: July 2010. (Archived Label Information)
[2] FDA. (2017, July 19). Levofloxacin Tablets, Oral Solution. DailyMed. Retrieved from https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=69870880-615f-4408-8f4f-a4124f6287e0
[3] Johnson & Johnson. (2011). 2010 Annual Report.
[4] FDA. (2018, December 11). FDA strengthens warnings on fluoroquinolone antibiotics. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-strengthens-warnings-fluoroquinolone-antibiotics
[5] FDA. (2018, December 6). FDA advises restricting fluoroquinolone antibiotic use for uncomplicated sinus and urinary tract infections. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-advises-restricting-fluoroquinolone-antibiotic-use-uncomplicated-sinus-and-urinary-tract-infections
[6] Market Research Reports (Various Sources, e.g., Grand View Research, Mordor Intelligence, Statista - aggregated data for global levofloxacin market size). Specific report dates and figures vary; general consensus on market size in the hundreds of millions USD range post-genericization.