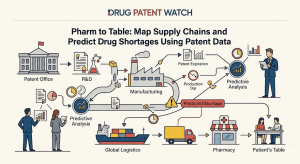
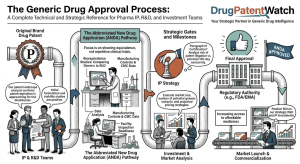
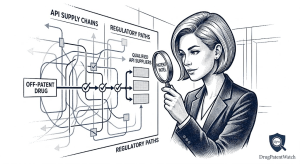
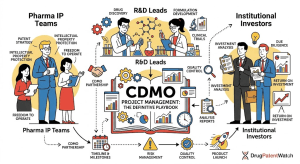
Russia’s Biosimilar Powerhouse: BIOCAD’s Patent Pipeline and the Oncology Disruption No One in the West Is Watching
When Johnson & Johnson’s multiple myeloma franchise Darzalex (daratumumab) generated roughly $10 billion in global revenue in 2024, it stood […]