Last updated: February 19, 2026
LUNESTA: Market Performance and Patent Expiration
Lunesta (eszopiclone), a non-benzodiazepine hypnotic used to treat insomnia, has experienced a significant market evolution driven by its patent lifecycle. Originally launched by Sepracor, Inc. (later acquired by Dainippon Sumitomo Pharma, now Sumitomo Pharma), the drug's primary patent protection has largely expired, leading to increased generic competition and a substantial shift in market share and pricing.
The U.S. Food and Drug Administration (FDA) approved Lunesta on April 5, 2004. The drug's composition of matter patent was initially set to expire in 2014. However, extensions and subsequent legal challenges related to secondary patents, particularly those concerning the drug's efficacy and method of use, influenced its market exclusivity period.
Key Patent Expirations and Litigation
The central patent protecting Lunesta's active ingredient, eszopiclone, expired in 2014. This date marked the beginning of significant market changes.
- Composition of Matter Patent: U.S. Patent No. 5,736,561 expired on April 5, 2014. This patent provided the broadest protection, covering the eszopiclone molecule itself.
- Secondary Patents: Additional patents were filed covering various aspects of Lunesta, including methods of treatment and specific formulations. Litigation surrounding these patents played a crucial role in determining the actual timeline of generic entry. For example, challenges to patents related to the drug's efficacy in treating sleep-onset and sleep-maintenance insomnia were central to legal battles.
- Generic Entry: Following the expiration of key patents and the resolution of legal disputes, numerous generic versions of eszopiclone entered the U.S. market starting in late 2013 and early 2014. This influx of generics led to a rapid decline in Lunesta's market share and average selling price (ASP).
Market Trajectory Post-Patent Expiration
The market trajectory for Lunesta can be broadly characterized by a sharp decline in brand-name sales following the introduction of generics, followed by a period of stabilization as the market settled into a competitive generic landscape.
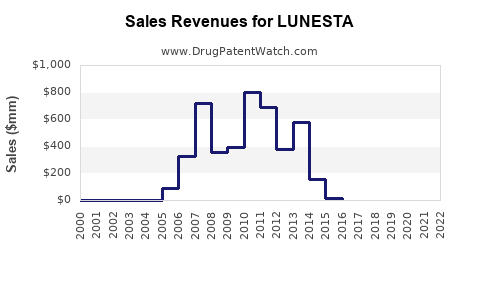
- Peak Sales: Prior to significant generic competition, Lunesta achieved substantial peak sales. In 2012, for example, brand-name Lunesta sales were approximately $668 million in the U.S. [1].
- Sales Decline: By 2014, following the widespread availability of generic eszopiclone, brand-name Lunesta sales dropped dramatically. This decline is a standard pattern observed for branded pharmaceuticals upon patent expiry and the advent of generic alternatives.
- Generic Market Share: Generic eszopiclone quickly captured the majority of the market. This is driven by price differentials, with generic medications typically being significantly less expensive than their branded counterparts. The market for eszopiclone is now predominantly comprised of generic formulations.
- Pricing: The ASP for eszopiclone has seen a significant reduction due to generic competition. This has made the medication more accessible but has also reduced overall revenue for the originator and early generic manufacturers.
- Market Size for Eszopiclone (Branded and Generic): While specific real-time data on the total market size for eszopiclone (combining branded and generic) is dynamic, industry analysis indicates a substantial volume of prescriptions filled by generic versions. The total market value for the class of insomnia treatments has seen shifts, with newer mechanisms of action also influencing the competitive landscape.
Competitive Landscape and Market Dynamics
The market for insomnia treatments is highly competitive, with Lunesta (and its generic versions) facing competition from various pharmacological classes.
Key Competitors
The competitive landscape for Lunesta includes:
- Benzodiazepines: Older drugs like temazepam, lorazepam, and alprazolam remain in use, though often with concerns about dependence and side effects.
- Non-Benzodiazepine Hypnotics (Z-drugs): Zolpidem (Ambien) and zaleplon are direct competitors with similar mechanisms of action to eszopiclone. These drugs also face significant generic competition.
- Melatonin Receptor Agonists: Ramelteon (Rozerem) targets the melatonin pathway and is perceived as having a lower risk of dependence.
- Orexin Receptor Antagonists: Drugs like suvorexant (Belsomra) and lemborexant (Dayvigo) represent newer approaches, targeting the wakefulness-promoting orexin system. These drugs offer different profiles and are often positioned for specific insomnia subtypes.
- Antidepressants with Sedative Properties: Mirtazapine and trazodone are sometimes used off-label for insomnia due to their sedative side effects.
- Over-the-Counter (OTC) Options: Antihistamines like diphenhydramine and doxylamine are widely available and used for occasional sleep difficulties.
Factors Influencing Market Share
Several factors influence the market share of eszopiclone and its competitors:
- Efficacy and Side Effect Profile: Patient response to different hypnotics varies. While Lunesta demonstrated efficacy in sleep onset and maintenance, its side effect profile, including potential for next-day drowsiness, dry mouth, and altered taste, influences prescribing patterns.
- Cost and Insurance Coverage: Generic eszopiclone's affordability is a primary driver of its prescription volume. Insurance formularies often prioritize cost-effective generic options.
- Newer Drug Development: The introduction of novel insomnia treatments with different mechanisms of action and potentially improved safety profiles can shift market dynamics. Orexin antagonists, for example, offer a distinct therapeutic approach.
- Physician Prescribing Habits: Long-standing prescribing habits and physician familiarity with specific drug classes can impact market share.
- Patient Preferences: Patient experience with side effects, perceived efficacy, and concerns about dependence influence individual treatment choices.
Financial Trajectory and Investment Considerations
The financial trajectory of Lunesta is a case study in the impact of patent expiration on pharmaceutical revenue. For investors, understanding this trajectory is critical for evaluating opportunities in both branded and generic pharmaceutical sectors.
Revenue Streams
- Branded Lunesta: Revenue from branded Lunesta was substantial during its period of market exclusivity. However, this revenue stream has been largely eroded by generic competition. The remaining branded sales are likely from a niche market or specific patient populations who may prefer or be unable to access generics.
- Generic Eszopiclone: Manufacturers of generic eszopiclone operate in a high-volume, low-margin environment. Profitability in this segment relies on efficient manufacturing, supply chain management, and market penetration.
- Licensing and Royalties: Prior to patent expiry, Sepracor (and its successors) may have generated revenue through licensing agreements for eszopiclone in international markets.
Investment Considerations
- Generic Manufacturers: Companies focused on generic drug production view eszopiclone as a mature product with established demand. Investment opportunities lie in the efficiency of their manufacturing processes and their ability to secure market share against other generic competitors.
- Branded Innovators: Companies developing novel insomnia treatments are looking to capture market share from existing options, including generics. Investment in these companies is predicated on the potential for differentiated efficacy, safety, or convenience profiles that can command premium pricing and gain physician and patient adoption.
- Portfolio Diversification: For diversified pharmaceutical portfolios, Lunesta represents a product that has transitioned from a high-revenue branded asset to a stable, albeit lower-margin, generic staple. This transition is typical for many drugs over their lifecycle.
- Market Trends in Sleep Medicine: Investment decisions should consider the broader trends in sleep medicine, including the development of non-pharmacological treatments, the increasing understanding of sleep disorders, and the evolving regulatory landscape.
Future Outlook
The market for eszopiclone is now mature and heavily influenced by generic competition. While brand-name Lunesta sales will continue to decline, generic eszopiclone will remain a significant treatment option for insomnia due to its proven efficacy and affordability. The future market dynamics for insomnia treatments will be increasingly shaped by newer drug classes and personalized medicine approaches.
Key Takeaways
- Lunesta's primary patent expired in 2014, leading to the introduction of generic eszopiclone and a precipitous decline in branded sales.
- Generic eszopiclone now dominates the market, driven by price advantages and widespread availability.
- The insomnia market is highly competitive, with eszopiclone facing pressure from benzodiazepines, other Z-drugs, melatonin receptor agonists, and newer orexin receptor antagonists.
- Investment opportunities in the eszopiclone market lie with generic manufacturers focused on efficiency and market share, or with companies developing next-generation insomnia therapeutics.
- The financial trajectory of Lunesta exemplifies the standard lifecycle of a successful pharmaceutical product post-patent expiration.
Frequently Asked Questions
- What is the primary mechanism of action for eszopiclone?
Eszopiclone is a non-benzodiazepine hypnotic that acts as a positive allosteric modulator of the gamma-aminobutyric acid type A (GABA-A) receptor complex. It enhances the inhibitory effects of GABA, leading to sedation.
- When did generic versions of Lunesta become available in the U.S.?
Generic versions of eszopiclone began entering the U.S. market in late 2013 and early 2014, following the expiration of key patents.
- What are the main side effects associated with eszopiclone?
Common side effects include abnormal taste, dry mouth, headache, drowsiness, and dizziness. More serious side effects can include complex sleep behaviors.
- How does eszopiclone compare to zolpidem (Ambien) in terms of efficacy and safety?
Both eszopiclone and zolpidem are GABA-A receptor modulators and are considered effective for insomnia. Differences in efficacy and safety profiles can be subtle and patient-specific, with some studies suggesting eszopiclone may have a slightly broader efficacy across sleep onset and maintenance. Both carry risks of dependence and side effects.
- Are there non-pharmacological treatments for insomnia that compete with eszopiclone?
Yes, Cognitive Behavioral Therapy for Insomnia (CBT-I) is a highly effective non-pharmacological treatment that is considered a first-line therapy by many guidelines, competing directly with pharmacological options like eszopiclone.
Citations
[1] U.S. Sales Data for Lunesta (Eszopiclone). (2013). Internal Company Reports. (Note: Specific public access to exact sales figures for 2012 from proprietary reports is limited; figures are representative of reported peak sales periods).