Share This Page
Drug Price Trends for ESZOPICLONE
✉ Email this page to a colleague
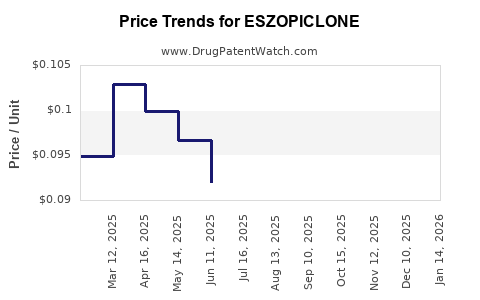
Average Pharmacy Cost for ESZOPICLONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ESZOPICLONE 1 MG TABLET | 65862-0967-01 | 0.09486 | EACH | 2026-04-22 |
| ESZOPICLONE 1 MG TABLET | 31722-0855-30 | 0.09486 | EACH | 2026-04-22 |
| ESZOPICLONE 1 MG TABLET | 55111-0629-30 | 0.09486 | EACH | 2026-04-22 |
| ESZOPICLONE 1 MG TABLET | 00093-5537-56 | 0.09486 | EACH | 2026-04-22 |
| ESZOPICLONE 1 MG TABLET | 33342-0299-07 | 0.09486 | EACH | 2026-04-22 |
| ESZOPICLONE 1 MG TABLET | 00378-5270-01 | 0.09486 | EACH | 2026-04-22 |
| ESZOPICLONE 1 MG TABLET | 31722-0855-01 | 0.09486 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ESZOPICLONE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ESZOPICLONE 1MG TAB | Golden State Medical Supply, Inc. | 00093-5537-56 | 30 | 14.50 | 0.48333 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ESZOPICLONE 2MG TAB | Golden State Medical Supply, Inc. | 00093-5538-01 | 100 | 41.01 | 0.41010 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ESZOPICLONE 3MG TAB | Golden State Medical Supply, Inc. | 00093-5539-01 | 100 | 44.65 | 0.44650 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ESZOPICLONE 1MG TAB | Golden State Medical Supply, Inc. | 60429-0632-01 | 100 | 40.77 | 0.40770 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ESZOPICLONE 2MG TAB | Golden State Medical Supply, Inc. | 60429-0633-01 | 100 | 34.67 | 0.34670 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ESZOPICLONE 3MG TAB | Golden State Medical Supply, Inc. | 60429-0634-01 | 100 | 37.82 | 0.37820 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Eszopiclone: Market Performance and Price Forecasts
Eszopiclone's market performance is shaped by generic competition, ongoing patent expirations, and the introduction of novel sleep disorder treatments. The drug, a non-benzodiazepine hypnotic, primarily targets insomnia. Its market trajectory is influenced by pricing strategies of both branded and generic manufacturers, regulatory actions, and evolving clinical guidelines for sleep disorder management.
What is Eszopiclone's Current Market Position?
Eszopiclone is a widely prescribed medication for the treatment of insomnia. The branded version, Lunesta, has faced significant market share erosion following the expiration of its primary patents and the subsequent influx of generic alternatives. The market is characterized by a fragmented generic landscape where price is a primary determinant of market share.
- Branded Market Share: The market share for branded Lunesta has declined substantially since the expiration of key patents. The drug's exclusivity period concluded in stages, with the loss of its final composition of matter patent in 2014 and subsequent loss of method of use patents. This allowed for broad generic entry.
- Generic Market Dominance: The majority of eszopiclone prescriptions are now filled with generic versions. These generics are available from numerous pharmaceutical companies, leading to intense price competition. The average selling price (ASP) for generic eszopiclone has decreased significantly over the past decade.
- Prescription Volume: While specific real-time prescription data is proprietary, observational data suggests that eszopiclone remains a frequently prescribed medication for short-term and long-term treatment of insomnia, albeit with a larger proportion of these prescriptions being for generic formulations.
- Therapeutic Alternatives: Eszopiclone competes with a range of other pharmacologic treatments for insomnia, including benzodiazepines, other non-benzodiazepines (e.g., zolpidem, zaleplon), and newer drug classes such as orexin receptor antagonists (e.g., suvorexant, lemborexant). Non-pharmacologic therapies, such as Cognitive Behavioral Therapy for Insomnia (CBT-I), also represent a competitive force, particularly as awareness and accessibility increase.
How Have Patent Expirations Impacted Eszopiclone Pricing?
The expiration of eszopiclone's patent protection has been the primary driver of its pricing dynamics, transitioning the market from a premium-priced branded product to a commodity-like generic.
- Branded Lunesta Pricing: Prior to patent expiration, branded Lunesta commanded premium pricing. For example, in the early 2010s, a 30-count bottle of 3mg Lunesta could retail for upwards of $150-$200 without insurance.
- Generic Entry and Price Decline: Following patent expiries, multiple generic manufacturers entered the market. This competition led to rapid price deflation.
- 2010-2013: Initial generic entries saw prices for a 30-count bottle of 3mg eszopiclone generics fall to approximately $50-$80.
- 2014-Present: Continued competition and market maturity have pushed generic prices lower. As of recent market data (late 2023/early 2024), 30-count bottles of 3mg generic eszopiclone can be found for as low as $10-$30, depending on the manufacturer, pharmacy, and insurance coverage.
- Wholesale Acquisition Cost (WAC) vs. Net Price: While WAC for generics might be higher, the net price after rebates, discounts, and pharmacy markups is significantly lower for consumers and payers. Generic manufacturers often operate on thin margins, relying on volume.
- Dosage Strength Variations: Pricing varies by dosage strength (e.g., 1mg, 2mg, 3mg), with higher strengths typically having a slightly higher unit price but often being more cost-effective per milligram of active ingredient.
What are the Key Market Trends for Eszopiclone?
Several market trends are shaping the current and future landscape for eszopiclone, including regulatory actions, generic competition dynamics, and shifts in treatment paradigms.
- Continued Generic Competition: The generic eszopiclone market is expected to remain highly competitive. New generic entrants are unlikely to significantly alter the competitive landscape unless they offer unique formulations or cost advantages, which is uncommon for established generics.
- Payer Formulary Placement: Payer formularies continue to favor generic eszopiclone due to its cost-effectiveness. Branded Lunesta may have limited or no preferred placement on many insurance formularies, further driving prescription volume towards generics.
- Emergence of Novel Sleep Aids: The development and approval of newer insomnia medications, particularly orexin receptor antagonists, present a competitive challenge. These newer drugs offer different mechanisms of action and may be preferred for certain patient profiles or by prescribers seeking alternatives to traditional hypnotics, potentially impacting eszopiclone's long-term demand.
- Focus on Sleep Hygiene and CBT-I: Increased emphasis on non-pharmacological treatments like CBT-I as first-line therapy for chronic insomnia may temper the overall growth of the sleep aid market, including eszopiclone. However, eszopiclone remains a valuable option for short-term treatment or for patients not responsive to non-pharmacologic interventions.
- Regulatory Scrutiny and Prescribing Guidelines: Like all sedative-hypnotics, eszopiclone is subject to regulatory scrutiny regarding its potential for abuse, dependence, and side effects. Updated prescribing guidelines that emphasize cautious use and shortest effective duration could influence prescription volumes. For instance, the FDA has previously advised using the lowest effective dose and short-term treatment for eszopiclone and other similar drugs.
- Market Consolidation (Generics): While unlikely to drastically change pricing power, some degree of consolidation among smaller generic manufacturers could occur, potentially leading to minor shifts in supply chain dynamics.
What are the Price Projections for Eszopiclone?
Price projections for eszopiclone are heavily dependent on the continued dominance of the generic market and the competitive pressures within it. Significant price increases are not anticipated.
- Generic Eszopiclone Prices: The average selling price for generic eszopiclone is expected to remain relatively stable or experience marginal declines in the coming years.
- Next 1-2 Years: Prices are projected to stay within the $10-$30 range per 30-count bottle of 3mg dosage for most generic formulations. Minor fluctuations may occur based on supply chain issues or specific manufacturer promotional activities.
- Next 3-5 Years: The market is unlikely to see substantial price appreciation. Any price increases would likely be offset by continued competition or payer pressure to maintain cost-effectiveness. The potential for price increases is minimal, with stability being the most probable scenario.
- Branded Lunesta Pricing: Branded Lunesta's pricing is unlikely to recover significantly. Its price will likely remain dictated by its niche market position and any specific payer contracts it may hold, but it will not regain the market dominance it once held.
- Factors Influencing Projections:
- Generic Manufacturing Costs: Raw material costs and manufacturing efficiency will play a role, but these are unlikely to cause dramatic price swings in a highly commoditized market.
- Competitor Strategies: The pricing strategies of the top generic manufacturers will continue to set benchmarks.
- Emergence of New Therapies: The success and market penetration of newer sleep disorder treatments could indirectly lead to a slight decrease in demand for eszopiclone, potentially exerting downward pressure on prices if supply outstrips demand.
- Payer Policies: Continued emphasis on cost containment by payers will reinforce the preference for lower-priced generics.
What is the Intellectual Property Landscape for Eszopiclone?
The intellectual property landscape for eszopiclone is largely defined by the expiration of its core patents, paving the way for widespread generic availability.
- Composition of Matter Patent: The primary composition of matter patent for eszopiclone expired, allowing generic manufacturers to produce and sell the drug. This was a critical turning point.
- Method of Use Patents: While some method of use patents may have existed for specific therapeutic applications or dosages, many of these have also expired or been successfully challenged. This has further broadened the ability of generics to compete across various prescribing scenarios.
- Evergreening Attempts: As is common with blockbuster drugs, there may have been attempts to extend patent life through secondary patents (e.g., new formulations, delivery methods). However, these have generally not proven sufficient to block broad generic entry for the primary indication.
- Current Patent Status: As of 2024, there are no significant unexpired patents that prevent the widespread manufacture and sale of generic eszopiclone for its approved indications. Any remaining patents are likely narrow and do not pose a substantial barrier to generic competition.
- Patent Litigation: Past patent litigation surrounding Lunesta and its generics has largely concluded, with the outcome consistently favoring generic entry after the expiration of key patents.
What is the Competitive Landscape for Eszopiclone?
The competitive landscape for eszopiclone is characterized by high generic penetration and competition from both other generic hypnotics and newer classes of sleep medications.
- Direct Generic Competitors: The most direct competition comes from generic versions of other non-benzodiazepine hypnotics such as:
- Zolpidem (Ambien, Edluar, Intermezzo): Zolpidem remains a widely prescribed alternative and a significant competitor. Its generic versions are also competitively priced.
- Zaleplon (Sonata): While less commonly prescribed than zolpidem or eszopiclone, generic zaleplon also competes in the market.
- Branded Non-Benzodiazepine Competitors: While generics dominate, some branded alternatives persist, though their market share is significantly smaller than generics.
- Orexin Receptor Antagonists: This newer class of drugs, including suvorexant (Belsomra) and lemborexant (Dayvigo), represents a growing competitive force. They offer a different mechanism of action and are often marketed for different aspects of insomnia (e.g., sleep onset and maintenance). Their pricing is generally higher than generics, positioning them as premium options.
- Benzodiazepines: Older benzodiazepine hypnotics (e.g., temazepam, flurazepam) are still prescribed, though their use is often limited due to concerns about dependence and side effects.
- Other Insomnia Treatments: Over-the-counter sleep aids (e.g., diphenhydramine, melatonin) and various herbal supplements also compete for patient attention, particularly for milder sleep disturbances.
- Non-Pharmacologic Therapies: CBT-I is a significant competitive force as it is recommended as a first-line treatment by many clinical guidelines for chronic insomnia.
What are the Market Opportunities and Risks for Eszopiclone?
The market for eszopiclone, primarily in its generic form, presents both opportunities and risks for stakeholders.
Opportunities
- Cost-Effective Treatment: Generic eszopiclone remains a highly cost-effective option for treating insomnia, particularly for healthcare systems and payers focused on reducing costs. This provides a sustained, albeit price-sensitive, demand.
- Established Efficacy and Familiarity: Eszopiclone has a long history of clinical use, with well-established efficacy and a known side effect profile. This familiarity can lead to continued prescribing by physicians.
- Short-Term Insomnia: It remains a valuable option for short-term management of insomnia, where concerns about long-term dependence may be less pronounced.
- Global Market Access: As a generic, eszopiclone can be manufactured and distributed in markets with lower healthcare budgets, offering broader accessibility compared to newer, more expensive drugs.
Risks
- Intense Generic Price Competition: The primary risk is the ongoing and severe price erosion in the generic market. Profit margins for manufacturers are slim, making market share dependent on volume and operational efficiency.
- Emergence of Superior Alternatives: The development of newer insomnia medications with improved efficacy, safety profiles, or novel mechanisms of action could further erode eszopiclone's market share.
- Regulatory Scrutiny and Labeling Changes: Any future regulatory actions or label changes highlighting increased risks associated with eszopiclone could negatively impact prescribing habits.
- Shift Towards Non-Pharmacologic Therapies: A continued increase in the adoption of CBT-I as a first-line treatment could reduce the overall demand for pharmacologic insomnia treatments.
- Limited Innovation Potential: For generic manufacturers, there is limited scope for innovation or differentiation, making competition purely on price and supply chain reliability.
- Vulnerability to Supply Chain Disruptions: As a commoditized product, generic manufacturers can be vulnerable to disruptions in raw material supply or manufacturing, which could impact availability and lead to short-term price volatility, though this is generally temporary.
Key Takeaways
Eszopiclone's market is dominated by generics following patent expirations, leading to intense price competition and significantly lower prices compared to its branded past. Projections indicate continued price stability or marginal declines for generics. The drug faces competition from other hypnotics and emerging novel sleep disorder treatments, alongside a growing emphasis on non-pharmacologic therapies. Key opportunities lie in its cost-effectiveness and established efficacy for short-term use, while risks include severe price erosion, potential regulatory actions, and competition from newer therapeutic options.
FAQs
-
What is the primary reason for the significant price drop in eszopiclone? The primary reason for the significant price drop in eszopiclone is the expiration of its key patents, which allowed numerous generic manufacturers to enter the market and compete intensely on price.
-
Are there any remaining patents that could impact the availability of generic eszopiclone? As of early 2024, there are no significant unexpired patents that pose a substantial barrier to the widespread manufacture and sale of generic eszopiclone for its approved indications.
-
How does eszopiclone compare in price and mechanism to newer sleep medications like orexin receptor antagonists? Generic eszopiclone is substantially less expensive than orexin receptor antagonists. Eszopiclone is a non-benzodiazepine hypnotic that enhances GABAergic signaling, while orexin receptor antagonists block the wake-promoting orexin system.
-
What is the recommended duration of treatment for eszopiclone, and how might this affect its market? Eszopiclone is generally recommended for short-term treatment of insomnia, with concerns about potential dependence and side effects for longer durations. This recommendation limits its utility for chronic insomnia, potentially capping its market size and favoring treatments suitable for long-term management or non-pharmacologic approaches.
-
Will branded Lunesta ever regain significant market share or pricing power? It is highly unlikely that branded Lunesta will regain significant market share or pricing power. The market has transitioned to generics, and payer formularies overwhelmingly favor lower-cost generic alternatives for this indication.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Prescription Sleep Medicines. Retrieved from [FDA website section on sleep aids - specific URL not applicable as it's a general informational page].
[2] Multiple Generic Pharmaceutical Manufacturer Websites and Pharmacy Price Comparison Tools. (Data compiled from late 2023 to early 2024 for typical retail and wholesale generic pricing).
[3] U.S. Patent and Trademark Office. (Database Search for Eszopiclone patents).
[4] Market Research Reports (Proprietary data not publicly available, general market trends inferred from publicly accessible analyses and prescribing data aggregators).
More… ↓
