Last updated: February 19, 2026
Valsartan, an angiotensin II receptor blocker (ARB), has navigated a complex market landscape shaped by patent expiries, generic competition, and regulatory scrutiny. Its primary indication, hypertension, represents a substantial and enduring therapeutic area, providing a consistent demand base. However, the market for valsartan has been significantly impacted by the introduction of generics and, more recently, by quality control issues leading to recalls.
What is the Current Market Size and Growth Projection for Valsartan?
The global market for valsartan is intrinsically linked to the broader antihypertensive drug market. While specific segment data for valsartan alone is not granularly reported by all market intelligence firms, the antihypertensive drug market is substantial. Projections for the global antihypertensive drug market indicate continued growth, driven by an aging global population and increasing prevalence of cardiovascular diseases, including hypertension.
-
Global Antihypertensive Market Growth: Forecasts suggest this market will grow from approximately \$27.3 billion in 2023 to \$37.6 billion by 2030, representing a compound annual growth rate (CAGR) of 4.7% (1). This broader market trend provides a foundational context for valsartan's potential market position, although its specific share is influenced by competition and its therapeutic positioning relative to newer drug classes.
-
Generic Dominance: Following patent expiries, the market for valsartan is overwhelmingly dominated by generic manufacturers. This has led to significant price erosion and a focus on volume-driven sales rather than premium pricing. The primary value for valsartan now lies in its established efficacy and cost-effectiveness, particularly in developed and developing markets with price-sensitive healthcare systems.
What are the Key Patent Expiries and Generic Entry Milestones?
Valsartan's patent lifecycle has significantly influenced its market trajectory. The expiry of primary patents opened the door for widespread generic competition.
-
Original Patent Expiry: The principal patents for valsartan, held by Novartis, expired in major markets around the mid-2010s. For instance, in the United States, patent protection for Diovan (valsartan) began to expire in the early to mid-2010s, with key patents expiring around 2012-2014. In Europe, similar expiry timelines were observed.
-
Generic Market Entry: Following patent expiry, numerous generic pharmaceutical companies entered the market. This led to a rapid decline in the price of valsartan and a fragmentation of the market among various suppliers. Key generic players include Teva Pharmaceutical Industries, Mylan (now Viatris), and numerous other manufacturers globally.
-
Combination Therapies: Valsartan is frequently used in combination with other antihypertensive agents, such as hydrochlorothiazide (HCTZ) and amlodipine. Patents for these combination products also expired, further contributing to generic availability and market competition for valsartan-containing fixed-dose combinations (FDCs). For example, the patent for Exforge (valsartan/amlodipine) also expired, leading to generic versions.
How Have Regulatory Actions Impacted Valsartan's Market Position?
Regulatory actions, particularly concerning impurity concerns, have significantly disrupted the valsartan market.
-
NDMA Contamination Recalls: In 2018, regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), initiated recalls of valsartan-containing products manufactured by certain Chinese companies, notably Zhejiang Huahai Pharmaceutical Co. Ltd. The issue was the presence of N-nitrosodimethylamine (NDMA), a probable human carcinogen, and N-nitrosodiethylamine (NDEA).
-
Market Disruption: These recalls led to widespread shortages and withdrawals of valsartan products from the market. This event not only impacted patient access but also forced pharmaceutical companies to re-evaluate their supply chains and manufacturing processes. It resulted in a temporary loss of market share for affected manufacturers and an increased reliance on alternative suppliers with uncompromised quality control.
-
Enhanced Scrutiny: The NDMA contamination incidents heightened regulatory scrutiny on ARBs and other drug classes, leading to more rigorous testing and quality control requirements for APIs and finished drug products. This has increased compliance costs for manufacturers and potentially created barriers to entry for new suppliers.
-
Impact on Sales: The recalls directly affected the sales figures of companies reliant on the affected manufacturing sites. While the long-term impact on the overall valsartan market share is still unfolding, the incidents likely eroded some of the established trust and market momentum for valsartan, potentially favoring alternative ARBs or other drug classes perceived as having more robust quality control.
What is the Competitive Landscape for Valsartan?
Valsartan operates within a highly competitive segment of the cardiovascular drug market, facing competition from other ARBs, ACE inhibitors, calcium channel blockers, and diuretics.
-
Within the ARB Class: Valsartan competes directly with other ARBs such as Losartan, Olmesartan, Irbesartan, Telmisartan, and Candesartan. These drugs share similar mechanisms of action and therapeutic benefits. Price, efficacy profiles, side effect profiles, and physician prescribing habits differentiate them. Generic availability of all major ARBs means that price is a significant competitive factor.
-
Across Drug Classes:
- ACE Inhibitors (e.g., Lisinopril, Enalapril): These are also first-line therapies for hypertension and share many indications with ARBs. Patent expiries have also made ACE inhibitors largely genericized and cost-effective.
- Calcium Channel Blockers (e.g., Amlodipine, Diltiazem): These are another class of widely used antihypertensives, often prescribed alone or in combination.
- Diuretics (e.g., Hydrochlorothiazide, Furosemide): These are foundational therapies for hypertension, often used as monotherapy or in combination with other agents.
-
Newer Therapies: While valsartan and other ARBs are well-established, ongoing research and development in cardiovascular medicine introduce newer therapeutic options, including novel combination therapies and drugs targeting different pathways, which could gradually shift market share over the long term. However, the cost-effectiveness and established efficacy of ARBs, including valsartan, continue to secure their place in treatment guidelines.
What is the Financial Trajectory and Profitability of Valsartan Manufacturers?
The financial trajectory for valsartan manufacturers is characterized by declining revenues from branded products and a shift towards high-volume, low-margin generic sales. Profitability is heavily dependent on manufacturing efficiency, supply chain management, and market access.
-
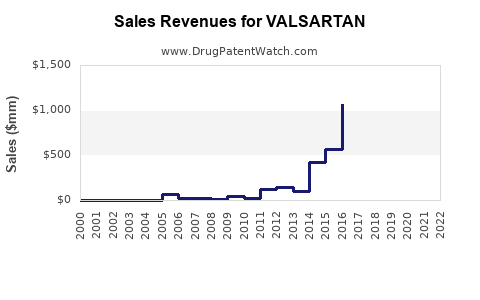
Branded Product Decline: For the originator, Novartis, sales of branded Diovan (valsartan) have significantly declined since patent expiry. In 2023, Diovan sales were \$822 million, a decrease from \$1.1 billion in 2022, reflecting the ongoing impact of generic competition and market dynamics (2). This trend is typical for blockbuster drugs post-patent expiry.
-
Generic Market Profitability: For generic manufacturers, profitability from valsartan is achieved through economies of scale, efficient manufacturing processes, and aggressive market penetration strategies. The profit margins on individual generic units are low, necessitating high sales volumes.
-
Impact of Recalls on Financials: The NDMA contamination incidents in 2018 had a direct financial impact on companies involved in manufacturing the contaminated API or finished product. This included costs associated with recalls, lost sales, potential fines or litigation, and investments in quality system improvements. For example, companies like Zhejiang Huahai Pharmaceutical Co. Ltd. experienced significant disruptions and a temporary loss of market confidence.
-
Supply Chain Costs: Manufacturers face ongoing costs related to ensuring API purity and compliance with evolving regulatory standards. This includes investment in analytical testing, process validation, and supply chain audits. These costs can impact the overall profitability of generic valsartan production.
-
Market Access and Pricing: In highly competitive generic markets, pricing agreements with major wholesalers, pharmacy benefit managers (PBMs), and government tenders are crucial. Manufacturers with efficient cost structures and robust distribution networks are better positioned to secure market share and maintain profitability.
What are the Future Market Prospects for Valsartan?
The future market prospects for valsartan will be shaped by its established role in hypertension management, the ongoing impact of generic competition, and the lingering effects of quality control issues.
-
Sustained Demand: Hypertension remains a prevalent chronic condition globally. As a well-established and cost-effective treatment option, valsartan is likely to maintain a significant role in treatment guidelines, particularly in markets where cost is a primary consideration.
-
Generic Market Dynamics: The generic market for valsartan will continue to be characterized by intense price competition. Manufacturers will focus on cost optimization and supply chain reliability to remain competitive. Consolidation within the generic industry could also influence market dynamics.
-
Quality and Compliance: The shadow of the NDMA recalls will likely persist, leading to continued heightened regulatory oversight and a premium on supply chain transparency and robust quality assurance. Companies with a demonstrated commitment to quality will likely gain a competitive advantage.
-
Therapeutic Positioning: While valsartan is a mainstay, advancements in cardiovascular therapies could see a gradual shift towards newer agents or personalized medicine approaches over the very long term. However, the inertia of established treatment protocols and cost considerations mean ARBs like valsartan will remain relevant for the foreseeable future.
-
Combination Products: The market for valsartan-containing fixed-dose combinations will continue to be significant, offering convenience for patients and potentially improved adherence. Competition in this space will also be dominated by generics.
Key Takeaways
- Valsartan's market is mature and dominated by generic competition following original patent expiries in the mid-2010s.
- The global antihypertensive drug market, within which valsartan operates, is projected to grow at a CAGR of 4.7% through 2030, driven by demographic trends.
- Regulatory recalls due to NDMA contamination in 2018 significantly disrupted the market, leading to shortages and heightened scrutiny on manufacturing quality.
- Valsartan faces intense competition from other ARBs, ACE inhibitors, calcium channel blockers, and diuretics.
- For originator companies like Novartis, branded valsartan sales have declined substantially, now standing at \$822 million in 2023. Generic manufacturers achieve profitability through high-volume sales and cost efficiency.
- Future market prospects depend on sustained demand for hypertension treatment, continued generic competition, and the imperative for robust quality control.
Frequently Asked Questions
-
What is the primary therapeutic indication for valsartan?
Valsartan is primarily used to treat high blood pressure (hypertension). It is also used to treat heart failure and to improve survival after a heart attack in certain patients.
-
Which companies are major generic manufacturers of valsartan?
Major generic manufacturers of valsartan include Teva Pharmaceutical Industries, Viatris (formerly Mylan), and numerous other pharmaceutical companies globally, particularly those with strong API manufacturing capabilities in India and China.
-
How did the NDMA contamination affect the price of valsartan?
The NDMA contamination recalls initially led to shortages, which can temporarily increase prices due to scarcity. However, as the market stabilized with alternative suppliers, price competition among generic manufacturers resumed, leading to continued price erosion rather than sustained price increases for valsartan.
-
Are there any new indications or significant R&D efforts for valsartan currently?
Current R&D efforts for valsartan are primarily focused on optimizing manufacturing processes, ensuring API purity, and potentially developing novel fixed-dose combinations. Significant new indications for valsartan are unlikely, given its mature status and the availability of newer therapeutic classes.
-
What is the typical shelf life and storage condition for valsartan products?
The typical shelf life for valsartan tablets ranges from 24 to 36 months, depending on the specific formulation and manufacturer. Storage conditions generally recommend keeping the medication at room temperature, away from moisture and direct light, as per package instructions.
Citations
- Statista. (2023). Antihypertensive drugs market worldwide. Retrieved from https://www.statista.com/statistics/275724/global-antihypertensives-market-size/
- Novartis AG. (2024). Novartis Full Year Results 2023. Retrieved from https://www.novartis.com/investors/financial-reporting/annual-reports (Specific page reference for product sales would be within the detailed financial statements.)