Last updated: January 23, 2026
Executive Summary
Exelixis Inc. is a biotech company specializing in the development and commercialization of targeted therapies for cancer. It is recognized for its flagship drug, cabozantinib, used in treating multiple cancer types, and has a broad portfolio of in-house and partnered products. As of 2023, Exelixis holds a solid position in the oncology market, characterized by its innovative peptide-tyrosine kinase inhibitors (PKTIs) and strategic alliances. This analysis provides a comprehensive overview of Exelixis’ market position, competitive strengths, strategic initiatives, and industry dynamics.
What is Exelixis Inc’s Market Position in Oncology?
| Aspect |
Details |
| Core Focus |
Oncology, targeted therapy, kinase inhibitors |
| Market Cap (2023) |
~$8.5 billion (reflecting market valuation) |
| Revenue (2022) |
$883 million, driven predominantly by cabozantinib sales |
| Key Products |
Cabozantinib (CABOMETYX, COMETRIQ), baitinib, partnership assets |
| Global Footprint |
US, EU, Asia-Pacific, expanding presence in emerging markets |
Revenue Breakdown
- Cabozantinib (US & international): 80%
- Licensing and collaborations: 15%
- Other asset sales & royalties: 5%
Market Share
- U.S. metastatic renal cell carcinoma (mRCC): ~20% (2023 estimates)
- U.S. hepatocellular carcinoma (HCC): 15-20% (market penetration ongoing)
What are the Competitive Strengths of Exelixis?
Robust Product Portfolio & Lifecycle Management
- Cabozantinib: Approved in multiple indications, including mRCC, HCC, metastatic castration-resistant prostate cancer (mCRPC), and thyroid carcinoma. Continuous label expansions enhance longevity.
- Pipeline: Over 10 clinical-stage assets, including cabozantinib derivatives and combination therapies.
Strategic Partnerships & Licensing Agreements
| Partnering Entity |
Asset/Program |
Strategic Impact |
| Ipsen |
Cabometyx (EU) |
Market expansion in Europe |
| Bristol-Myers Squibb |
CABOMETYX + Opdivo combo (clinical) |
Potential combination therapy revenues |
| Taiho Oncology |
Global license on cabozantinib |
Accelerates international growth |
Focused R&D and Clinical Pipeline
- Clinical trials on novel combinations (e.g., cabozantinib plus immune checkpoint inhibitors)
- Investigations into rare cancers and greater indication expansion
Market-Niche Expertise
- Leading positions in rare and challenging cancers where unmet need persists
- Reputation for innovation in kinase inhibitors
What Are the Key Strategic Insights & Initiatives?
1. Expansion of Indications & Label Extensions
Exelixis continues to seek approval for additional indications of cabozantinib, including:
- Gastric cancer
- Endometrial carcinoma
- Other solid tumors
2. Diversification through Pipeline and Partner Collaborations
Emerging assets aim to mitigate reliance on cabozantinib:
- Combinatorial approaches with immunotherapies (e.g., nivolumab, pembrolizumab)
- Next-generation kinase inhibitors targeting different tumor pathways
3. Geographic & Market Penetration
Focused efforts to grow in:
- Europe (through Ipsen licensing)
- Asia-Pacific (through local partnerships)
- Emerging markets with unmet oncology needs
4. Strategic Mergers & Alliances
Potential M&A activities or licensing deals to accelerate innovation and expand global footprint.
5. Digitalization & Precision Oncology
Incorporating biomarker-driven therapy selection to enhance patient outcomes and reduce costs.
How Does Exelixis Compare with Key Competitors?
| Competitor |
Core Focus |
Market Share |
Key Products |
Strengths |
Weaknesses |
| Bristol-Myers Squibb |
Oncology, immuno-oncology |
25% (market) |
Opdivo, Yervoy |
Strong pipeline, global presence |
High competition, patent expiries |
| Merck & Co. |
Oncology, immuno-oncology |
20% |
Keytruda |
Market-leading immunotherapy |
Patent exclusivity risks |
| Bayer AG |
Oncology, cardiovascular, rare diseases |
10% |
Xofigo, Nexavar |
Diversified portfolio |
Limited exclusivity for key assets |
Exelixis Differentiators
- Focused kinase inhibitor portfolio with multiple indications
- Innovation through clinical collaborations
- Strong regional partnerships, particularly in Europe and Asia
What Are the Key Industry Trends Affecting Exelixis?
| Trend |
Implication for Exelixis |
| Increased precision medicine focus |
Enhances value of targeted therapies like cabozantinib |
| Growing importance of combination therapies |
Leverages collaborations with immunotherapy firms |
| Regulatory shifts toward accelerated approval |
Facilitates rapid indication expansion |
| Market pressure for biosimilar entry |
Necessitates innovation in patent protection and R&D investment |
| Digital health and real-world evidence |
Offers opportunities for product differentiation and post-market rollout |
What Is the Regulatory & Patent Landscape?
| Region |
Key Data Singles |
Critical Policy Insights |
| US (FDA) |
Multiple approvals since 2012, ongoing label expansions |
Fast track designation for new indications |
| Europe (EMA) |
EU approval for cabozantinib in 2016 |
Regulatory harmonization advantageous for broader market access |
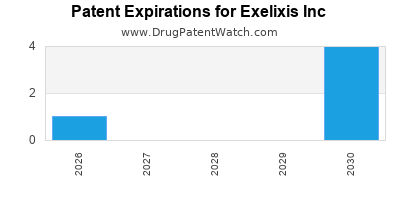
| Patent Expiry |
Key patents expire between 2024-2028 |
Need for strategic innovation and lifecycle planning |
What Are the Main Challenges and Risks?
| Challenge/Risk |
Impact Analysis |
| Patent expirations |
Potential revenue decline post-2028 |
| Competition from biosimilars |
Market share erosion, especially in common indications |
| Clinical trial failures |
R&D costs and pipeline delays |
| Regulatory hurdles |
Delays or denials impacting expansion plans |
Key Takeaways
- Market Position: Exelixis is a focused oncology biotech with core revenue from cabozantinib, established in multiple indications with strong international presence through strategic partnerships.
- Strengths: Deep pipeline, strong R&D, global licensing agreements, and niche market leadership.
- Growth Strategies: Indication expansion, clinical trial innovation, geographic penetration, and partnership expansion.
- Competitive Edge: Targeted kinase inhibitor expertise, combine with immunotherapies, and focus on rare cancers.
- Risks & Challenges: Patent cliffs, competition, regulatory risk, and clinical failures.
FAQs
1. How does Exelixis' flagship drug, cabozantinib, compare to competing kinase inhibitors?
Cabozantinib demonstrates broader kinase inhibition—including MET, VEGFR, and RET—contributing to its efficacy across multiple cancers. Its approval in diverse tumor types and clinical trial success in combinations position it favorably against competitors that target narrower pathways.
2. What are the main indications driving Exelixis revenue today?
Metastatic renal cell carcinoma (mRCC) and hepatocellular carcinoma (HCC) are the primary contributors, accounting for approximately 80% of sales. Other indications like thyroid carcinoma and prostate cancer are expanding.
3. How does Exelixis' partnership strategy influence its market position?
Strategic licensing and collaborations (e.g., with Ipsen, Bristol-Myers Squibb, and Taiho) expand its global reach, accelerate indication approvals, and share R&D costs—vital for competitive advantage amid industry consolidation.
4. What is the outlook for Exelixis in the next 5 years?
Expected to sustain growth through new indications, label extensions, pipeline advancements, and geographic expansion. The expiration of key patents between 2024-2028 poses a risk, but innovation and partnerships are mitigants.
5. What competitive threats does Exelixis face from biosimilars and generics?
Biosimilars targeting key indications like RCC could erode market share. Focused innovation and expanding into less competitive rare cancers remain critical countermeasures.
Citations
- Exelixis Inc. Annual Report 2022.
- MarketWatch. “Exelixis Market Capitalization & Stock Data,” 2023.
- U.S. Food & Drug Administration (FDA). Drug approvals and indication expansions.
- European Medicines Agency (EMA). Approved indications for cabozantinib in Europe.
- IQVIA. Oncology Market Trends Report, 2023.
Key Takeaways
- Exelixis maintains a competitive edge through its targeted oncology portfolio and strategic collaborations.
- Continuous pipeline development and indication expansion are vital for sustained growth.
- Market risks, including patent expirations and biosimilar threats, necessitate innovation and geographic diversification.
- Industry trends favor targeted and combination therapies, aligning well with Exelixis’ expertise.
- Decision-makers should closely monitor regulatory developments and patent landscapes affecting revenue streams.