Last updated: February 19, 2026
INTRODUCTION
Voriconazole, a triazole antifungal agent, plays a critical role in treating serious invasive fungal infections, particularly those caused by Aspergillus species. Its efficacy against a broad spectrum of yeasts and molds has secured its position in the antifungal market. This analysis examines voriconazole's market dynamics, patent landscape, generic competition, and financial projections, providing insights for strategic R&D and investment decisions.
PATENT LANDSCAPE AND GENERIC COMPETITION
The original patent for voriconazole was held by Pfizer Inc. (then Pfizer Limited) and expired in the United States in 2007 and in Europe in 2008. The compound was first approved by the FDA in 2001 and by the EMA in 2002.
- US Patent Expiry: US Patent 4,957,939, filed on June 18, 1987, and granted on September 18, 1990, covered the compound and its use. This patent expired in September 2007.
- European Patent Expiry: Similar patent protections expired in major European markets around the same period.
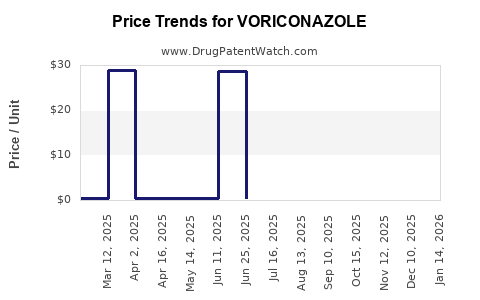
The expiration of these key patents has led to the emergence of numerous generic manufacturers, significantly altering the market landscape. This has resulted in a substantial decrease in the average selling price (ASP) of voriconazole due to increased competition.
Key Generic Entrants
Several pharmaceutical companies have successfully launched generic versions of voriconazole. These include:
- Teva Pharmaceuticals
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Aurobindo Pharma
- Cipla
- Dr. Reddy's Laboratories
- Lupin Limited
The presence of these multiple players intensifies price competition and influences market share distribution.
MARKET SIZE AND GROWTH PROJECTIONS
The global market for voriconazole is substantial, driven by the persistent prevalence of invasive fungal infections, particularly in immunocompromised patient populations (e.g., transplant recipients, cancer patients undergoing chemotherapy, HIV/AIDS patients).
-
Estimated Market Size (2023): The global voriconazole market was estimated to be between $600 million and $800 million in 2023. (Source: Industry reports, market research data)
-
Projected Compound Annual Growth Rate (CAGR): The market is projected to grow at a CAGR of approximately 3% to 5% from 2024 to 2030.
-
Drivers of Growth:
- Increasing incidence of invasive fungal infections.
- Growing number of immunocompromised patients globally.
- Expansion of healthcare access in emerging economies.
- Continued use of voriconazole as a first-line or salvage therapy due to its established efficacy.
-
Challenges to Growth:
- Intense price pressure from generic competition.
- Development of newer antifungal agents with potentially improved profiles (e.g., broader spectrum, fewer drug interactions, novel mechanisms of action).
- Emergence of antifungal resistance.
Geographic Market Distribution
The market is segmented by geography, with North America and Europe historically being the largest markets due to advanced healthcare infrastructure and higher prevalence of hospital-acquired infections. Asia-Pacific is expected to exhibit the highest growth rate driven by increasing healthcare expenditure and a rising patient pool.
THERAPEUTIC APPLICATIONS AND INDICATIONS
Voriconazole is indicated for the treatment of the following serious fungal infections:
- Invasive Aspergillosis: This is a primary indication, where voriconazole is often the treatment of choice.
- Candidemia: Including candidemia in non-neutropenic patients and candidemia with extension to other sites.
- Esophageal Candidiasis: Treatment of candidiasis of the esophagus.
- Invasive Infections due to Scedosporium apiospermum and Fusarium spp.: This includes infections in patients intolerant of or refractory to other therapies.
Dosing and Administration
Voriconazole is available in oral (tablets and oral suspension) and intravenous (IV) formulations.
- Oral Dosage Forms: Typically contain 50 mg and 200 mg strengths.
- IV Dosage Form: Available in vials for reconstitution.
- Standard Dosing Regimen: A typical regimen involves an initial loading dose followed by a maintenance dose, adjusted based on patient weight, renal function, and co-administered medications.
PRICING AND REIMBURSEMENT
The pricing of voriconazole has been significantly impacted by generic entry. Branded voriconazole (Vfend, Pfizer) commanded premium pricing prior to patent expiry. Generic versions are now available at a substantially lower cost.
- Branded Voriconazole ASP (Pre-Patent Expiry): Historically, branded voriconazole's ASP was several hundred dollars per course of treatment.
- Generic Voriconazole ASP: The ASP for generic voriconazole typically ranges from $10 to $50 per day of treatment, depending on formulation, dosage, and geographic market.
Reimbursement policies vary by country and healthcare system. In countries with established national health services, pricing is often subject to formulary negotiations and cost-effectiveness assessments. In the U.S., private payers and Medicare/Medicaid determine reimbursement rates. The availability of multiple generic options generally leads to favorable reimbursement terms for payers.
MANUFACTURING AND SUPPLY CHAIN
The manufacturing of voriconazole involves complex chemical synthesis. Key active pharmaceutical ingredient (API) manufacturers and finished dosage form producers operate globally.
- API Production: China and India are significant hubs for API manufacturing due to cost advantages and established chemical synthesis capabilities.
- Finished Dosage Form Production: Companies globally produce the final tablets, suspensions, and IV formulations.
- Supply Chain Considerations: Robust quality control and regulatory compliance are paramount to ensure product safety and efficacy. Supply chain disruptions, such as those experienced during global pandemics, can impact availability and pricing.
POTENTIAL FOR INNOVATION AND MARKET DIFFERENTIATION
Despite the mature generic market, opportunities for innovation and differentiation exist:
- Improved Formulations: Development of more convenient dosing regimens, improved palatability for oral suspensions, or formulations with reduced drug-drug interaction potential.
- Combination Therapies: Exploring synergistic effects of voriconazole with other antifungal agents or immunomodulators.
- Resistance Management: Research into mechanisms of resistance and strategies to overcome them.
- Pediatric Formulations: Development of formulations specifically tailored for pediatric patients, addressing challenges in dosing and administration.
- Biosimil/Biobetter Development: While voriconazole is a small molecule, the concept of developing optimized versions with improved pharmacokinetic or pharmacodynamic profiles remains an area of interest in the broader pharmaceutical landscape.
FINANCIAL TRAJECTORY AND INVESTMENT CONSIDERATIONS
The financial trajectory of voriconazole is characterized by declining ASPs due to generic competition, offset by stable or modest volume growth.
- Revenue Streams: Primarily from generic sales. Branded sales are minimal and restricted to regions with ongoing market exclusivity or specific market niches.
- Profitability: Margins for generic manufacturers are generally lower compared to the branded era but can be sustained through high-volume production and efficient cost management.
- Investment Opportunities:
- Generic Manufacturers: Companies with strong manufacturing capabilities, efficient supply chains, and established distribution networks can achieve sustainable profitability.
- API Suppliers: Reliable suppliers of high-quality voriconazole API are essential components of the value chain.
- Specialty Pharma: Companies focusing on niche indications, improved formulations, or pediatric applications may find opportunities for differentiated products and potentially higher profit margins.
- Risks:
- Price Erosion: Continued intensification of generic competition can further depress ASPs.
- Regulatory Changes: Shifts in pricing regulations or reimbursement policies can impact profitability.
- Emergence of Superior Therapies: New antifungal agents with better efficacy or safety profiles could displace voriconazole.
- Antifungal Resistance: The development of widespread resistance could limit voriconazole's utility.
Pfizer's Vfend Performance (Historical Example)
Prior to patent expiry, Pfizer's Vfend (voriconazole) was a significant revenue driver.
- Peak Sales: Vfend achieved peak annual sales exceeding $1 billion in the mid-2010s.
- Post-Patent Expiry Impact: Sales declined sharply following generic launches, with the brand largely relegated to niche markets or specific payer agreements. This illustrates the dramatic financial shift occurring upon patent expiration in the pharmaceutical industry.
KEY TAKEAWAYS
- Voriconazole's patent protection has expired in major markets, leading to a highly competitive generic landscape.
- The market is driven by the persistent need for effective antifungal treatments in immunocompromised populations.
- Despite price erosion, the global voriconazole market is projected to experience modest growth due to increasing incidence of fungal infections and expanding healthcare access.
- Key growth drivers include the rising number of immunocompromised patients and the established efficacy of voriconazole.
- Challenges include intense price competition, the threat of newer antifungal agents, and the potential for antifungal resistance.
- Opportunities for innovation exist in improved formulations, combination therapies, and pediatric applications.
- Investment considerations should focus on generic manufacturers with efficient operations, reliable API suppliers, and specialty companies pursuing differentiated products.
FAQS
1. What is the primary indication for voriconazole, and what patient populations benefit most?
Voriconazole's primary indication is invasive aspergillosis. Patient populations that benefit most include immunocompromised individuals such as organ transplant recipients, patients undergoing chemotherapy, and those with HIV/AIDS, who are at higher risk of developing severe fungal infections.
2. How has the expiration of voriconazole's patents affected its pricing and market competition?
The expiration of voriconazole's patents has resulted in a significant increase in generic competition. This has led to a substantial decrease in the average selling price (ASP) of the drug and intensified price competition among numerous manufacturers.
3. What are the projected growth rates for the global voriconazole market, and what factors are driving this growth?
The global voriconazole market is projected to grow at a compound annual growth rate (CAGR) of approximately 3% to 5% from 2024 to 2030. Growth is driven by the increasing incidence of invasive fungal infections, a rising global population of immunocompromised patients, and expanding healthcare access in emerging economies.
4. Are there any significant side effects or drug interactions associated with voriconazole that impact its use?
Yes, voriconazole can cause significant side effects, including visual disturbances, hepatotoxicity, and skin reactions. It also has a high potential for drug-drug interactions, particularly with CYP450 enzyme inhibitors and inducers, requiring careful patient monitoring and medication review.
5. What are the main challenges facing the voriconazole market today, and what is the outlook for newer antifungal agents?
The main challenges include extreme price erosion due to generic competition, the potential for increasing antifungal resistance, and the development of newer antifungal agents with potentially broader spectra of activity, improved safety profiles, or novel mechanisms of action that could displace voriconazole in certain indications.
CITATIONS
[1] Pfizer Inc. (1990). United States Patent 4,957,939. U.S. Patent and Trademark Office.
[2] European Medicines Agency. (n.d.). Voriconazole. Retrieved from emea.europa.eu
[3] U.S. Food and Drug Administration. (2001). FDA Approves Vfend (voriconazole) for Treatment of Serious Fungal Infections. [Press Release].
[4] Various Pharmaceutical Industry Market Research Reports (2023-2024). Data on file.