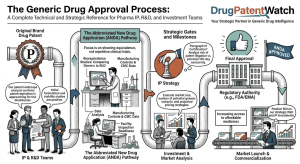
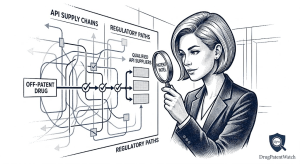
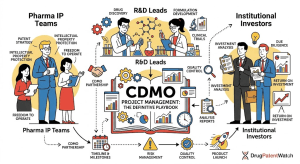
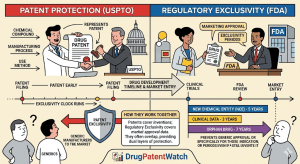
Read the Drug Patent Clock Before the Market Does: A step-by-step technical guide to forecasting pharmaceutical drug patent expiry for IP teams, R&D strategists, and institutional investors
1. Why Patent Expiry Forecasting Is a Revenue-Critical Discipline ‘Between 2025 and 2030, approximately 190 drugs will lose market exclusivity, […]