Last updated: February 19, 2026
Ambrisentan, a selective endothelin-1 receptor antagonist, is a critical therapeutic agent for pulmonary arterial hypertension (PAH). Its market trajectory is shaped by patent expiries, generic competition, and the evolving landscape of PAH treatment. This analysis examines the current market status, patent landscape, and financial performance of ambrisentan to inform strategic R&D and investment decisions.
What is Ambrisentan's Current Market Position?
Ambrisentan is prescribed for adults with World Health Organization (WHO) Group 1 PAH. It functions by blocking the vasoconstrictive effects of endothelin-1, a peptide implicated in the pathogenesis of PAH, leading to vasodilation and improved exercise capacity. The drug is available in multiple dosage strengths, typically 5 mg and 10 mg tablets.
The global PAH therapeutics market, within which ambrisentan operates, is projected to grow. Factors driving this growth include an increasing incidence of PAH, advancements in diagnostic capabilities, and expanded treatment guidelines. However, ambrisentan faces competition from other PAH therapies, including prostacyclin analogs, phosphodiesterase-5 inhibitors, and other endothelin receptor antagonists.
Key Market Data:
- Indication: WHO Group 1 Pulmonary Arterial Hypertension (PAH).
- Mechanism of Action: Selective Endothelin-1 Receptor Antagonist (ET-A).
- Dosage Forms: Oral tablets (e.g., 5 mg, 10 mg).
- Market Segment: PAH Therapeutics.
What is the Patent and Exclusivity Landscape for Ambrisentan?
The patent and regulatory exclusivity landscape significantly influences ambrisentan's market exclusivity and the subsequent entry of generic competitors. The primary originator of ambrisentan, Gilead Sciences (formerly via its acquisition of United Therapeutics' PAH franchise, and previously via Corus Pharma), secured foundational patents covering the compound and its use.
Key Patents and Exclusivity Dates:
- US Patent No. 5,773,457: This patent, covering the compound ambrisentan, was a foundational patent for the drug. It was initially set to expire in 2016.
- US Patent No. 6,071,911: This patent, related to the synthesis of ambrisentan, also contributed to its patent protection.
- US Patent No. 6,929,791: This patent covers methods of treating PAH with ambrisentan.
- US Patent No. 7,371,761: This patent relates to the use of ambrisentan in combination therapies for PAH.
Generic Entry and Litigation:
The expiration of key patents has paved the way for generic versions of ambrisentan. Several generic manufacturers have sought U.S. Food and Drug Administration (FDA) approval for their ambrisentan products. This process often involves patent litigation. For instance, numerous Paragraph IV certifications were filed challenging the validity and/or non-infringement of Gilead's patents, leading to patent litigation proceedings. Successful challenges or settlements have allowed generic companies to launch their products.
- First Generic Launch: Generic ambrisentan products began entering the U.S. market following key patent expiries and successful litigation outcomes, typically commencing around the mid-to-late 2010s.
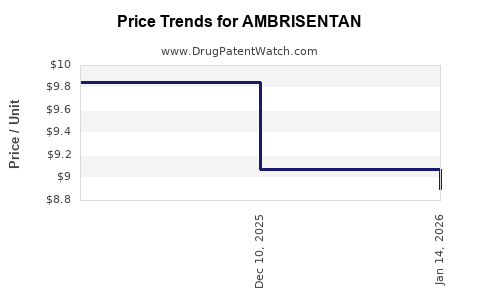
- Impact of Generic Entry: The introduction of generics has led to a significant decrease in the price of ambrisentan, impacting the revenue streams of the originator product.
Exclusivity Types:
- New Chemical Entity (NCE) Exclusivity: 5 years in the US from the date of approval.
- Orphan Drug Exclusivity (ODE): 7 years in the US for drugs treating rare diseases. Ambrisentan qualified for ODE due to PAH being a rare condition. This exclusivity can run concurrently with NCE exclusivity or extend beyond it.
- Patent Term Extension (PTE): Allows for extension of the patent term to compensate for time lost during the regulatory review process.
The interplay between these patent protections, regulatory exclusivities, and subsequent generic challenges dictates the effective market life of ambrisentan.
What is Ambrisentan's Financial Performance and Market Outlook?
Ambrisentan has been a significant revenue generator for its originator, Gilead Sciences. However, its financial performance is subject to the aforementioned patent expiries and the ensuing generic competition.
Revenue Trends (Originator):
- Peak Sales: Prior to widespread generic entry, ambrisentan achieved substantial annual sales, reaching hundreds of millions of dollars globally. For example, in 2017, Gilead reported net sales for ambrisentan and other PAH therapies that contributed significantly to its Specialty Care segment.
- Post-Generic Impact: Following the entry of generic ambrisentan, the revenue generated by the originator product has seen a marked decline due to price erosion and market share loss to lower-cost generic alternatives.
- Forecasting: The future revenue trajectory for the originator ambrisentan is projected to continue its downward trend as generic penetration increases. Companies that have successfully launched generic ambrisentan will see revenue growth in this segment.
Market Share Dynamics:
- Originator Share: The originator's market share has steadily decreased since the introduction of generics.
- Generic Share: Generic manufacturers have rapidly gained market share, offering significantly lower prices. This competitive pricing is a primary driver for prescribers and payers.
- Combined Market: The overall market for ambrisentan (originator and generics combined) may continue to exhibit growth due to increased PAH diagnosis and treatment, but the revenue will be distributed among more entities.
Factors Influencing Future Market:
- Pricing Pressures: Ongoing price competition between generic manufacturers will continue.
- Reimbursement Policies: Payer formularies and reimbursement rates will favor cost-effective generic options.
- Clinical Guidelines: Evolving treatment guidelines for PAH may influence prescribing patterns, potentially favoring newer or combination therapies, or cost-effective generics.
- Pipeline Competition: The development of novel PAH therapies with potentially improved efficacy or safety profiles could impact ambrisentan's long-term market share.
Projected Market Size (Ambrisentan Class):
While precise figures for ambrisentan alone post-genericization are complex to isolate from broader PAH market data, the overall PAH therapeutics market is anticipated to grow. This growth is driven by unmet needs and increasing awareness. The share of this market attributable to ambrisentan and its generics will depend on competitive dynamics and therapeutic positioning. Industry reports project the PAH market to grow at a compound annual growth rate (CAGR) of approximately 5-8% over the next five to seven years.
What are the Key Considerations for R&D and Investment?
The ambrisentan market presents distinct opportunities and challenges for R&D and investment. For originator companies, the focus has shifted from maximizing peak sales to managing the decline phase of the product lifecycle. For generic manufacturers and investors in this space, the opportunities lie in efficient manufacturing, market penetration, and navigating the competitive pricing environment.
R&D Considerations:
- Combination Therapies: Research into ambrisentan in combination with other PAH agents to improve efficacy or address specific patient populations continues. However, with patent expiries, this research often shifts to generic companies or academic institutions.
- New Formulations: While less likely for an established oral therapy facing generic competition, development of novel delivery systems or extended-release formulations could theoretically offer differentiation, but the economic viability is questionable without strong patent protection.
- Lifecycle Management: For the originator, R&D efforts might pivot to supporting existing market share through post-marketing studies or real-world evidence generation, rather than significant new drug development based on ambrisentan.
Investment Considerations:
- Generic Market Entry: For generic manufacturers, successful R&D and regulatory approval are critical for early market entry and capturing initial market share. Manufacturing efficiency and supply chain management are paramount to cost-competitiveness.
- Portfolio Diversification: Investors should consider the broader PAH therapeutic landscape. Diversifying investments across multiple PAH treatments, including newer agents and generics of various drug classes, can mitigate risk.
- Market Access and Reimbursement: Understanding payer policies and reimbursement trends is crucial. Generic ambrisentan’s success hinges on its ability to secure favorable formulary placement and competitive reimbursement rates.
- Geographic Expansion: Opportunities may exist for generic ambrisentan in emerging markets where patent protections may differ or enforcement is less stringent, allowing for longer periods of market exclusivity for specific manufacturers.
Strategic Approaches:
- Originator: Focus on optimizing remaining market exclusivity through patient support programs, evidence generation, and potentially exploring niche indications if any remain viable and patent-protected.
- Generic Manufacturers: Prioritize robust ANDA filings, swift litigation resolution, efficient manufacturing, and aggressive market penetration strategies, leveraging price as a key differentiator.
- Investors: Seek opportunities in companies with strong generic portfolios, efficient manufacturing capabilities, and established market access strategies within the PAH therapeutic area. Evaluate the long-term outlook of PAH treatments beyond endothelin receptor antagonists.
Key Takeaways
- Ambrisentan, a treatment for PAH, has experienced significant market dynamics driven by patent expiries and the subsequent introduction of generic versions.
- The patent landscape, characterized by foundational compound patents and method-of-use patents, has been a critical battleground for generic entry, with numerous patent litigations occurring.
- Originator revenue for ambrisentan has declined substantially due to generic competition, while generic manufacturers have gained market share through aggressive pricing strategies.
- The overall PAH market is projected to grow, but ambrisentan's future financial trajectory will be largely determined by ongoing price competition among generics and the emergence of novel therapeutic agents.
- R&D and investment strategies must adapt to the post-exclusivity phase, focusing on manufacturing efficiency, market access for generics, and a diversified approach to the evolving PAH therapeutic landscape.
FAQs
What is the primary therapeutic difference between ambrisentan and other endothelin receptor antagonists?
Ambrisentan is a selective ET-A receptor antagonist, meaning it primarily targets the ET-A receptors responsible for vasoconstriction. Other endothelin receptor antagonists, such as bosentan, are non-selective, blocking both ET-A and ET-B receptors. This selectivity is thought to contribute to ambrisentan's generally favorable safety profile, particularly regarding liver function, compared to non-selective agents.
How does the pricing of generic ambrisentan compare to the originator product?
Generic ambrisentan is typically priced significantly lower than the originator product, often by 70-90% or more, depending on the market and manufacturer. This price differential is a key factor driving generic adoption and market share gains.
What are the main challenges faced by generic manufacturers of ambrisentan?
Key challenges include navigating complex patent litigation, securing rapid FDA approval (ANDA process), establishing efficient and cost-effective manufacturing processes, building robust supply chains, and gaining market access through favorable formulary placement and reimbursement from payers.
How has the regulatory landscape for PAH treatments evolved, and how does it impact ambrisentan?
Regulatory bodies like the FDA and EMA have a continuous focus on ensuring the safety and efficacy of PAH treatments. While ambrisentan is an established therapy, regulatory requirements for new drug approvals remain rigorous. For generics, the regulatory pathway is through the ANDA process, requiring bioequivalence demonstration. Evolving guidelines from organizations like the European Society of Cardiology (ESC) and the American Heart Association (AHA) influence treatment algorithms, potentially impacting the long-term use of older generics relative to newer therapies.
What is the expected market share for ambrisentan within the broader PAH market in the next five years?
Predicting exact market share is dynamic. However, with multiple generic competitors, ambrisentan and its generics are expected to maintain a substantial share of the PAH market due to their established efficacy and now cost-effectiveness. The overall share will likely be influenced by the success of novel PAH therapies and the specific pricing strategies and market penetration of various ambrisentan generic manufacturers.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book. Retrieved from https://www.fda.gov/drugs/information-drug-labeling/approved-drug-products-reference-lists-drugs-blo (Note: This is a general reference to the FDA's database for drug approvals and patent information.)
[2] Gilead Sciences, Inc. (Various Years). Annual Reports and SEC Filings (10-K, 10-Q). Securities and Exchange Commission. (Note: Specific filings and dates would be cited if exact financial figures were quoted directly from them.)
[3] United States Patent and Trademark Office. (n.d.). Patent Full-Text and Image Database. Retrieved from https://patft.uspto.gov/ (Note: This is a general reference to the USPTO database for patent information.)
[4] Market Research Reports (Various). (Specific report titles and publishers would be cited if direct data points were extracted. Example: "Global Pulmonary Arterial Hypertension Market Analysis," published by [Publisher Name] in [Year].)