Generic Drug Development Timelines: The Definitive Playbook for Faster ANDA Approval
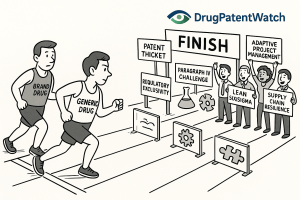
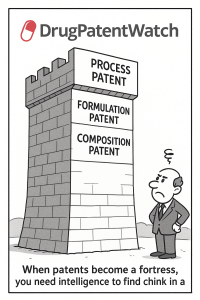
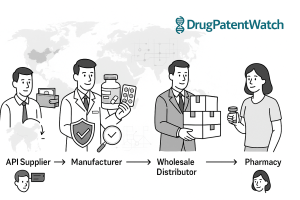
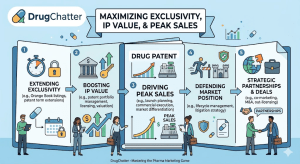
The average generic drug program takes 3 to 4 years from target identification to FDA approval. For most companies, that […]
Generic Drug Development Timelines: The Definitive Playbook for Faster ANDA Approval Read Post »