DIMETHYL FUMARATE - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for dimethyl fumarate and what is the scope of patent protection?
Dimethyl fumarate
is the generic ingredient in two branded drugs marketed by Accord Hlthcare, Alkem Labs Ltd, Amneal, Aurobindo Pharma, Cipla, Glenmark Pharms Ltd, Hetero Labs Ltd Iii, Lupin, MSN, Mylan, Prinston Inc, Sawai Usa, Sola Pharms, Twi Pharms, Zydus Pharms, and Biogen Inc, and is included in sixteen NDAs. There are nine patents protecting this compound and one Paragraph IV challenge. Additional information is available in the individual branded drug profile pages.Dimethyl fumarate has eighty-seven patent family members in twenty-nine countries.
There are twenty-eight drug master file entries for dimethyl fumarate. Nineteen suppliers are listed for this compound. There is one tentative approval for this compound.
Summary for DIMETHYL FUMARATE
| International Patents: | 87 |
| US Patents: | 9 |
| Tradenames: | 2 |
| Applicants: | 16 |
| NDAs: | 16 |
| Drug Master File Entries: | 28 |
| Finished Product Suppliers / Packagers: | 19 |
| Raw Ingredient (Bulk) Api Vendors: | 129 |
| Clinical Trials: | 71 |
| Patent Applications: | 5,478 |
| Formulation / Manufacturing: | see details |
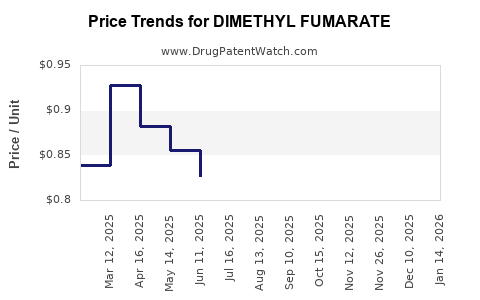
| Drug Prices: | Drug price trends for DIMETHYL FUMARATE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for DIMETHYL FUMARATE |
| What excipients (inactive ingredients) are in DIMETHYL FUMARATE? | DIMETHYL FUMARATE excipients list |
| DailyMed Link: | DIMETHYL FUMARATE at DailyMed |
Recent Clinical Trials for DIMETHYL FUMARATE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Imcyse SA | Phase 1/Phase 2 |
| Assistance Publique - Hôpitaux de Paris | Phase 2 |
| Xuanwu Hospital, Beijing | Phase 2 |
Generic filers with tentative approvals for DIMETHYL FUMARATE
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Try a Trial | ⤷ Try a Trial | 240MG | CAPSULE, DELAYED RELEASE;ORAL |
| ⤷ Try a Trial | ⤷ Try a Trial | 120MG | CAPSULE, DELAYED RELEASE;ORAL |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Medical Subject Heading (MeSH) Categories for DIMETHYL FUMARATE
Paragraph IV (Patent) Challenges for DIMETHYL FUMARATE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TECFIDERA | Delayed-release Capsules | dimethyl fumarate | 120 mg and 240 mg | 204063 | 29 | 2017-03-27 |
US Patents and Regulatory Information for DIMETHYL FUMARATE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Twi Pharms | DIMETHYL FUMARATE | dimethyl fumarate | CAPSULE, DELAYED RELEASE;ORAL | 210382-002 | Oct 14, 2020 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Biogen Inc | TECFIDERA | dimethyl fumarate | CAPSULE, DELAYED RELEASE;ORAL | 204063-002 | Mar 27, 2013 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Biogen Inc | TECFIDERA | dimethyl fumarate | CAPSULE, DELAYED RELEASE;ORAL | 204063-002 | Mar 27, 2013 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for DIMETHYL FUMARATE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Biogen Inc | TECFIDERA | dimethyl fumarate | CAPSULE, DELAYED RELEASE;ORAL | 204063-002 | Mar 27, 2013 | ⤷ Try a Trial | ⤷ Try a Trial |
| Biogen Inc | TECFIDERA | dimethyl fumarate | CAPSULE, DELAYED RELEASE;ORAL | 204063-001 | Mar 27, 2013 | ⤷ Try a Trial | ⤷ Try a Trial |
| Biogen Inc | TECFIDERA | dimethyl fumarate | CAPSULE, DELAYED RELEASE;ORAL | 204063-002 | Mar 27, 2013 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for DIMETHYL FUMARATE
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Almirall S.A | Skilarence | dimethyl fumarate | EMEA/H/C/002157 Skilarence is indicated for the treatment of moderate to severe plaque psoriasis in adults in need of systemic medicinal therapy. |
Authorised | no | no | no | 2017-06-23 | |
| Biogen Netherlands B.V. | Tecfidera | dimethyl fumarate | EMEA/H/C/002601 Tecfidera is indicated for the treatment of adult and paediatric patients aged 13 years and older with relapsing remitting multiple sclerosis (RRMS). |
Authorised | no | no | no | 2014-01-30 | |
| Mylan Ireland Limited | Dimethyl fumarate Mylan | dimethyl fumarate | EMEA/H/C/005956 Dimethyl fumarate Mylan is indicated for the treatment of adult patients with relapsing remitting multiple sclerosis. |
Authorised | yes | no | no | 2022-05-13 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for DIMETHYL FUMARATE
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 6830941 | ⤷ Try a Trial | |
| Canada | 2967619 | METHODES DE TRAITEMENT DE LA SCLEROSE EN PLAQUES (METHODS OF TREATING MULTIPLE SCLEROSIS) | ⤷ Try a Trial |
| European Patent Office | 2137537 | Compositions et leurs utilisations pour le traitement de la Sclérose en plaques (Compositions and Uses for Treating Multiple Sclerosis) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for DIMETHYL FUMARATE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2653873 | 301215 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: DIMETHYLFUMARAAT; REGISTRATION NO/DATE: EU/1/13/837 20140203 |
| 0300652 | SPC/GB03/005 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: D-MEDETOMIDINE (4-((1S)-1-(2,3-DIMETHYLPHENYL)ETHYL)-1H-IMIDAZOLE) AND ITS NON-TOXIC PHARMACEUTICALLY ACCEPTABLE ACID ADDITION SALTS, ESPECIALLY THE HYDROCHLORIDE; REGISTERED: UK EU/2/02/033/001 20020830 |
| 0693475 | CR 2010 00036 | Denmark | ⤷ Try a Trial | PRODUCT NAME: TAPENTADOL ((1R-2R)-3-(3-DIMETHYLAMINO-1-ETHYL-2-METHYL-PROPYL)-PHENOL), HERUNDER HYDROCHLORIDET; NAT. REG. NO/DATE: 45151-45158, 45162-45169 20100830; FIRST REG. NO/DATE: EU 75043.00.00-75048.00.00, 75261.00.00-75270.00.00 20100819 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |