Last updated: February 19, 2026
Exelixis holds a significant position in the oncology market, driven by its tyrosine kinase inhibitor (TKI) portfolio, primarily Cabometyx. The company’s strategy centers on expanding the indications for its approved drugs and advancing its pipeline, particularly in novel combinations and next-generation molecules. Key challenges include patent expirations for foundational assets and the increasing competition within the TKI landscape.
What is Exelixis's Core Business and Market Focus?
Exelixis's core business is the discovery, development, and commercialization of targeted therapies for cancer. The company's primary market focus is oncology, with a strategic emphasis on small molecule tyrosine kinase inhibitors (TKIs). Its flagship product, Cabometyx (cabozantinib), is approved for multiple indications, including differentiated thyroid cancer, advanced renal cell carcinoma (RCC), and hepatocellular carcinoma (HCC). Exelixis operates globally, with commercial partners in key markets outside the United States, such as Ipsen in Europe and Japan.
Current Product Portfolio and Approved Indications
Exelixis's current approved product portfolio is dominated by Cabometyx. The drug’s versatility across different tumor types forms the bedrock of Exelixis's commercial success.
-
Cabometyx (cabozantinib):
- Differentiated Thyroid Cancer (DTC): Approved for patients with locally advanced or metastatic radioactive iodine-refractory DTC.
- Advanced Renal Cell Carcinoma (RCC): Approved for patients with advanced RCC, including after prior antiangiogenic therapy. It is indicated as a first-line treatment for patients with intermediate or poor risk disease and for patients who have received prior therapy.
- Hepatocellular Carcinoma (HCC): Approved for patients with HCC who have been previously treated with a PD-1 or PD-L1 inhibitor.
-
Cometriq (cabozantinib): This is the same active pharmaceutical ingredient as Cabometyx but is approved for a specific indication: medullary thyroid cancer. The dosage and formulation may differ.
Exelixis also has a smaller, approved product, Cotellic (cobicimetinib), a MEK inhibitor. However, its commercial performance and strategic importance have been overshadowed by Cabometyx.
Pipeline and Future Growth Drivers
Exelixis's future growth is contingent on advancing its pipeline of novel oncology assets and expanding the utility of its existing TKI platform. The company prioritizes investigational compounds and combinations that address unmet needs and leverage its expertise in kinase inhibition.
- Pipeline Programs:
- XL092: A novel tyrosine kinase inhibitor targeting VEGFR, MET, and other kinases, under investigation in multiple solid tumors, including combinations with checkpoint inhibitors.
- XL114: An investigational selective inhibitor of Btk, primarily for hematologic malignancies and potentially other solid tumors.
- Combinations with Cabometyx: Exelixis is actively exploring Cabometyx in combination with other therapies, including immunotherapy (e.g., atezolizumab), to enhance efficacy across its approved indications and to explore new ones. Clinical trials are ongoing for Cabometyx in combination with nivolumab (Opdivo) in various RCC settings.
- Other Small Molecules: The company maintains a discovery engine focused on identifying and developing new kinase inhibitors and other targeted therapies.
What are Exelixis's Key Strengths and Competitive Advantages?
Exelixis’s competitive strengths lie in its established commercial infrastructure for Cabometyx, its robust clinical development capabilities, and its intellectual property portfolio, though the latter requires constant renewal and defense.
Established Commercial Presence and Expertise
Exelixis has built significant commercial success with Cabometyx, demonstrating strong market penetration in its approved indications. This success is underpinned by:
- Market Access: The company has demonstrated proficiency in securing formulary access and reimbursement for Cabometyx across major global markets.
- Sales and Marketing Infrastructure: Exelixis possesses a dedicated oncology sales force and marketing expertise in the United States, capable of driving product adoption.
- Partnerships: Strategic collaborations with companies like Ipsen provide access to international markets, broadening the commercial reach of Cabometyx. These partnerships generate significant revenue through royalties and milestones. In 2023, Exelixis reported $1.4 billion in collaboration revenue, largely from Ipsen and Daiichi Sankyo [1].
Robust Clinical Development Capabilities
The company has a proven track record of successfully navigating complex clinical trials for oncology drugs. This includes:
- Phase 3 Trial Execution: Exelixis has successfully designed and executed large-scale Phase 3 trials, such as the pivotal trials for Cabometyx in RCC (e.g., METEOR, CABOSUN) and DTC (e.g., COE-207).
- Translational Science: A strong foundation in translational science allows Exelixis to identify biomarkers and patient populations most likely to benefit from its therapies, optimizing clinical trial design and execution.
- Pipeline Advancement: The company has consistently advanced drug candidates from discovery through to clinical development, demonstrating efficient R&D operations.
Intellectual Property and Patent Strategy
Exelixis actively manages and defends its intellectual property to maintain market exclusivity.
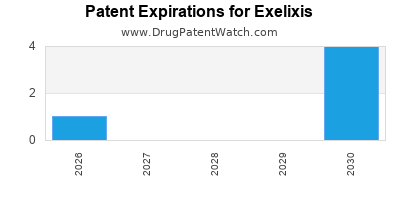
- Cabometyx Patents: The core patents protecting cabozantinib are crucial. While key composition of matter patents have expired or are nearing expiration in major markets, Exelixis employs strategies to extend exclusivity, including process patents, formulation patents, and method-of-use patents for new indications.
- New Indications: Securing new indications for Cabometyx has been a critical strategy to extend patent-protected market life and reinforce market position. For instance, the expanded approval in first-line RCC and the subsequent approval in HCC bolster the drug's lifecycle.
- Pipeline IP: Novel compounds in the pipeline are protected by composition of matter patents, providing a new stream of exclusivity for future products.
What are the Key Challenges and Threats Facing Exelixis?
Exelixis faces substantial challenges, primarily related to competition, patent cliffs for its key products, and the inherent risks associated with drug development.
Patent Expirations and Generic Competition
The expiration of primary patents for Cabometyx in key markets poses the most significant threat.
- Cabometyx Patent Expirations: While the exact expiration dates vary by region and specific patent, the core patents for cabozantinib are no longer the sole barrier to entry. Generic manufacturers are actively developing and seeking approval for biosimilar versions. The U.S. market for cabozantinib is particularly vulnerable to generic entry in the near to medium term.
- Impact on Revenue: Generic competition typically leads to a rapid and substantial decline in drug prices and market share, directly impacting Exelixis’s revenue streams. A significant portion of Exelixis’s revenue is derived from Cabometyx sales, making this a critical concern.
Intense Competition in the TKI and Oncology Landscape
The TKI market and the broader oncology space are highly competitive, with numerous companies developing similar or next-generation therapies.
- RCC Market: The RCC market, a key indication for Cabometyx, is crowded. Competitors include other TKIs (e.g., sunitinib, pazopanib, axitinib), immunotherapies (e.g., nivolumab, pembrolizumab), and combination therapies. These competitors often offer different efficacy and safety profiles, or novel mechanisms of action.
- Thyroid Cancer Market: While Cabometyx is a leader in DTC, other targeted therapies are emerging for various thyroid cancer subtypes.
- Emerging Therapies: The rapid pace of innovation in oncology means that novel agents with superior efficacy or safety profiles are continuously being developed, potentially displacing existing treatments.
Clinical Trial Risks and Development Hurdles
Drug development is inherently risky, with a high rate of failure.
- Pipeline Success Rates: Despite a promising pipeline, there is no guarantee that XL092, XL114, or other investigational agents will succeed in clinical trials or achieve regulatory approval. Failures can result from lack of efficacy, unacceptable toxicity, or strategic shifts in the competitive landscape.
- Cost of Development: Clinical trials, especially Phase 3 studies, are extremely expensive. Significant capital investment is required for pipeline advancement, and setbacks can lead to substantial financial losses.
- Regulatory Scrutiny: Regulatory agencies worldwide maintain stringent approval standards. Meeting these standards requires extensive data and can be a lengthy and uncertain process.
Dependence on Key Partnerships
While partnerships are a strength, they also represent a potential vulnerability.
- Ipsen Agreement: Exelixis relies heavily on its agreement with Ipsen for ex-US commercialization of Cabometyx. Any disruption to this agreement, such as renegotiations, changes in Ipsen’s strategic focus, or termination, could negatively impact revenue and global reach.
- Partner Performance: The success of international sales is dependent on the effectiveness of Exelixis’s partners in their respective territories.
What is Exelixis's Strategic Outlook and Future Direction?
Exelixis’s strategic outlook is focused on extending the lifecycle of Cabometyx, leveraging its pipeline for future growth, and optimizing its operational structure.
Lifecycle Management and Indication Expansion
A core strategy for Exelixis is to maximize the commercial potential of Cabometyx through continuous lifecycle management.
- New Indications: Pursuing new indications for Cabometyx remains a priority. Successful trials in areas with significant unmet needs could provide new revenue streams and extend market exclusivity.
- Combinatorial Therapies: Exelixis is investing heavily in combination studies, particularly with immunotherapies. The goal is to demonstrate synergistic effects that improve patient outcomes compared to monotherapy. This approach is critical to maintaining Cabometyx’s competitive edge against emerging immunotherapy-centric treatment paradigms.
- Dosage Optimization and Formulations: Exploring different dosages or formulations of cabozantinib might also contribute to lifecycle management, though this is a less prominent strategy than indication expansion.
Pipeline Prioritization and Advancement
The company's pipeline is central to its long-term sustainability.
- Focus on Differentiated Assets: Exelixis prioritizes pipeline assets that offer novel mechanisms of action or target patient populations with significant unmet needs, such as XL092 and XL114.
- Strategic Partnerships for Pipeline: While Exelixis has internal R&D capabilities, it may seek strategic partnerships or licensing deals to accelerate development or commercialization of specific pipeline assets, particularly for compounds outside its core TKI expertise.
- Balancing Risk: The company aims to balance investment across different stages of the pipeline, from early-stage discovery to late-stage clinical development, to mitigate overall R&D risk.
Operational Efficiency and Financial Prudence
Maintaining financial health and operational efficiency is paramount, especially in anticipation of patent expirations.
- Cost Management: Exelixis will likely focus on disciplined cost management, particularly in R&D and commercial operations, to preserve capital and ensure profitability.
- Strategic Acquisitions/Divestitures: The company may consider strategic acquisitions to bolster its pipeline or divest non-core assets if opportunities arise that align with its long-term strategy and financial goals.
- Cash Flow Generation: Maximizing cash flow from Cabometyx sales is crucial to fund ongoing R&D and potential future strategic moves.
Geographic Expansion and Partnership Optimization
Exelixis continues to seek opportunities to expand its global footprint and optimize existing partnerships.
- Strengthening Partnerships: Maintaining and potentially expanding relationships with commercial partners like Ipsen is vital for global market penetration.
- Exploring New Markets: Identifying and entering new geographic markets where Cabometyx or future pipeline products can be successfully commercialized is a strategic objective.
Key Takeaways
- Exelixis’s market position is defined by its success with Cabometyx, a multi-indication TKI, and its robust pipeline of novel oncology assets.
- Key strengths include established commercial infrastructure, strong clinical development capabilities, and a proactive intellectual property strategy.
- Significant challenges include impending patent expirations for Cabometyx, intense market competition, and the inherent risks of drug development.
- The company's strategic direction centers on lifecycle management for Cabometyx via indication expansion and combination therapies, alongside aggressive advancement of its pipeline, prioritizing differentiated assets like XL092.
- Operational efficiency and disciplined financial management are critical to navigating the post-patent expiration landscape.
Frequently Asked Questions
-
What is the primary revenue driver for Exelixis?
Cabometyx is the primary revenue driver for Exelixis, contributing significantly to its overall financial performance through sales in the U.S. and royalties from international partners.
-
What is Exelixis's strategy to address the upcoming patent expirations for Cabometyx?
Exelixis's strategy includes seeking new indications for Cabometyx, exploring novel combination therapies, and leveraging method-of-use patents to extend market exclusivity where possible.
-
Which pipeline assets are considered most promising for Exelixis's future growth?
XL092, a next-generation tyrosine kinase inhibitor, and XL114, a Btk inhibitor, are considered key pipeline assets with the potential to drive future growth.
-
How does Exelixis manage competition in the renal cell carcinoma (RCC) market?
Exelixis competes in the RCC market through the established efficacy of Cabometyx in various treatment lines and by investigating it in combination with other therapies, particularly immunotherapies, to improve outcomes.
-
What role do international partnerships play in Exelixis's business model?
International partnerships, notably with Ipsen for Europe and Japan, are crucial for global commercialization of Cabometyx, generating significant collaboration revenue and expanding market reach beyond the United States.
Citations
[1] Exelixis, Inc. (2024, February 28). Exelixis Reports Fourth Quarter and Full Year 2023 Financial Results. Retrieved from https://investors.exelixis.com/news-releases/news-release-details/exelixis-reports-fourth-quarter-and-full-year-2023-financial-results