Last updated: April 25, 2026
What is the calcitriol commercial footprint?
Calcitriol (1,25-dihydroxyvitamin D3) is the hormonally active vitamin D metabolite used to treat disorders driven by impaired mineral metabolism. Commercially, calcitriol sits in the established segment of endocrine and mineral metabolism drugs where pricing and demand are shaped by chronic prescribing patterns, generic erosion, and payer controls.
Product form factors
- Oral capsules and oral solutions (most common).
- Topical formulations (used for dermatologic indications such as psoriasis in some jurisdictions).
- Dose dispersion across strengths (drives inventory and NDC-level complexity).
Demand drivers
- Chronic conditions that require continuous dosing.
- Patient adherence dependence (titration and monitoring via calcium and phosphate levels).
- Substitution effects when generics exist for oral brands.
How do market dynamics shape pricing and volume?
Calcitriol pricing and volume are influenced by three interacting forces: (1) generic penetration, (2) payer rules for vitamin D analogs, and (3) safety and monitoring behavior in real-world prescribing.
1) Generic and supply dynamics
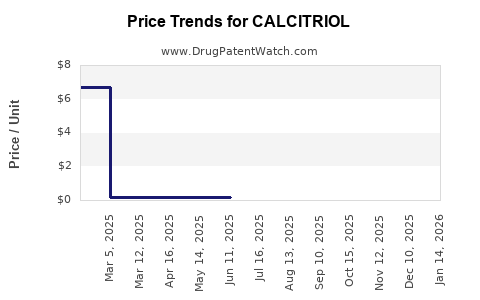
- In most developed markets, calcitriol has multiple generic manufacturers for oral products, which compresses net price through competitive tendering and formularies.
- Supply stability often improves over time as production scales and low-cost entrants consolidate, pushing the market toward benchmark-driven pricing rather than innovation-led pricing.
Typical commercial outcome
- Volume tends to remain resilient because the molecule treats chronic disease and has established prescriber habits.
- Net revenue tends to flatten or decline as wholesalers and PBMs push pricing toward cost-plus or competitive benchmarks.
2) Payer controls and formulary positioning
Payers generally treat vitamin D agents as a cost-controlled drug class and apply:
- Step edits within vitamin D products (when clinically acceptable).
- Prior authorization for higher-cost formulations or nonpreferred strengths.
- Quantity limits based on dosing regimens.
This structure tends to reduce price volatility but limits upside from incremental brand differentiation.
3) Clinical monitoring and adherence
Calcitriol requires monitoring of calcium, phosphate, and renal function in many patient populations. This affects market dynamics by:
- Creating a “discipline” barrier against frequent switching (physicians prefer stable regimens).
- Increasing the importance of drug availability and consistent formulation performance.
- Slowing uptake in regions or segments where monitoring infrastructure is limited.
Net effect
- Uptake is steadier than for acute therapies.
- Sales growth is usually not step-change driven; it is incremental and tied to epidemiology plus prescribing persistence.
Which indications drive sales and how does that change over time?
Calcitriol’s revenue is distributed across endocrine-mineral indications and, in some markets, dermatology. The relative mix shifts with local practice patterns and availability of alternative therapies.
Primary revenue pools
- Hypocalcemia due to hypoparathyroidism or other causes where active vitamin D is clinically required.
- Chronic kidney disease-related mineral disorders (where active vitamin D analogs may be preferred depending on guideline alignment and payer constraints).
- Other mineral metabolism disorders where calcitriol is selected for active vitamin D replacement rather than nutritional vitamin D.
Dermatology contribution
- Topical calcitriol is used in psoriasis in some jurisdictions.
- Dermatology contributes differently by country due to competitive topical steroid and vitamin D analog landscapes.
Trajectory implication
- Oral calcitriol typically anchors the bulk of durable demand.
- Dermatology can add variability depending on competitive intensity and guideline preferences, but it usually does not override the core oral market.
What is the financial trajectory pattern for calcitriol?
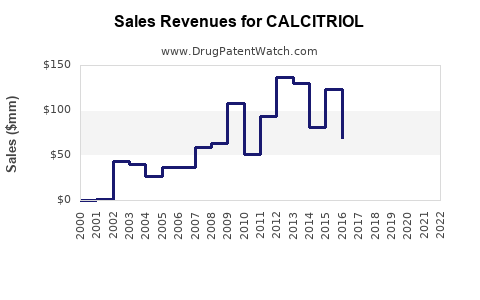
Calcitriol is a mature, off-patent molecule in most markets. Financial trajectories typically show:
- Early brand-led growth around line extensions and formulation wins.
- Sustained volume as generics enter and prescriptions normalize.
- Margin compression as net prices decline with competitive procurement.
- Late-stage stabilization at lower price points where unit volume becomes the main lever.
Revenue behavior by phase (typical for mature calcitriol markets)
- Unit volumes: stable to moderately growing with treated prevalence.
- Net price: declines after generic entry or formulary renegotiation cycles.
- Total revenue: often flattens after price erosion, with occasional recoveries from temporary supply tightness, dosage mix shifts, or tender-based pricing resets.
How do competitive products and substitution affect the financial line?
Calcitriol faces substitution in two directions: different vitamin D forms and different dosing/route options.
1) Substitution within vitamin D therapy
- Nutritional vitamin D (e.g., cholecalciferol/ergocalciferol) can substitute in some cases where conversion is adequate.
- Other active vitamin D analogs (calcifediol, alfacalcidol, and newer agents depending on market) may displace calcitriol where guidelines or payer policy prefer them.
Substitution reduces the addressable pool when clinical alternatives are viewed as equivalent.
2) Dose and formulation competition
- Alternative strengths and formulations influence adherence and reduce switching friction.
- Payers may push to “preferred NDCs,” narrowing the effective commercial footprint even when branded products remain listed.
Financial impact
- Generic competition usually drives the biggest net price hits.
- Therapeutic substitution drives the biggest volume displacement in specific patient subsegments.
What do market pricing and reimbursement dynamics imply for profitability?
For off-patent molecules like calcitriol, profitability depends less on premium pricing and more on:
- Contracting discipline (PBM and wholesaler terms).
- Manufacturing scale and cost per unit.
- Portfolio mix (oral vs topical; high vs low strength utilization).
- Inventory management around tender cycles and demand spikes after formulary changes.
Operating profile
- Revenue volatility is usually lower than in innovation cycles.
- Cash generation is typically steady but margin is sensitive to price renegotiation, especially in tenders.
Where are the main near-term risk and upside levers?
Risk levers
- Further formulary tightening and preferred product consolidation.
- Additional generic supply increases in regions with tender-driven pricing.
- Therapeutic substitution toward other vitamin D therapies where payer policies align with clinical guidelines.
Upside levers
- Epidemiology-driven growth in treated populations for mineral disorders.
- Switching to active vitamin D therapies when nutritional forms fail in conversion-limited patients.
- Topical dermatology demand retention where topical vitamin D analog preferences remain.
How does calcitriol’s safety profile influence market behavior and spend?
Calcitriol safety risk is a major determinant of long-term prescribing patterns. The need to manage hypercalcemia risk affects:
- Initiation caution and slower uptake in borderline populations.
- Continued use among stabilized patients once monitoring protocols are in place.
- Risk-driven payer scrutiny around dosing and monitoring requirements.
This typically supports stable long-term demand among established users while limiting aggressive expansion.
What does the competitive landscape suggest for financial trajectory by geography?
Calcitriol’s trajectory varies across countries based on:
- Generic penetration speed.
- Reimbursement rules for active vitamin D agents.
- Strength-by-strength prescribing preferences.
- Availability of topical calcitriol versus alternative dermatology drugs.
Common global pattern
- North America and Europe show maturity with net price compression post-generic entry.
- Emerging markets often show later penetration dynamics, with slower erosion when distribution is less consolidated.
Key Takeaways
- Calcitriol is a mature, off-patent active vitamin D drug with revenue primarily anchored in chronic endocrine-mineral disorder management.
- Market dynamics are dominated by generic competition and payer formulary controls, which typically drive net price compression while keeping unit demand comparatively resilient.
- Financial trajectory follows a mature-product pattern: early brand-led growth transitions into stable volume with flattening revenue and margin pressure after generic entry and tender renegotiations.
- Therapeutic substitution within vitamin D therapy and across active analogs affects both addressable volume and net pricing.
- Safety and monitoring requirements support patient retention but limit rapid expansion, making long-term sales more dependent on treated prevalence than on innovation-led uptake.
FAQs
1) Is calcitriol’s market growth driven by new launches?
No. Growth is typically driven by treated prevalence, prescribing persistence, and occasional mix shifts across strengths and formulations rather than new innovation.
2) What matters most for net revenue in calcitriol?
Net pricing from PBM/wholesaler contracting and formulary preference, since generic supply and therapeutic substitution compress margins.
3) Does calcitriol have meaningful competitive threats beyond generics?
Yes. Active vitamin D analogs and other vitamin D forms can displace calcitriol in subpopulations where conversion or guideline preferences differ.
4) Why is calcitriol less volatile than specialty oncology drugs?
Because it treats chronic conditions with long-term dosing and monitoring routines, which stabilizes baseline demand even when prices adjust.
5) What would most likely improve calcitriol’s financial trajectory?
Improved formulary access for preferred NDCs, stabilized contract pricing, and volume protection in subsegments that require active vitamin D rather than nutritional supplementation.
References
[1] WHO. (n.d.). Calcitriol (1,25-dihydroxyvitamin D3). World Health Organization classification and related documentation.
[2] US FDA. (n.d.). Drug Safety and Availability for calcitriol-containing products (labeling and product information). U.S. Food and Drug Administration.
[3] EMA. (n.d.). Product information and assessment reports for calcitriol medicines in EU member states. European Medicines Agency.