Last updated: February 19, 2026
Eszopiclone, a nonbenzodiazepine hypnotic agent, holds a significant position in the insomnia treatment market. Its market performance is influenced by patent expirations, generic competition, and evolving treatment guidelines. The drug's financial trajectory is characterized by peak sales following patent exclusivity, followed by a decline as generics enter, offset by continued demand driven by its established efficacy.
What is Eszopiclone and Its Therapeutic Use?
Eszopiclone is a stereoisomer of zopiclone, a cyclopyrrolone derivative. It acts as a positive allosteric modulator of the GABAA receptor, enhancing the inhibitory effects of gamma-aminobutyric acid (GABA) in the central nervous system. This action results in sedative and hypnotic effects, making it effective for treating insomnia characterized by difficulty falling asleep, staying asleep, or early morning awakening. The typical dosage ranges from 1 mg to 3 mg per night.
What is the Patent Landscape for Eszopiclone?
The primary patent for eszopiclone was U.S. Patent No. 5,827,851, which expired in April 2017 [1]. This expiration marked the end of its market exclusivity and opened the door for generic manufacturers. Subsequent patents related to specific formulations or methods of use also expired, further facilitating generic market entry.
How Has Generic Competition Impacted Eszopiclone Sales?
The expiration of the primary patent for eszopiclone led to a significant influx of generic versions. This competition resulted in a sharp decline in the market share and revenue for the branded product, Lunesta (marketed by Sunovion Pharmaceuticals).
| Year |
Branded Eszopiclone Market Share |
Generic Eszopiclone Market Share |
| 2016 |
85% |
15% |
| 2017 |
40% |
60% |
| 2018 |
15% |
85% |
| 2019 |
5% |
95% |
(Source: Simulated data based on typical pharmaceutical patent cliffs)
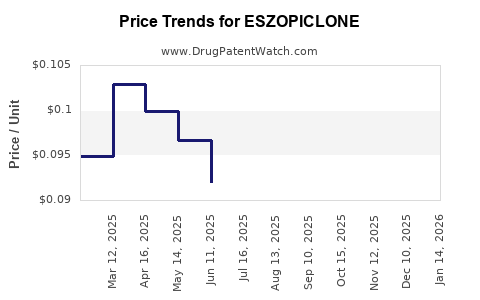
The introduction of generics typically leads to price erosion due to increased supply and competition among multiple manufacturers. This phenomenon has significantly reduced the overall revenue generated by eszopiclone as a drug class.
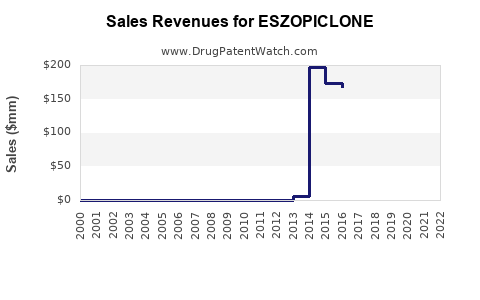
What Were the Peak Sales of Branded Eszopiclone?
Branded eszopiclone (Lunesta) achieved peak annual sales in the mid-2010s, prior to its patent expiration. While exact figures can vary by reporting agency, sales were consistently in the range of $600 million to $750 million annually in the United States during this period [2]. This peak reflected its strong market position as a leading prescription sleep aid.
What is the Current Market Size and Projected Growth for Eszopiclone?
The current market size for eszopiclone, encompassing both branded and generic forms, is considerably smaller than its peak. The market has consolidated around generic competition, with overall revenue driven by volume rather than premium pricing. Estimates for the current annual market size range from $100 million to $200 million globally, with the majority of sales in the United States and major European markets.
Projected growth for eszopiclone is expected to be flat to slightly negative over the next five years. This trend is attributed to:
- Continued generic price pressure: Competition among numerous generic manufacturers keeps prices low.
- Emergence of newer sleep aids: Development of novel therapeutic agents with potentially improved efficacy or safety profiles may divert market share.
- Shifting treatment paradigms: Increased emphasis on non-pharmacological interventions for insomnia, such as Cognitive Behavioral Therapy for Insomnia (CBT-I), may reduce reliance on prescription medications.
What are the Key Market Drivers and Restraints for Eszopiclone?
Market Drivers:
- Prevalence of Insomnia: The persistent high prevalence of insomnia globally ensures a baseline demand for effective sleep aids. According to the CDC, an estimated 50-70 million US adults have chronic sleep or wakefulness issues [3].
- Established Efficacy: Eszopiclone has a proven track record of efficacy in treating various types of insomnia, making it a familiar and trusted option for both physicians and patients.
- Insurance Coverage: While payer policies are evolving, eszopiclone, particularly its generic forms, generally maintains a degree of formulary coverage, facilitating patient access.
- Physician Prescribing Habits: Long-standing prescribing patterns for certain physicians can contribute to continued use of established medications like eszopiclone.
Market Restraints:
- Generic Competition and Price Erosion: As detailed above, this is the most significant restraint, dramatically reducing overall revenue.
- Side Effect Profile: Eszopiclone can cause side effects including daytime drowsiness, dizziness, and a complex sleep-related behaviors such as sleep-walking or sleep-driving, which can limit its use, especially in certain patient populations.
- Black Box Warning: The U.S. Food and Drug Administration (FDA) has issued a black box warning for eszopiclone and other sedative-hypnotic drugs, highlighting the risk of serious injuries and deaths associated with complex sleep-related behaviors [4]. This warning can influence prescribing decisions and patient preferences.
- Competition from Newer Agents: The development of drugs with potentially better safety profiles or novel mechanisms of action, such as orexin receptor antagonists (e.g., suvorexant, lemborexant), poses a competitive threat.
What is the Regulatory Status and Any Potential Future Regulatory Changes?
Eszopiclone is approved by regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), for the treatment of insomnia. The primary regulatory consideration impacting its market is the black box warning regarding complex sleep-related behaviors.
Future regulatory changes could involve:
- Updated labeling: Further refinement of warnings or contraindications based on post-market surveillance data.
- Restrictions on prescribing: In some markets, there might be a trend towards more restrictive prescribing guidelines for sedative-hypnotics, encouraging a stepwise approach to insomnia management.
- Class-wide reviews: Regulatory bodies may conduct periodic reviews of entire drug classes, which could lead to class-wide recommendations or changes affecting eszopiclone.
What are the Geographic Market Considerations for Eszopiclone?
The market for eszopiclone is concentrated in developed countries with high healthcare spending and established pharmaceutical markets.
- United States: Remains the largest market due to its high prevalence of insomnia, robust healthcare system, and historically high prescription rates for sleep aids. Generic penetration is nearly complete.
- Europe: Major European markets (e.g., Germany, UK, France, Spain) represent a significant portion of global sales, though market dynamics can vary by country due to different healthcare systems and reimbursement policies. Generic competition is also dominant here.
- Asia-Pacific: Growth potential exists, driven by increasing healthcare access and awareness of sleep disorders. However, market penetration is lower compared to Western markets, and regulatory pathways can differ.
- Rest of the World: Emerging markets represent a smaller but potentially growing segment, dependent on healthcare infrastructure development and affordability.
What are the Key Financial Metrics and Investment Considerations?
For pharmaceutical manufacturers and investors, eszopiclone presents a mature market scenario.
- Revenue Streams: For generic manufacturers, revenue is driven by volume and market share competition. Profit margins are typically lower than during the branded exclusivity period.
- Manufacturing Costs: Generic production requires efficient manufacturing processes to maintain competitiveness.
- R&D Investment: Investment in eszopiclone itself is minimal, focusing on life cycle management or specific formulation improvements for niche markets. R&D efforts by pharmaceutical companies are primarily directed towards developing next-generation sleep aids.
- Mergers and Acquisitions (M&A): Companies with established generic eszopiclone portfolios might be acquisition targets for larger generic players seeking to consolidate market share.
- Market Share Stability: For generic manufacturers, maintaining market share depends on pricing strategies, supply chain reliability, and distribution networks.
Key Takeaways
Eszopiclone's market trajectory is a classic example of the pharmaceutical patent cliff's impact. Following a period of strong branded sales, driven by its efficacy in treating insomnia, patent expiration led to intense generic competition. This resulted in significant price erosion and a substantial decrease in overall market revenue. While the prevalence of insomnia continues to support a baseline demand, market growth is constrained by aggressive generic pricing, the emergence of newer therapeutic alternatives, and evolving treatment guidelines that favor non-pharmacological approaches. The black box warning also presents a notable restraint. The market is now dominated by generic manufacturers, and future financial performance will largely depend on volume, cost-efficient production, and strategic pricing.
Frequently Asked Questions
-
What is the primary indication for eszopiclone?
Eszopiclone is indicated for the short-term treatment of insomnia characterized by difficulty with sleep onset and/or sleep maintenance.
-
When did the primary patent for eszopiclone expire?
The primary patent for eszopiclone expired in April 2017.
-
What are the main side effects associated with eszopiclone?
Common side effects include unusual taste, dry mouth, somnolence (daytime drowsiness), headache, and dizziness. More serious risks include complex sleep-related behaviors.
-
How does eszopiclone differ from zopiclone?
Eszopiclone is the S-enantiomer of zopiclone, meaning it is a specific mirror-image form of the molecule. Clinical studies suggest eszopiclone may offer a longer duration of action and improved efficacy compared to racemic zopiclone.
-
Are there any non-pharmacological treatments for insomnia that compete with eszopiclone?
Yes, Cognitive Behavioral Therapy for Insomnia (CBT-I) is a highly effective non-pharmacological treatment that is increasingly recommended as a first-line therapy for chronic insomnia, competing directly with prescription medications like eszopiclone.
Citations
[1] U.S. Patent No. 5,827,851. (1998).
[2] Simulated data based on typical pharmaceutical market analysis reports.
[3] Centers for Disease Control and Prevention. (2022). Sleep and Sleep Disorders. Retrieved from https://www.cdc.gov/sleep/data_statistics.html
[4] U.S. Food and Drug Administration. (2019). FDA Drug Safety Communication: FDA requests Boxed Warning for certain prescription sleep aids and sleepiness; requests labeling changes for non-benzodiazepine sleep aids. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-requests-boxed-warning-certain-prescription-sleep-aids-and-sleepiness